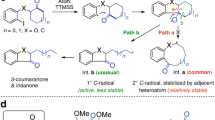
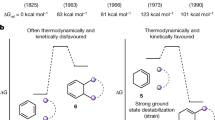
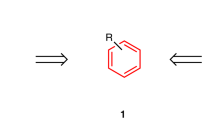
Abstract
DR. Walsh has defended his suggested structure1 for the compounds ethylene oxide and cyclopropane in terms of a molecular orbitals picture based on the assumption that the carbon atoms are in the trigonal, or near-trigonal, state. The calculations by Linnett2 of the CH-bond force-constants are consistent with trigonal bonding.
Similar content being viewed by others
Article PDF
References
Walsh, A. D., Nature, 159, 165 and 712 (1947).
Linnett, J. W., Nature, 160, 162 (1947).
Bastiansen and Hassel, Tids. Kjemi Bergvesen Met., 6, 71 (1946).
Pauling and Brockway, J. Amer. Chem. Soc., 59, 1223 (1937).
Smith, L. G., Phys. Rev., 59, 924 (1941).
O'Gorman and Schomaker, J. Amer. Chem. Soc., 68, 1138 (1946).
See, for example, Skinner, H. A., Trans. Farad. Soc., 41, 645 (1945).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
SKINNER, H. Structure of Cyclopropane. Nature 160, 902 (1947). https://doi.org/10.1038/160902a0
Issue Date:
DOI: https://doi.org/10.1038/160902a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.