Summary:
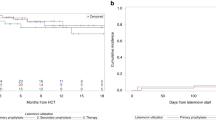
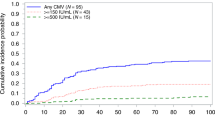
In contrast to allogeneic bone marrow transplantation (allo-BMT), there is a paucity of data on cytomegalovirus (CMV) infection and pre-emptive therapy (PT) strategies following allogeneic peripheral blood stem cell (allo-PBSC) transplantation. We report here on the patterns of CMV infection in a cohort of 225 patients following sibling donor allo-PBSC transplantation. In an attempt to reduce neutropenia, we used intravenous low-dose short-course (LDSC) ganciclovir (GCV) 5 mg/kg once daily for 21 days as preemptive therapy. A total of 165 recipient–donor pairs were CMV seropositive. An initial episode of viremia (detected by shell vial/tube culture) occurred in 75/165 (45%) at a median of day +35 (17–445) post allo-PBSC. In all, 58 patients received PT with LDSC GCV. Among 58, 55 (94%) completed the 21-day course of PT. A second episode of viremia occurred in 19/58 (33%) at day+80 (50–174) and a third episode in 5/58 (9%) at day+134 (103–218). Among patients receiving LDSC GCV, 5/58(9%) developed disease (four pneumonia, one colitis) at day+211 (63–487). No patient on LDSC GCV exhibited a decline in their ANC below 500/μl and none required growth factors. LDSC GCV is extremely well tolerated and cost-effective as PT for CMV viremia following allo-PBSC transplantation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bowden RA, Slichter SJ, Sayers MH et al. Use of leukocyte-depleted platelets and cytomegalovirus-seronegative red blood cells for prevention of primary cytomegalovirus infection after marrow transplant. Blood 1991; 78: 246–250.
Miller WJ, McCullough J, Balfour HH et al. Prevention of cytomegalovirus infection following bone marrow transplantation: a randomized trial of blood product screening. Bone Marrow Transplant 1991; 7: 227–234.
Winston DJ, Ho WG, Bartoni K et al. Ganciclovir prophylaxis of cytomegalovirus infection and disease in allogeneic bone marrow transplant recipients. Results of a placebo- controlled, double-blind trial. Ann Intern Med 1993; 118: 179–184.
Goodrich JM, Bowden RA, Fisher L et al. Ganciclovir prophylaxis to prevent cytomegalovirus disease after allogeneic marrow transplant. Ann Intern Med 1993; 118: 173–178.
Salzberger B, Bowden RA, Hackman RC et al. Neutropenia in allogeneic marrow transplant recipients receiving ganciclovir for prevention of cytomegalovirus disease: risk factors and outcome. Blood 1997; 90: 2502–2508.
Goodrich JM, Mori M, Gleaves CA et al. Early treatment with ganciclovir to prevent cytomegalovirus disease after allogeneic bone marrow transplantation. N Engl J Med 1991; 325: 1601–1607.
Schmidt GM, Horak DA, Niland JC et al. A randomized, controlled trial of prophylactic ganciclovir for cytomegalovirus pulmonary infection in recipients of allogeneic bone marrow transplants; The City of Hope-Stanford-Syntex CMV Study Group. N Engl J Med 1991; 324: 1005–1011.
Singhal S, Mehta J, Powles R . Prevention of cytomegalovirus disease by a short course of preemptive ganciclovir or foscarnet. Blood 1994; 84: 2055.
Peggs KS, Preiser W, Kottaridis PD et al. Extended routine polymerase chain reaction surveillance and pre-emptive antiviral therapy for cytomegalovirus after allogeneic transplantation. Br J Haematol 2000; 111: 782–790.
Reusser P, Einsele H, Lee J et al. Randomized multicenter trial of foscarnet versus ganciclovir for preemptive therapy of cytomegalovirus infection after allogeneic stem cell transplantation. Blood 2002; 99: 1159–1164.
Einsele H, Ehninger G, Hebart H et al. Polymerase chain reaction monitoring reduces the incidence of cytomegalovirus disease and the duration and side effects of antiviral therapy after bone marrow transplantation. Blood 1995; 86: 2815–2820.
Machado CM, Menezes RX, Macedo MC et al. Extended antigenemia surveillance and late cytomegalovirus infection after allogeneic BMT. Bone Marrow Transplant 2001; 28: 1053–1059.
Mori T, Okamoto S, Matsuoka S et al. Risk-adapted pre-emptive therapy for cytomegalovirus disease in patients undergoing allogeneic bone marrow transplantation. Bone Marrow Transplant 2000; 25: 765–769.
Kanda Y, Mineishi S, Saito T et al. Pre-emptive therapy against cytomegalovirus (CMV) disease guided by CMV antigenemia assay after allogeneic hematopoietic stem cell transplantation: a single-center experience in Japan. Bone Marrow Transplant 2001; 27: 437–444.
Nichols WG, Corey L, Gooley T et al. Rising pp65 antigenemia during preemptive anticytomegalovirus therapy after allogeneic hematopoietic stem cell transplantation: risk factors, correlation with DNA load, and outcomes. Blood 2001; 97: 867–874.
Moretti S, Zikos P, VanLint MT et al. Forscarnet vs ganciclovir for cytomegalovirus (CMV) antigenemia after allogeneic hemopoietic stem cell transplantation (HSCT): a randomised study. Bone Marrow Transplant 1998; 22: 175–180.
Boeckh M, Gooley TA, Myerson D et al. Cytomegalovirus pp65 antigenemia-guided early treatment with ganciclovir versus ganciclovir at engraftment after allogeneic marrow transplantation: a randomized double-blind study. Blood 1996; 88: 4063–4071.
Sakuma H, Hosoya M, Kanno H et al. Risk of cytomegalovirus infection after peripheral blood stem cell transplantation. Bone Marrow Transplant 1997; 19: 49–53.
Trenschel R, Ross S, Husing J et al. Reduced risk of persisting cytomegalovirus pp65 antigenemia and cytomegalovirus interstitial pneumonia following allogeneic PBSCT. Bone Marrow Transplant 2000; 25: 665–672.
Manteiga R, Martino R, Sureda A et al. Cytomegalovirus pp65 antigenemia-guided pre-emptive treatment with ganciclovir after allogeneic stem transplantation: a single-center experience. Bone Marrow Transplant 1998; 22: 899–904.
Link H, Arseniev L, Ljungman P et al. Cytomegalovirus infections after allogeneic peripheral blood progenitor cell transplantation (PBSCT) – a survey of the infectious disease working party of EBMT. Blood 1997; 90(Suppl. 1): 544a (Abstr. 2424).
Gleaves CA, Smith TF, Shuster EA et al. Rapid detection of cytomegalovirus in MRC-5 cells inoculated with urine specimens by using low-speed centrifugation and monoclonal antibody to an early antigen. J Clin Microbiol 1984; 19: 917–919.
Swenson PD, Kaplan MH . Rapid detection of cytomegalovirus in cell culture by indirect immunoperoxidase staining with monoclonal antibody to an early nuclear antigen. J Clin Microbiol 1985; 21: 669–673.
Przepiorka D, Weisdorf D, Martin P et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant 1995; 15: 825–828.
Vij R, DiPersio J, Venkatraman P et al. Donor CMV serostatus has no impact on CMV viremia or disease when prophylactic granulocyte transfusions are given following allogeneic peripheral blood stem cell transplantation. Blood 2003; 101: 2067–2069.
Meyers JD, Flournoy N, Thomas ED et al. Risk factors for cytomegalovirus infection after human marrow transplantation. J Infect Dis 1986; 153: 478–488.
Storch GA, Buller RS, Bailey TC et al. Comparison of PCR and pp65 antigenemia assay with quantitative shell vial culture for detection of cytomegalovirus in blood leukocytes from solid-organ transplant recipients. J Clin Microbiol 1994; 32: 997–1003.
Brown RA, Adkins D, Khoury H et al. Long-term follow-up of high-risk allogeneic peripheral-blood stem-cell transplant recipients: graft-versus-host disease and transplant-related mortality. J Clin Oncol 1999; 17: 806–812.
Korbling M, Przepiorka D, Huh YO et al. Allogeneic blood stem cell transplantation for refractory leukemia and lymphoma: potential advantage of blood over marrow allografts. Blood 1995; 85: 1659–1665.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vij, R., Khoury, H., Brown, R. et al. Low-dose short-course intravenous ganciclovir as pre-emptive therapy for CMV viremia post allo-PBSC transplantation. Bone Marrow Transplant 32, 703–707 (2003). https://doi.org/10.1038/sj.bmt.1704216
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704216
Keywords
This article is cited by
-
A Low Incidence of Cytomegalo Virus Infection Following Allogeneic Hematopoietic Stem Cell Transplantation Despite a High Seroprevalence
Indian Journal of Hematology and Blood Transfusion (2018)
-
A randomized trial of preemptive therapy for prevention of cytomegalovirus disease after allogeneic hematopoietic stem cell transplantation
International Journal of Hematology (2010)
-
Preemptive therapy with ganciclovir 5 mg/kg once daily for cytomegalovirus infection after unrelated cord blood transplantation
Bone Marrow Transplantation (2008)
-
Once daily ganciclovir as initial pre-emptive therapy delayed until threshold CMV load ⩾10000 copies/ml: a safe and effective strategy for allogeneic stem cell transplant patients
Bone Marrow Transplantation (2006)
-
Anti-CD25 monoclonal antibody (basiliximab) for prevention of graft-versus-host disease after haploidentical bone marrow transplantation for hematological malignancies
Bone Marrow Transplantation (2005)