Abstract
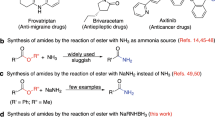
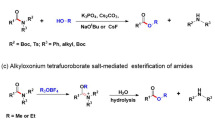
FOR the synthesis of esters from carboxylic acids and alcohols or phenols, it is customary to heat the reactants for several hours in the presence of a strong mineral acid catalyst, or else to proceed via the acid chloride or the acid anhydride. We now report a method of esterifleation which obviates the necessity of a two-stage process and, at the same time, enables a rapid direct reaction to occur between the acid and the hydroxy-compound under mild conditions. The new method, which entails the use of the anhydride of trifluoracetic acid, gives the purified esters in yields of 60–90 per cent.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
STACEY, M., BOURNE, E., TATLOW, J. et al. A General Method of Esterification using Trifluoracetic Anhydride. Nature 164, 705 (1949). https://doi.org/10.1038/164705a0
Issue Date:
DOI: https://doi.org/10.1038/164705a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.