Abstract
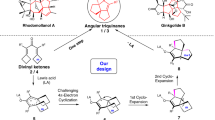
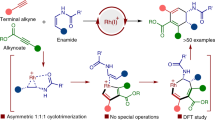
THE claim of Dr. Qudrat-i-Khuda1 to the isolation of the four stereoisomeric forms of l-carboxy-4-methyleycloZohexane-1-acetic acid required by the Sasche-Mohr hypothesis which it has not been possible to substantiate in this laboratory2 led us to search for further possible examples of this phenomenon in reactions involving the methylcyclo-hexanones. Attention was directed, therefore, to the condensation of the cyanhydrins of methylcyclo-hexanones with arylamines, by an extension of the method used by Walther and Hubner in the case of the simple cyclohexanone3
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
NATURE, 136, 301; August 24, 1935.
NATURE, 136, 608; October 12, 1935.
J. prakt. Chem., 93, 124; 1916.
J. Chem. Soc., 2073; 1928.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DESAI, R., HUNTER, R. Multiplanar Cyclohexane Rings. Nature 136, 953–954 (1935). https://doi.org/10.1038/136953b0
Issue Date:
DOI: https://doi.org/10.1038/136953b0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.