Summary:
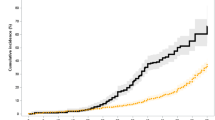
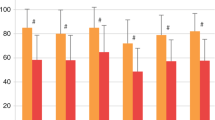
Malignant diseases (MD) occurring after stem cell transplantation (SCT) are of particular concern as increasing number of patients survive and remain free of their original disease. The cumulative incidence at 15 years is 10–12%. The B-cell proliferative disorders (BCLP) are the most common MD in the first year after SCT; the incidence probability is 1% in allogeneic transplants but is much higher (until 14%) after HLA-identical, T-cell-depleted SCT in which Campath 1G or ATG are given. BCLP develop because of reactivation of the EBV and a depressed cellular immunity. Prediction of risk of BCLP can be made by frequent monitoring of EBV load in patients with risk factors. The most effective therapies are the early administration of anti-CD20 monoclonal antibody and adoptive immunotherapy with in vitro generated EBV-specific cytotoxic T cells. Myelodysplasia and acute myeloid leukemia with very poor prognosis have been described in 4–18% of patients with non-Hodgkin lymphoma and Hodgkin disease, 12–24 months after autologous SCT. The risk of development of solid tumors increases over time and the cumulative incidence among children who underwent an SCT at less than 10 years of age is 6–11% at 15 years. There are few studies evaluating quality of life (QOL) in children and adolescents who had received an SCT. The findings of these studies can be summarized as follows: (a) The majority of long survivors enjoy good QOL and return successfully to school or work. (b) A minority (10–15%) complain of physical problems or present moderate cognitive or psychological dysfunctions. (c) The importance of family, other social support and psychological adjustments is generally recognized. More extensive, longitudinal and comparative studies with other alternative therapies are required.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bathia S, Ramsay NKC, Steinbuch M et al. Malignant neoplasms following bone marrow transplantation. Blood 1996; 87: 3633–3639.
Lowsky R, Lipton J, Fyles G et al. Secondary malignancies after bone marrow transplantation in adults. J Clin Oncol 1994; 12: 2187–2192.
Kolb HJ, Socié G, Duell T et al. Malignant neoplasms in long-term survivors of bone marrow transplantation. Ann Intern Med 1999; 131: 738–744.
Bathia S, Bathia R . Secondary malignancies after hematopoietic cell transplantation. In: KG Blume, SJ Forman, FR Appelbaum (eds). Thomas Hematopoietic Cell Transplantation, 3rd edn. Blackwell Publishers: Malden, Mass., 2004, 962–977 (Chapter 70).
Wheeler C, Khurshid A, Ibrahim J et al. Incidence of post-transplantation myelodysplasia/acute leukaemia in non-Hodgkin's lymphoma patients compared with Hodgkin's disease patient undergoing autologous transplantation following cyclophosphamide, carmustine and etoposide. Leuk Lymphoma 2001; 40: 499–509.
Milligan DW, Ruiz de Elvvia MC, Kolb HJ et al. Secondary leukemia and myelodysplasia after autografting for lymphoma: results from the EBMT. Br J Haematol 1999; 106: 1020–1027.
Friedberg JW, Neuberg D, Stone RM et al. Outcome of patients with myelodysplastic syndrome after autologous bone marrow transplantation for non-Hodgkin's lymphoma. J Clin Oncol 1999; 17: 3128–3136.
Curtis RE, travis LB, Rowlings PA et al. Risk of lymphoproliferative disorders after bone marrow transplantation: a multi-institutional study. Blood 1999; 94: 2208–2216.
Socié G, Curtis RE, Deeg J et al. New malignant diseases after allogeneic marrow transplantation for childhood leukemia. J Clin Oncol 2000; 18: 348–357.
Gerritsen EJA, Stam ED, Hermans J et al. Risk factors for developing EBV-related B cell lymphoproliferative disorders after non-HLA-identical BMT in children. Bone Marrow Transplant 1996; 18: 377–382.
van Esser JW, van Der HB, Meijer E et al. Epstein–Barr virus reactivation is a frequent event after allogeneic stem cell transplantation and quantitatively predicts EBV-lymphoppro-liferative disease following T-cell-depleted SCT. Blood 2001; 98: 972–978.
Shapiro RS, Chauvenet A, Mc Guire W et al. Treatment of B-cell lymphoproliferative disorders with interferon alpha and intravenous gammaglobulin. N Eng J Med 1988; 318: 1334–1335.
Dotti E, Rambaldi A, Fiocchi R et al. Anti-CD20 antibody (ritumab) administration in patients with late occurring lymphomas after solid organ. Haematologica 2001; 86: 618–623.
Straathof KC, Savoldo B, Heslop HE, Rooney CM . Immunotherapy for post-transplat lymphoproliferative diseases. Br J Haemat 2002; 118: 728–740.
Faye A, Quartier P, Regerre Y et al. Chimaeric anti-CD20 monoclonal antibody (Rituximab) in post-transplant B-lymphoproliferative disorder following stem cell transplantation in children. Br J Haematol 2001; 115: 112–118.
Rooney LM, Smith CA, Nug CYC et al. Use of gene-modified virus-specific T lymphocytes to control Epstein Barr-virus-related lymphoproliferation. Lancet 1995; 345: 9–13.
Comoli P, Labirio M, Basso S et al. Infusion of autologous Epstein–Barr virus (EBV)-specific cytotoxic T cells for prevention of EBV-related lymphoproliferative disorder in solid organ transplant: recipients with evidence of active virus replication. Blood 2002; 99: 2592–2598.
Curtis SRE, Rowlings PA, Deeg HJ et al. Solid cancers after bone marrow transplantation. N Engl J Med 1997; 336: 897–904.
Deeg H, Socié G . Malignancies after hematopoietic stem cell transplantation: many questions, some answers. Blood 1998; 91: 1833–1844.
Deeg HJ, Spocié G, Henry-Amar M et al. Malignancies after marrow transplantation for aplastic anemia and Fanconi anemia: a joint Seattle and Paris analysis of results in 700 patients. Blood 1996; 87: 386–392.
Favre-Schmuzig G, Hofers S, Passweg S et al. Treatment of solid tumors following allogeneic bone marrow transplantation. Bone Marrow Transplant 2000; 25: 895–898.
Syrjala K . Assessment of quality of life in hematopoietic cell transplantation recipients. In: KG Blume, SJ Forman, FR Appelbaum (eds). Thomas Hematopoietic Cell Transplantation, 3rd edn. Blackwell Publishers: Malden, Mass., 2004, 962–977 (Chapter 70).
Duell S, van Lint MT, Ljungmann P et al. Health and functional status of long-term survivors of bone marrow transplantation. Ann Intern Med 1997; 126: 184–192.
Bush NE, Donaldson GN, Haberman MH et al. Conditional quality of life after hematopoietic stem cell transplantation: a longitudinal follow-up of 415 patients. Biol Blood Marrow Transplant 2000; 6: 576–591.
Kanabar DJ, Attard-Montalto S, Saha V et al. Quality of life in survivors of childhood cancer after megatherapy with autologous bone marrow rescue. Ped Hematol Oncol 1995; 12: 29–36.
Felder-Puig R, Peters C, Matthers-Martin S et al. Physicosocial adjustment of pediatric patients after allogeneic stem cell transplantation. Bone Marrow Transplant 1999; 24: 75–80.
Nespoli L, Verrri AP, Locatelli F et al. The impact of pediatric bone marrow transplantation on quality of life. Qual Life Res 1995; 4: 233–240.
Badell I, Igual L, Gómez P et al. Quality of life in young adults having received a BMT during childhood: a GETMON study. Bone Marrow Transplant 1998; 21 (Suppl. 2): S68–S71.
Sanders JE, Syrkala KL, Hoffmister PH et al. Quality of life of adult survivors of childhood leukemia treated with chemotherapy or bone marrow transplant. Blood 2001; 98: 741a–742a.
Phipps S, Dunavant M, Garvie PA et al. Acute health-related quality of life in children undergoing stem-cell transplants. I-Descriptive outcomes. II Medical and demographic determinants. Bone Marrow Transplant 2002; 29: 425–442.
Barrera M, Boyd-Pringle LA, Sumbler K, Saunders S . Quality of life and behavioural adjustment after pediatric bone marrow transplantation. Bone Marrow Transplant 2000; 26: 427–435.
Kramer JH, Crittenden MR, De Santes H, Cowan MJ . Cognitive and adaptive behavior 1 and 3 years following bone marrow transplantation. Bone Marrow Transplant 1997; 19: 607–613.
Phipps S, Dunavant M, Srivastave DK et al. Cognitive and academic functioning in survivors of pediatric bone marrow transplantation. J Clin Oncol 2000; 18: 1004–1011.
Calaminus G, Weinspach S, Teske C, Göbel U . Quality of life in children and adolescents with cancer. Klin Pädiatr 2000; 212: 211–215.
Ortega JJ, Díaz de Heredia C, Olivé T et al. Allogeneic and autologous stem cell transplantation for AML in first remission: long-term results, late effects and quality of life. Pediatr Blood Cancer 2004; 43: 375 (Abstract).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ortega, J., Olivé, T., de Heredia, C. et al. Secondary malignancies and quality of life after stem cell transplantation. Bone Marrow Transplant 35 (Suppl 1), S83–S87 (2005). https://doi.org/10.1038/sj.bmt.1704854
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704854
Keywords
This article is cited by
-
Occurrence of long-term effects after hematopoietic stem cell transplantation in children affected by acute leukemia receiving either busulfan or total body irradiation: results of an AIEOP (Associazione Italiana Ematologia Oncologia Pediatrica) retrospective study
Bone Marrow Transplantation (2020)
-
Long-term Effects of Myeloablative Allogeneic Hematopoietic Stem Cell Transplantation in Pediatric Patients with Acute Lymphoblastic Leukemia
Current Oncology Reports (2018)
-
Secondary malignancies following high dose therapy and autologous hematopoietic cell transplantation-systematic review and meta-analysis
Bone Marrow Transplantation (2015)
-
Treosulfan-based preparative regimens for allo-HSCT in childhood hematological malignancies: a retrospective study on behalf of the EBMT pediatric diseases working party
Bone Marrow Transplantation (2011)
-
Longitudinal health-related quality of life outcomes and related factors after pediatric SCT
Bone Marrow Transplantation (2009)