Abstract
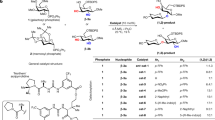
ANHYDRO-SUGARS have already been the subject of diligent study by several groups of workers, and the chief methods of obtaining these compounds have been through the hydrolysis of sulphonic or halogen acid esters of sugar derivatives. For example, 2: 3-anhydro?-methyltaloside was obtained by alkaline hydrolysis of 2-p-toluene sulphonyl?-methylgalactoside1; 1: 6-2: 3-dianhydro?-talose was isolated after the alkaline hydrolysis of 2-mesyl 1: 6-anhydro?galactose2, and 3: 6-anhydro?-methylglucoside has been obtained by the hydrolysis of 6-bromo?-methylglucoside3. Sulphuric acid esters of sugars have also been made to yield anhydro-derivatives. For example, Duff and Percival4 obtained 3:6-anhydro?-methyl glucopyranoside from barium -methyl glucopyranoside sulphate. Finally, the same type of reaction has been applied to the nitric acid esters of sugars. Gladding and Purves5 obtained a product which probably contained 2: 3-anhydro?-methyl-mannoside from 3:4: 6-triacetyl?-methylglucoaide 2-nitrate and 3: 6-anhydro?-methyl-glucoside from 2: 3: 4-triacetyl?-methylglucoside 6-nitrate.
Similar content being viewed by others
Article PDF
References
Wiggins, J. Chem. Soc., 522 (1944).
James, Smith, Stacey and Wiggins, Nature, 156, 308 (1945).
Fischer and Zach, Ber., 45, 456 (1912).
Duff and Percival, J. Chem. Soc., 830 (1941).
Gladding and Purves, J. Amer. Chem. Soc., 66, 76 (1944).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
WIGGINS, L. Reversible Conversion of Amino- into Anhydro-sugars. Nature 157, 300 (1946). https://doi.org/10.1038/157300a0
Issue Date:
DOI: https://doi.org/10.1038/157300a0
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.