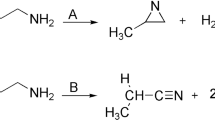
Abstract
IN a letter in NATURE of March 30, I asked for suspension of judgment regarding a difference of opinion between Mr. Hinshelwood and his colleagues on one hand, and myself and my co-workers on the other, with regard to the possibility of measuring the rate of chemical reaction in the thermal decomposition of gaseous acetaldehyde by observing the rate of increase of pressure at constant volume, and also as to the mechanism of the chemical process involved. Mr. Hinshelwood has published an account of his latest work on this subject, and Mr. R. V. Seddon and I have carried on our own work to a point at which publication is possible. We believe that our results support our views on the case.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
TRAVERS, M. Thermal Decomposition of Certain Gaseous Organic Compounds. Nature 136, 909–910 (1935). https://doi.org/10.1038/136909b0
Issue Date:
DOI: https://doi.org/10.1038/136909b0
This article is cited by
-
Thermal Decomposition of Ethylene Oxide
Nature (1936)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.