Summary:
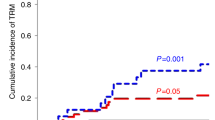
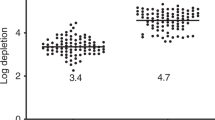
Transient T cell immunodeficiency is a common complication following hematopoietic stem cell transplantation. In breast cancer patients transplanted with autologous peripheral blood progenitor cells (PBPC) harvested after cytotoxic treatment with either cyclophosphamide or epirubicin plus paclitaxel, we evaluated T cells infused in grafts and in peripheral blood during the early reconstitution phase. We found that PBPC grafts harvested after treatment with epirubicin plus paclitaxel contained substantially larger numbers of T cells with less altered composition than after cyclophosphamide. Three months after high-dose cytotoxic chemotherapy, the numbers and the kinetics of circulating naive T cells, but not of memory and CD28− T cells, correlated positively with the number of naive T cells infused PBPC grafts. Finally, retrospective analysis of two cohorts of patients transplanted in different clinical settings with PBPC grafts harvested following cyclophosphamide or epirubicin plus paclitaxel showed apparently different susceptibilities to develop endogenous varicella zoster virus reactivation in the first year after high-dose cytotoxic chemotherapy. On the whole, these data indicate that number and composition of T cells in PBPC grafts vary according to the former cytotoxic therapy, and suggest that autologous transfer of T cells may accelerate the early T cell reconstitution phase and possibly ameliorate immune competence in patients rendered lymphopenic by high-dose chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Guillaume T, Rubinstein DB, Symann M . Immune reconstitution and immunotherapy after autologous hematopoietic stem cell transplantation. Blood 1998; 92: 1471–1490.
Sugita K, Soiffer RJ, Murray C et al. The phenotype and reconstitution of immunoregulatory T cell subsets after T cell-depleted allogeneic and autologous bone marrow transplantation. Transplantation 1994; 57: 1465–1473.
Mackall CL, Fleisher TA, Brown MR et al. Distinctions between CD8+ and CD4+ T-cell regenerative pathways result in prolonged T-cell subset imbalance after intensive chemotherapy. Blood 1997; 89: 3700–3707.
Fagnoni FF, Lozza L, Zibera C et al. T-cell dynamics after high-dose chemotherapy in adults: elucidation of the elusive CD8+ subset reveals multiple homeostatic T-cell compartments with distinct implications for immune competence. Immunology 2002; 106: 26–37.
Pennesi G, Liu Z, Ciubotariu R et al. TCR repertoire of suppressor CD8+ CD28− T cell populations. Hum Immunol 1999; 60: 291–304.
Batliwalla F, Monteiro J, Serrano D, Gregersen PK . Oligoclonality of CD8+ T cells in health and disease: aging, infection, or immune regulation? Hum Immunol 1996; 48: 68–76.
Avigan D, Pirofski LA, Lazarus HM . Vaccination against infectious disease following hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2001; 7: 171–183.
Freitas AA, Rocha B . Population biology of lymphocytes: the flight for survival. Annu Rev Immunol 2000; 18: 83–111.
Mackall CL, Fleisher TA, Brown MR et al. Age, thymopoiesis, and CD4+ T-lymphocyte regeneration after intensive chemotherapy. New Engl J Med 1995; 332: 143–149.
Vavassori M, Maccario R, Moretta A et al. Restricted ~TCR repertoire and long-term persistence of donor-derived antigen-experienced CD4+ T cells in allogeneic bone ~marrow transplantation recipients. J Immunol 1996; 157: 5739–5747.
Bomberger C, Singh-Jairam M, Rodey G et al. Lymphoid reconstitution after autologous PBSC transplantation with FACS-sorted CD34+ hematopoietic progenitors. Blood 1998; 91: 2588–2600.
Mackall CL, Stein D, Fleisher TA et al. Prolonged CD4 depletion after sequential autologous peripheral blood progenitor cell infusions in children and young adults. Blood 2000; 96: 754–762.
Fagnoni FF, Vescovini R, Passeri G et al. Shortage of circulating naive CD8(+ ) T cells provides new insights on immunodeficiency in aging. Blood 2000; 95: 2860–2868.
Fagnoni FF, Vescovini R, Mazzola M et al. Expansion of cytotoxic CD8+ CD28− T cells in healthy ageing people, including centenarians. Immunology 1996; 88: ~501–507.
Hamann D, Baars PA, Rep MH et al. Phenotypic and functional separation of memory and effector human CD8+ T cells. J Exp Med 1997; 186: 1407–1418.
Hamann D, Roos MT, van Lier RA . Faces and phases of human CD8 T-cell development. Immunol Today 1999; 20: 177–180.
Zibera C, Pedrazzoli P, Ponchio L et al. Efficacy of epirubicin/paclitaxel combination in mobilizing large amounts of hematopoietic progenitor cells in patients with metastatic breast cancer showing optimal response to the same chemotherapy regimen. Haematologica 1999; 84: 924–929.
Pedrazzoli P, Perotti C, Da Prada GA et al. Collection of circulating progenitor cells after epirubicin, paclitaxel and filgrastim in patients with metastatic breast cancer. Br J Cancer 1997; 75: 1368–1372.
Brando B, Barnett D, Janossy G et al. Cytofluorometric methods for assessing absolute numbers of cell subsets in blood. European Working Group on Clinical Cell Analysis. Cytometry 2000; 42: 327–346.
De Rosa SC, Herzenberg LA, Herzenberg LA, Roederer M . 11-color, 13-parameter flow cytometry: identification of human naive T cells by phenotype, function, and T-cell receptor diversity. Nat Med 2001; 7: 245–248.
von Andrian UH, Mackay CR . T-cell function and migration. Two sides of the same coin. New Engl J Med 2000; 343: 1020–1034.
Mackall CL . T-cell immunodeficiency following cytotoxic antineoplastic therapy: a review. Stem Cells 2000; 18: ~10–18.
Rutella S, Pierelli L, Sica S et al. Transplantation of autologous peripheral blood progenitor cells: impact of CD34-cell selection on immunological reconstitution. Leuk Lymphoma 2001; 42: 1207–1220.
Douek DC, Vescio RA, Betts MR et al. Assessment of thymic output in adults after haematopoietic stem-cell transplantation and prediction of T-cell reconstitution. Lancet 2000; 355: 1875–1881.
Singh RK, Ino K, Varney ML et al. Immunoregulatory cytokines in bone marrow and peripheral blood stem cell products. Bone Marrow Transplant 1999; 23: 53–62.
Dummer W, Ernst B, LeRoy E et al. Autologous regulation of naive T cell homeostasis within the T cell compartment. J Immunol 2001; 166: 2460–2468.
Almeida AR, Borghans JA, Freitas AA . T cell homeostasis: thymus regeneration and peripheral T cell restoration in mice with a reduced fraction of competent precursors. J Exp Med 2001; 194: 591–599.
Arstila TP, Casrouge A, Baron V et al. A direct estimate of the human alphabeta T cell receptor diversity. Science 1999; 286: 958–961.
Mariani S, Coscia M, Even J et al. Severe and long-lasting disruption of T-cell receptor diversity in human myeloma after high-dose chemotherapy and autologous peripheral blood progenitor cell infusion. Br J Haematol 2001; 113: 1051–1059.
Porrata LF, Ingle JN, Litzow MR et al. Prolonged survival associated with early lymphocyte recovery after autologous hematopoietic stem cell transplantation for patients with metastatic breast cancer. Bone Marrow Transplant 2001; 28: 865–871.
Hsu FJ, Benike C, Fagnoni F et al. Vaccination of patients with B-cell lymphoma using autologous antigen-pulsed dendritic cells. Nat Med 1996; 2: 52–58.
Fagnoni FF, Robustelli della Cuna G . Immunotherapy: on the edge between experimental and clinical oncology. J Chemother 2001; 13: 15–23.
Acknowledgements
The authors wish to thank Orietta Moscato and all nurses and personnel of the Medical Oncology Division for their assistance to patients and helping in blood samples collection.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fagnoni, F., Lozza, L., Zibera, C. et al. Cytotoxic chemotherapy preceding apheresis of peripheral blood progenitor cells can affect the early reconstitution phase of naive T cells after autologous transplantation. Bone Marrow Transplant 31, 31–38 (2003). https://doi.org/10.1038/sj.bmt.1703782
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703782
Keywords
This article is cited by
-
T-lymphocyte reconstitution following rigorously T-cell-depleted versus unmodified autologous stem cell transplants
Bone Marrow Transplantation (2006)