Abstract
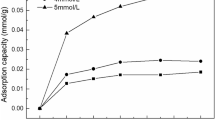
ALUMINIUM hydroxide is readily adsorbed by kieselguhr to form an adsorption complex which is remarkably stable. The hydroxide is deposited by the slow addition of a small excess of ammonia to a de-aerated suspension of 10 gm. of specially purified Superfloss kieselguhr in 100 c.c. of 2 per cent ammonium nitrate solution containing a known amount of aluminium nitrate, the suspension being vigorously stirred. The curve (Fig. 1) shows the relation between the value of the preparations in N/10 acetic acid and the amount of aluminium hydroxide adsorbed by 100 gm. of kieselguhr.
Similar content being viewed by others
Article PDF
References
Physica, 1, 753, 935, 953, 960; 1934.
B. A. Report, 1928, p. 35.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BALY, E., PEPPER, W. Adsorption of Aluminium Hydroxide by Kieselguhr. Nature 136, 28 (1935). https://doi.org/10.1038/136028a0
Issue Date:
DOI: https://doi.org/10.1038/136028a0
This article is cited by
-
Fixation of sulphur in the muds of Lake Victoria
Hydrobiologia (1958)
-
Fibre-Forms in Animal Hairs
Nature (1935)
-
Kinetics of Heterogeneous Catalysis and of Enzyme Action
Nature (1935)
-
Fibre Forms in Animal Hairs
Nature (1935)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.