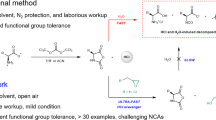
Abstract
FOR a long time it has been known that heating under various degrees of the ester of glycine1, glycyl-glycine2 and diglycyl-glycine3 results in the formation of certain condensation products with liberation of alcohol. If the hexapeptide ester of glycine would undergo the type of condensation exhibited by the dipeptide ester, then the simplest model of a ‘cyclol 6’ postulated by the Wrinch theory4 could be prepared. The formation of such ‘cyclo-hoxapeptide’ of glycine could then be considered as evidence for the hexagonal folding by hydrogen bonds of the polypeptide chains.
Similar content being viewed by others
Article PDF
References
Curtius, Th., Ber., 37, 1289 (1904).
Fischer, E., and Fourneau, E., Ber., 34, 2868 (1901).
Fischer, E., Ber., 39, 453 (1906).
Wrinch, D. M., NATURE, 137, 411 (1936), et seq.; Proc. Roy. Soc., A, 160, 59 (1937).
Fischer, E., Ber., 39, 2893 (1906).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
PACSU, E. Condensation of the Hexapeptide Ester of Glycine into the 96- and Higher (3 × 2n) Peptide Esters. Nature 144, 551 (1939). https://doi.org/10.1038/144551a0
Issue Date:
DOI: https://doi.org/10.1038/144551a0
This article is cited by
-
High molecular compounds
Bulletin of the Academy of Sciences of the USSR Division of Chemical Science (1954)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.