Summary:
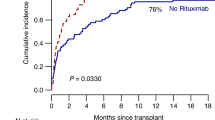
The use of recombinant human erythropoietin (rHuEPO) has been controversial after myeloablative allogeneic Stem cell transplantation (allo-SCT). Reduced intensity conditioning regimens (RIC) offer a novel approach that might translate into a different profile of erythropoietic recovery. We treated 20 consecutive patients with rHuEPO early after matched sibling RIC allo-SCT. Conditioning included fludarabine, busulfan and antithymocyte globulin. EPO treatment was analyzed in terms of toxicity, impact on the frequency of Red blood cell transfusions (RBCT) and kinetics of Hemoglobin recovery within the 60 days post-allo-SCT. Results were compared with 27 matched patients who did not receive rHuEPO. In the first 2 months after allo-SCT all patients receiving rHuEPO (100%) achieved an Hb level>11 g/dl at a median of 30 (15–35) days post-allo-SCT, as compared to only 63% of the patients not receiving rHuEPO (P=0.007) at a median of 35 (20–55) days (P=0.03). A total of 70% (95% CI, 50–90) of rHuEPO patients maintained an Hb over 11 g/dl in the second month as compared to only 19% (95% CI, 4–34) in the other group (P=0.0004). For patients receiving RBCT, the use of rHuEPO was associated with a trend towards reduced RBCT requirements. This pilot study suggests a potential benefit of early administration of rHuEPO after RIC allo-SCT on early erythropoietic recovery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Miller CB, Lazarus HM . Erythropoietin in stem cell transplantation. Bone Marrow Transplant 2001; 27: 1011–1016.
Ludwig H . Epoetin in cancer-related anaemia. Nephrol Dial Transplant 1999; 14 (Suppl. 2): 85–92.
Kasper C . Recombinant human erythropoietin in the treatment of anemic patients with hematological malignancies. Ann Hematol 2001; 80: 319–329.
Bosi A, Vannucchi AM, Grossi A et al. Inadequate erythropoietin production in allogeneic bone marrow transplant patients. Haematologica 1991; 76: 280–284.
Miller CB, Jones RJ, Zahurak ML et al. Impaired erythropoietin response to anemia after bone marrow transplantation. Blood 1992; 80: 2677–2682.
Steegmann JL, Lopez J, Otero MJ et al. Erythropoietin treatment in allogeneic BMT accelerates erythroid reconstitution: results of a prospective controlled randomized trial. Bone Marrow Transplant 1992; 10: 541–546.
Klaesson S, Ringden O, Ljungman P et al. Treatment with erythropoietin after allogeneic bone marrow transplantation: a randomized, double-blind study. Transplant Proc 1994; 26: 1827–1828.
Biggs JC, Atkinson KA, Booker V et al. Prospective randomised double-blind trial of the in vivo use of recombinant human erythropoietin in bone marrow transplantation from HLA-identical sibling donors. The Australian Bone Marrow Transplant Study Group. Bone Marrow Transplant 1995; 15: 129–134.
Link H, Boogaerts MA, Fauser AA et al. A controlled trial of recombinant human erythropoietin after bone marrow transplantation. Blood 1994; 84: 3327–3335.
Bokemeyer C, Aapro MS, Courdi A et al. EORTC guidelines for the use of erythropoietic proteins in anaemic patients with cancer. Eur J Cancer 2004; 40: 2201–2216.
Slavin S, Nagler A, Naparstek E et al. Nonmyeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and nonmalignant hematologic diseases. Blood 1998; 91: 756–763.
Giralt S, Estey E, Albitar M et al. Engraftment of allogeneic hematopoietic progenitor cells with purine analog-containing chemotherapy: harnessing graft-versus-leukemia without myeloablative therapy. Blood 1997; 89: 4531–4536.
Storb RF, Champlin R, Riddell SR et al. Non-myeloablative transplants for malignant disease. Hematology (Am Soc Hematol Educ Program) 2001; 98: 375–391.
Djulbegovic B, Seidenfeld J, Bonnell C, Kumar A . Nonmyeloablative allogeneic stem-cell transplantation for hematologic malignancies: a systematic review. Cancer Control 2003; 10: 17–41.
Blaise D, Bay JO, Faucher C et al. Reduced-intensity preparative regimen and allogeneic stem cell transplantation for advanced solid tumors. Blood 2004; 103: 435–441.
Weissinger F, Sandmaier BM, Maloney DG et al. Decreased transfusion requirements for patients receiving nonmyeloablative compared with conventional peripheral blood stem cell transplants from HLA-identical siblings. Blood 2001; 98: 3584–3588.
Ivanov V, Faucher C, Mohty M et al. Decreased RBCTs after reduced intensity conditioning allogeneic stem cell transplantation: predictive value of prior Hb level. Transfusion 2004; 44: 501–508.
Klink A, Kasper C, Scholl S et al. First report of a prospective randomised controlled trial of recombinant human erythropoietin after allogenic blood stem cell transplantation. Blood 2004; 104: P1833.
Glucksberg H, Storb R, Fefer A et al. Clinical manifestations of graft-versus-host disease in human recipients from HL-A-matched sibling donors. Transplantation 1974; 18: 295–304.
Przepiorka D, Weisdorf D, Martin P et al. 1994 Consensus Conference on acute GVHD grading. Bone Marrow Transplant 1995; 15: 825–828.
Crawford J, Cella D, Cleeland CS et al. Relationship between changes in hemoglobin level and quality of life during chemotherapy in anemic cancer patients receiving epoetin alfa therapy. Cancer 2002; 95: 888–895.
Bennett CL, Luminari S, Nissenson AR et al. Pure red-cell aplasia and epoetin therapy. N Engl J Med 2004; 351: 1403–1408.
Beguin Y, Clemons GK, Oris R, Fillet G . Circulating erythropoietin levels after bone marrow transplantation: inappropriate response to anemia in allogeneic transplants. Blood 1991; 77: 868–873.
Beguin Y, Oris R, Fillet G . Dynamics of erythropoietic recovery following bone marrow transplantation: role of marrow proliferative capacity and erythropoietin production in autologous vs allogeneic transplants. Bone Marrow Transplant 1993; 11: 285–292.
Baron F, Sautois B, Baudoux E et al. Optimization of recombinant human erythropoietin therapy after allogeneic hematopoietic stem cell transplantation. Exp Hematol 2002; 30: 546–554.
Baron F, Fillet G, Beguin Y . Erythropoiesis after nonmyeloablative stem-cell transplantation is not impaired by inadequate erythropoietin production as observed after conventional allogeneic transplantation. Transplantation 2002; 74: 1692–1696.
Lemoli RM, Tafuri A, Fortuna A et al. Cycling status of CD34+ cells mobilized into peripheral blood of healthy donors by recombinant human granulocyte colony-stimulating factor. Blood. 1997; 89: 1189–1196.
Groopman JE, Itri LM . Chemotherapy-induced anemia in adults: incidence and treatment. J Natl Cancer Inst 1999; 91: 1616–1634.
Acknowledgements
We thank Dr G Ivanov (University of Cardiff, Cardiff, UK) for critical reading of the manuscript, the nursing staff for providing excellent care for our patients and thank the following physicians at the Institute Paoli-Calmettes for their important study contributions and dedicated patient care: AC Braud, A Charbonnier, JM Schiano de Collela, GL Damaj, JA Gastaut, AM Stoppa and F Viret.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ivanov, V., Faucher, C., Mohty, M. et al. Early administration of recombinant erythropoietin improves hemoglobin recovery after reduced intensity conditioned allogeneic stem cell transplantation. Bone Marrow Transplant 36, 901–906 (2005). https://doi.org/10.1038/sj.bmt.1705152
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1705152
Keywords
This article is cited by
-
Granulocyte and erythropoietic stimulating proteins after high-dose chemotherapy for myeloma
Bone Marrow Transplantation (2007)
-
Transfusions after nonmyeloablative or reduced-intensity conditioning regimens
Leukemia (2006)