Abstract
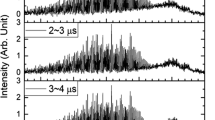
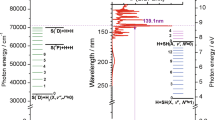
IN continuation of earlier work1 on SO2, the emission bands of SeO and the absorption bands of SeO2 have been analysed. The former consist of a long v = 0 progression and a short v = 1 progression. The origin of the system cannot be definitely located but the v = 0 progression can be expressed by the following formula v = 30433(882 v6 v2). Considerations of intensity distribution lead us to 33167 cm.1 as v0 and = 910 cm.–1, and the energy of dissociation of unexcited SeO will therefore be 4·17 volts. The vibrational frequency of the excited state co is 513 cm.–1
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
R. K. Asundi and R. Samuel, Proc. Ind. Ac. Soc. (Bangalore), 2, 30 ; 1935.
G. Herzberg and E. Teller, Z. Phys. Chem., B, 21, 410 ; 1933.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
ASUNDI, R., JAN-KHAN, M. & SAMUEL, R. Spectra of SeO and SeO2. Nature 136, 642–643 (1935). https://doi.org/10.1038/136642c0
Issue Date:
DOI: https://doi.org/10.1038/136642c0
This article is cited by
-
Ground State Vibrational Frequencies
Nature (1936)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.