Summary:
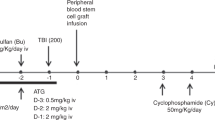
The primary object of the conditioning regimen for allogeneic reduced-intensity stem cell transplantation (RIST) is immunosuppression to achieve stable engraftment of donor cells, rather than bone marrow ablation. Therefore, immune reconstitution after RIST might be different from that after conventional stem cell transplantation (CST). In this study, 22 patients underwent RIST and 28 underwent CST. The RIST regimen consisted of cladribine (2-CdA; 0.11 mg/kg/day for 6 days), BU (4 mg/kg/day for 2 days), and rabbit anti-thymocyte globulin (ATG; 2.5 mg/kg/day for 2–4 days). The CST group received either the BU (4 mg/kg/day × 4 days)/CY (60 mg/kg/day × 2 days) (n=13) or CY (60 mg/kg/day × 2 days)/TBI (4 Gy/day × 3 days) regimen (n=15). All patients underwent transplantation with G-CSF-mobilized blood stem cells. Engraftment speed after RIST was fast and seven of 22 patients did not require platelet transfusion. We noted that the numbers of CD4+, CD4+CD45RA+, and CD4+CD45RO+ T cells after transplant in the RIST group were significantly lower than those in the CST group (P=0.0001 for both the comparisons). However, the reconstitution of CD20+ B cells was faster in the RIST group (P=0.0001). The response of T cells to PHA stimulation was lower in the RIST group (P=0.0001 on day 30 and P=0.02 on day 90). Nevertheless, there were no significant differences in the incidence of bacterial, fungal, or viral infections between the two groups. We concluded that our RIST regimen might delay laboratory-evaluated T-cell immune reconstitution compared to CST; however, the observed setbacks did not directly translate into clinically significant increases in infectious episodes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ottinger HD, Beelen DW, Scheulen B et al. Improved immune reconstitution after allotransplantation of peripheral blood stem cells instead of bone marrow. Blood 1996; 88: 2775–2779.
Champlin RE, Schmitz N, Horowitz MM et al. Blood stem cells compared with bone marrow as a source of hematopoietic cells for allogeneic transplantation. Blood 2000; 95: 3702–3709.
Stork J, Dawson MA, Storer B et al. Immune reconstitution after allogeneic marrow transplantation compared with blood stem cell transplantation. Blood 2001; 97: 3380–3389.
Wagner JE, Broxmeyer HE, Byrd RL et al. Transplantation of umbilical cord blood after myeloablative therapy: analysis of engraftment. Blood 1992; 79: 1874–1881.
Small TN, Papadopoulos EB, Boulad F et al. Comparison of immune reconstitution after unrelated and related T-cell-depleted bone marrow transplantation: effect of patient's age and donor leukocyte infusions. Blood 1999; 93: 467–480.
Behringer D, Bertz H, Schmoor C et al. Quantitative lymphocyte subset reconstitution after allogeneic hematopoietic transplantation from matched related donors with CD34+ selected PBPC grafts, unselected PBPC grafts or BM grafts. Bone Marrow Transplant 1999; 24: 295–302.
Champlin R, Khouri I, Shimoni A et al. Harnessing graft-versus-malignancy: non myeloablative preparative regimens for allogeneic haematopoietic transplantation, and an evolving strategy for adoptive immuno therapy. Br J Haematol 2000; 111: 18–29.
Childs R, Chernoff A, Contentin N et al. Regression of metastatic renal-cell carcinoma after nonmyeloablative allogeneic peripheral-blood stem-cell transplantation. N Engl J Med 2000; 343: 750–758.
Saito T, Kanda Y, Kami M et al. Therapeutic potential of a reduced-intensity preparative regimen for allogeneic transplantation with cladribine, busulfan and anti-thymocyte globulin against advanced/refractory acute leukemia/lymphoma. Clin Cancer Res 2002; 8: 1014–1020.
Ziegler SF, Ramsdell F, Alderson MR et al. The activation antigen CD69. Stem Cells 1994; 12: 456–465.
Kook H, Goldman F, Padley D et al. Reconstitution of immune system after unrelated or partially matched T-cell-depleted bone marrow transplantation in children: immunophenotypic analysis and factors affecting the speed. Blood 1996; 88: 1087–1097.
Amrolia P, Gaspar HB, Hassan A et al. Nonmyeloablative stem cell transplantation for congenital immunodeficiencies. Blood 2000; 96: 1239–1246.
Morecki S, Gelfand A, Nagler A et al. Immune reconstitution following stem cell transplantation in recipients conditioned by low-intensity vs myeloablative regimen. Bone Marrow Transplant 2001; 28: 243–249.
Finke J, Bertz H, Schmoor C et al. Allogeneic bone marrow transplantation from unrelated donors using in vivo anti-T-cell globulin. Br J Haematol 2000; 111: 303–313.
Naparstek E, Delukina M, Or R et al. Engraftment of marrow allografts treated with Campath-1 monoclonal antibodies. Exp Hematol 1999; 27: 1210–1218.
Slavin S, Nagler A, Naparstek E et al. Nonmyeloablative stem cell transplantation and cell therapy as an alternative to conventional bone marrow transplantation with lethal cytoreduction for the treatment of malignant and nonmalignant hematologic disease. Blood 1998; 91: 756–763.
Kottaridis PD, Milligan DW, Chopra R et al. In vivo CAMPATH-1H prevents graft-versus-host disease following nonmyeloablative stem cell transplantation. Blood 2000; 96: 2419–2425.
Mohty M, Faucher C, Vey N et al. High rate of secondary viral and bacterial infections in patients undergoing allogeneic bone marrow mini-transplantation. Bone Marrow Transplant 2000; 26: 251–255.
Michael J, Robertson J, Ritz J . Biology and clinical relevance of human natural killer cells. Blood 1990; 76: 2421–2438.
Soiffer RJ, Murray C, Cochran K et al. Clinical and immunologic effects of prolonged infusion of low-dose recombinant interleukin-2 after autologous and T-cell-depleted allogeneic bone marrow transplantation. Blood 1992; 79: 517–526.
Nieda M, Nicol A, Koezuka Y et al. TRAIL expression by activated human CD4+Vα24 NKT cells induces in vitro and vivo apoptosis of human acute myeloid leukemia cells. Blood 2001; 97: 2067–2074.
Moingeon P, Chang HC, Sayre PH et al. The structural biology of CD2. Immunol Rev 1989; 111: 111–144.
Berney SM, Schaan T, Wolf RE et al. CD2 (OKT11) augments CD3-mediated intracellular signaling events in human T lymphocytes. J Invest Med 2000; 48: 102–109.
Acknowledgements
This research was supported by a Grant-in-Aid for Scientific Research from the Japanese Ministry of Health, Labor and Welfare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saito, T., Kanda, Y., Nakai, K. et al. Immune reconstitution following reduced-intensity transplantation with cladribine, busulfan, and antithymocyte globulin: serial comparison with conventional myeloablative transplantation. Bone Marrow Transplant 32, 601–608 (2003). https://doi.org/10.1038/sj.bmt.1704205
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704205
Keywords
This article is cited by
-
Post-transplantation lymphoproliferative disorder after haematopoietic stem cell transplantation
Annals of Hematology (2021)
-
Epstein-Barr virus-related post-transplant lymphoproliferative disease (EBV-PTLD) in the setting of allogeneic stem cell transplantation: a comprehensive review from pathogenesis to forthcoming treatment modalities
Bone Marrow Transplantation (2020)
-
Late-onset hemorrhagic cystitis after haploidentical hematopoietic stem cell transplantation in patients with advanced leukemia: differences in ATG dosage are key
International Journal of Hematology (2013)
-
Immune reconstitution after allogeneic stem cell transplantation with reduced-intensity conditioning regimens
Leukemia (2007)
-
Lymphocyte reconstitution following allogeneic hematopoietic stem cell transplantation: a retrospective study including 148 patients
Bone Marrow Transplantation (2007)