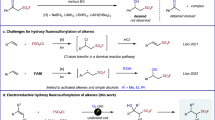
Abstract
A SHORTT time ago we reported1 that hypobromous acid became a powerful brominating agent in the presence of mineral acids owing to the production of bromine cations: 
Similar content being viewed by others
Article PDF
References
Derbyshire and Waters, Nature, 164, 446 (1949).
Evans, M. G., and Uri, N., Trans. Farad. Soc., 45, 224 (1949).
Swern, D., Chemical Reviews, 45, 1, (1949).
Swern, D., J. Amer. Chem. Soc., 69, 1692 (1947).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DERBYSHIRE, D., WATERS, W. An Oxidation Involving the Hydroxyl Cation, (OH)+. Nature 165, 401 (1950). https://doi.org/10.1038/165401a0
Issue Date:
DOI: https://doi.org/10.1038/165401a0
This article is cited by
-
Preparation of phenols by phthaloyl peroxide–mediated oxidation of arenes
Nature Protocols (2014)
-
Oxidation of mesitylene by hydrogen peroxide in AcOH-Ac2O-H2SO4
Bulletin of the Academy of Sciences of the USSR Division of Chemical Science (1990)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.