Abstract
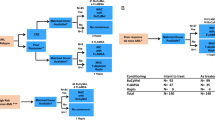
We performed a survey of the European Cooperative Group for Blood and Marrow Transplantation to analyze the outcome of 625 acute promyelocytic leukemia (APL) patients transplanted with auto- or allogeneic-hematopoietic stem cell transplantation (autoHSCT, alloHSCT) after 1993, in first (CR1) or in second complete remission (CR2). Leukemia-free survival (LFS) at 5 years in CR1 was 69% for 149 patients autografted and 68% for 144 patients allografted, whereas in CR2, LFS was 51% in 195 autoHSCT and 59% in 137 alloHSCT recipients, respectively. In the group of autoHSCT for CR1 (n=149), higher relapse incidence (RI) was associated with shorter time from diagnosis to transplant (<7.6 months); transplant-related mortality (TRM) was increased in older patients (>47 years), whereas for CR2, longer time from diagnosis to transplant (>18 months) was associated with increased LFS and decreased RI. In the alloHSCT group for CR1 (n=144), age (<33 years) was associated with increased LFS and decreased TRM and for CR2 (n=137), the use of mobilized peripheral blood stem cells was associated with decreased TRM. Female recipient, a female donor to male recipient and transplants performed before 1997 were associated with decreased RI. In conclusion, HSCT still appears to have a role in APL, especially for patients in CR2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Mandelli F, Diverio D, Avvisati G, Luciano A, Barbui T, Bernasconi C et al. Molecular remission in PML/RAR alpha-positive acute promyelocytic leukemia by combined all-trans retinoic acid and idarubicin (AIDA) therapy. Gruppo Italiano-Malattie Ematologiche Maligne dell'Adulto and Associazione Italiana di Ematologia ed Oncologia Pediatrica Cooperative Groups. Blood 1997; 90: 1014–1021.
Asou N, Adachi K, Tamura J, Kanamaru A, Kageyama S, Hiraoka A et al. Analysis of prognostic factors in newly diagnosed acute promyelocytic leukemia treated with all-trans retinoic acid and chemotherapy. Japan Adult Leukemia Study Group. J Clin Oncol 1998; 16: 78–85.
Burnett AK, Grimwade D, Solomon E, Wheatley K, Goldstone AH . Presenting white blood cell count and kinetics of molecular remission predict prognosis in acute promyelocytic leukemia treated with all- trans retinoic acid: result of the Randomized MRC Trial. Blood 1999; 93: 4131–4143.
Fenaux P, Chastang C, Chevret S, Sanz M, Dombret H, Archimbaud E et al. A randomized comparison of all transretinoic acid (ATRA) followed by chemotherapy and ATRA plus chemotherapy and the role of maintenance therapy in newly diagnosed acute promyelocytic leukemia. The European APL Group. Blood 1999; 94: 1192–1200.
Sanz MA, Martin G, Rayon C, Esteve J, González M, Díaz-Mediavilla J et al. A modified AIDA protocol with anthracycline-based consolidation results in high antileukemic efficacy and reduced toxicity in newly diagnosed PML/RARalpha-positive acute promyelocytic leukemia. PETHEMA group. Blood 1999; 94: 3015–3021.
Lengfelder E, Reichert A, Schoch C, Haase D, Haferlach T, Loffler H et al. Double induction strategy including high dose cytarabine in combination with all-trans retinoic acid: effects in patients with newly diagnosed acute promyelocytic leukemia. German AML Cooperative Group. Leukemia 2000; 14: 1362–1370.
Sanz MA, Martín G, González M, León A, Rayón C, Rivas C et al. Risk-adapted treatment of acute promyelocytic leukemia with all-trans retinoic acid and anthracycline monochemotherapy: a multicenter study by the PETHEMA Group. Blood 2004; 103: 1237–1243.
Diverio D, Rossi V, Avvisati G, De Santis S, Pistilli A, Pane F et al. Early detection of relapse by prospective reverse transcriptase–polymerase chain reaction analysis of the PML/RARalpha fusion gene in patients with acute promyelocytic leukemia enrolled in the GIMEMA-AIEOP multicenter ‘AIDA’ trial. GIMEMA-AIEOP Multicenter ‘AIDA’ Trial. Blood 1998; 92: 784–789.
Meloni G, Diverio D, Vignetti M, Avvisati G, Capria S, Petti MC et al. Autologous bone marrow transplantation for acute promyelocytic leukemia in second remission: prognostic relevance of pretransplant minimal residual disease assessment by reverse-transcription polymerase chain reaction of the PML/RAR alpha fusion gene. Blood 1997; 90: 1321–1325.
Lo Coco F, Diverio D, Avvisati G, Petti MC, Meloni G, Pogliani EM et al. Therapy of molecular relapse in acute promyelocytic leukemia. Blood 1999; 94: 2225–2229.
Thomas X, Dombret H, Cordonnier C, Pigneux A, Gardin C, Guerci A et al. Treatment of relapsing acute promyelocytic leukemia by all-trans retinoic acid therapy followed by timed sequential chemotherapy and stem cell transplantation. APL Study Group. Acute promyelocytic leukemia. Leukemia 2000; 14: 1006–1013.
Roman J, Martin C, Torres A, Jimenez MA, Andrés P, Flores R et al. Absence of detectable PML-RAR alpha fusion transcripts in long-term remission patients after BMT for acute promyelocytic leukemia. Bone Marrow Transplant 1997; 19: 679–683.
de Botton S, Fawaz A, Chevret S, Dombret H, Thomas X, Sanz M et al. Autologous and allogeneic stem-cell transplantation as salvage treatment of acute promyelocytic leukemia initially treated with all-trans-retinoic acid: a retrospective analysis of the European acute promyelocytic leukemia group. J Clin Oncol 2005; 23: 120–126.
Mandelli F, Labopin M, Granena A, Iriondo A, Prentice G, Bacigalupo A et al. European survey of the role of bone marrow transplantation in acute promyelocytic leukemia (M3). Working Party on Acute Leukemia of the European Cooperative Group for Bone Marrow Transplantation (EBMT). Bone Marrow Transplant 1994; 14: 293–298.
Bennet JM, Catovsky D, Daniel MT, Flandrin G, Galton D, Gralnick H et al. Proposals for the classification of the acute leukemias. Br J Haematol 1976; 33: 451–458.
Kaplan EL, Meier P . Non parametric estimation from incomplete observations. J Am Stat Assoc 1958; 53: 457–481.
Cox DR . Regression models and life tables. J R Stat Soc 1972; 34: 187–202.
Fine JP, Gray RJ . A proportional hazard model for the subdistribution of a competing risk. JASA 1999; 94: 496–509.
Nabhan C, Mehta J, Tallman MS . The role of bone marrow transplantation in acute promyelocytic leukemia. Bone Marrow Transplant 2001; 28: 219–226.
Bensinger WI, Martin PJ, Storer B, Clift R, Forman SJ, Negrin R et al. Transplantation of bone marrow as compared with peripheral blood cells from HLA-identical relatives in patients with hematologic cancers. N Engl J Med 2001; 344: 175–181.
Blaise D, Kuentz M, Fortainer C, Bourhis JH, Milpied N, Sutton L et al. Randomized trial of bone marrow versus lenograstim-primed blood cell allogeneic transplantation in patients with early-stage leukemia: a report from the Société Française de Greffe de Moelle. J Clin Oncol 2000; 18: 537–546.
Heldal D, Tjonnfjord G, Brinch L, Albrechtsen D, Egeland T, Steen R et al. A randomised study of allogeneic transplantation with stem cells from blood or bone marrow. Bone Marrow Transplant 2000; 25: 1129–1136.
Powles R, Mehta J, Kulkarni S, Treleaven J, Millar B, Marsden J et al. Allogeneic blood and bone marrow stem-cell transplantation in hematological malignant diseases. A randomized trial. Lancet 2000; 355: 1231–1237.
Schmitz N, Linch DC, Dreger P, Goldstone AH, Boogaerts MA, Ferrant A et al. Randomised trial of filgrastim-mobilised peripheral blood progenitor cell transplantation versus autologous bone marrow transplantation in lymphoma patients. Lancet 1996; 347: 353–357.
Vigorito AC, Azevedo WM, Marques JFC, Azevedo AM, Eid KA, Aranha FJ et al. A randomised, prospective comparison of allogeneic bone marrow and peripheral blood progenitor cell transplantation in the treatment of haematological malignancies. Bone Marrow Transplant 1998; 22: 1145–1151.
Couban S, Simpson DR, Barnett MJ, Bredeson C, Hubesch L, Howson-Jan K et al. A randomized multicenter comparison of bone marrow and peripheral blood in recipients of matched sibling allogeneic transplants for myeloid malignancies. Blood 2002; 100: 1525–1531.
Champlin RE, Schmitz N, Horowitz M, Chapuis B, Chopra R, Cornelissen JJ et al. Blood stem cells compared with bone marrow as source of hematopoietic cells for allogeneic transplantation. Blood 2000; 95: 3702–3709.
Randolph SS, Gooley TA, Warren EH, Appelbaum FR, Riddell SR . Female donors contribute to a selective graft-versus-leukemia effect in male recipients of HLA-matched, related hematopoietic stem cell transplants. Blood 2004; 103: 347–352.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Sanz, M., Labopin, M., Gorin, NC. et al. Hematopoietic stem cell transplantation for adults with acute promyelocytic leukemia in the ATRA era: a survey of the European Cooperative Group for Blood and Marrow Transplantation. Bone Marrow Transplant 39, 461–469 (2007). https://doi.org/10.1038/sj.bmt.1705620
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705620
Keywords
This article is cited by
-
Hematopoietic stem cell transplantation for adults with relapsed acute promyelocytic leukemia in second complete remission
Bone Marrow Transplantation (2021)
-
Autologous hematopoietic cell transplantation for acute myeloid leukemia in adults: 25 years of experience in Japan
International Journal of Hematology (2020)
-
Acute Promyelocytic Leukemia: A History over 60 Years—From the Most Malignant to the most Curable Form of Acute Leukemia
Oncology and Therapy (2019)
-
Management of patients with acute promyelocytic leukemia
Leukemia (2018)