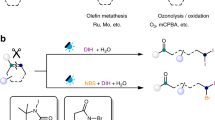
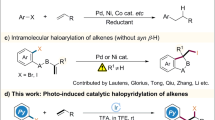
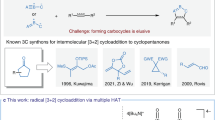
Abstract
ACCORDING to the theory of the mechanism of the catalysed bromination of ketones advanced by me1, presented in outline by Nathan Watson and Laurie2, and more recently extended by Watson Evans and Morgan3, the function of the catalytic molecule is the introduction of the translational energy of collision specifically into the carbonyl bond through the formation of transient addition complexes, which leave the bond in an electronically activated condition: ” the rupture of one link of the double bond, in chemical changes such as the formation of addition compounds, is an asymmetrical process, which can be formulated as depending on the transient formation of a semi-polar double bond in an activated phase of the molecule”4.
Similar content being viewed by others
Article PDF
References
Thesis, University of London (1933).
J. Chem. Phys., 3, 170 (1935).
J. Chem. Soc., 1167, 1173 (1935).
Cf. Lowry, Chem. and Ind., 456 (1925).
Cf. Lowry, J. Chem. Soc., 620 (1926).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
NATHAN, W. Photo-Activation of the Carbpnyl Group in Prototropic Reactions. Nature 137, 460 (1936). https://doi.org/10.1038/137460b0
Issue Date:
DOI: https://doi.org/10.1038/137460b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.