Abstract
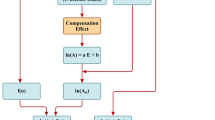
Kelsey and Clarke1 have recently discussed the variation with temperature of the integral heat of sorption of water by wood (ΔH), and its bearing on the specific heat (cm) of wood containing m gm. of moisture per gm. of dry wood. Empirically, it is found that cm is greater than the specific heat (c) calculated from the specific heats and proportions of the components, assuming a simple mixture law2. This effect is not confined to cellulosic materials, but has been observed on starch3, gelatin4, dried foods5 and other hygroscopic substances. The suggestion has previously been made5 that the excess specific heat could be accounted for in terms of the change in ΔH with temperature (T); Kelsey and Clarke quote the appropriate thermodynamic equation as  where cw is the specific heat of the water, and c0 the specific heat of the dry wood. Putting cw = 1, and noting that
where cw is the specific heat of the water, and c0 the specific heat of the dry wood. Putting cw = 1, and noting that  then
then  where Δc is the excess specific heat.
where Δc is the excess specific heat.
Similar content being viewed by others
Article PDF
References
Kelsey, K. E., and Clarke, L. N., Nature, 176, 83 (1955).
Forest Products Research 1953 (H.M.S.O., London, 1954).
Freeman, M. E., Arch. Biochem., 1, 27 (1942).
Hampton, W. F., and Mennie, J. H., Canad. J. Res., 10, 452 (1934). Horn, W. R., and Mennie, J. H., ibid., 12, 702 (1935).
Stitt, F., and Kennedy, E. K., Food Res., 10, 426 (1945). Wahba, M., and Nashed, S., Nature, 166, 998 (1950).
Forest Products Research 1954 (H.M.S.O., London, 1955).
Kull, W., Holz als Roh- u. Werkst., 12, 413 (1954).
Dunlap, F., U.S. Dep. Agric. For. Serv. Bull. No. 110 (1912).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
HEARMON, R., BURCHAM, J. Specific Heat and Heat of Wetting of Wood. Nature 176, 978 (1955). https://doi.org/10.1038/176978a0
Issue Date:
DOI: https://doi.org/10.1038/176978a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.