Abstract
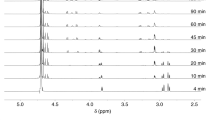
IN dealing with my 1 comments upon their communication 2 under this title, Speakman and Hirst 3 describe as erroneous my statement that “the guanidine group of arginine does not … yield nitrogen with nitrous acid under the usual conditions”. By “usual conditions” I naturally imply those generally adopted in determinations of amino-nitrogen in the course of protein analysis as described by van Slyke,4 5 that is to say, contact between acid nitrate solution and amino-body at room temperature, for 3–5 minutes in the case of mono-amino acids and for ½–1 hour in the case of the hexone bases or proteins. Under these conditions the guanidine group of arginine is practically unaffected. The work of Plimmer 6 to which Speakman and Hirst refer is, of course, well known to me, and in that paper it is stated that any error introduced by reaction of the guanidine group of arginine in a 1-hour determination is inappreciable. With longer reaction times, correspondingly greater decomposition does occur.
Similar content being viewed by others
Article PDF
References
Rimington, NATURE, 129, 580, April 16, 1932.
Speakman and Hirst, NATURE, 128, 1073, Dec. 26, 1931.
Speakman and Hirst, NATURE, 129, 938, June 25, 1932.
Van Slyke, J. Biol. Chem., 9, 185; 1911.
Van Slyke, J. Biol. Chem., 10, 15; 1911–12.
Plimmer, Biochem. J., 18, 105; 1924.
Marston, Council of Sci. and Ind. Res., Commonwealth of Australia, Bulletin 38; 1928.
Abderhalden and Voitinovici, Z. Physiol. Chem., 52, 368; 1907.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
REMINGTON, C. Constitution of the Keratin Molecule. Nature 130, 401 (1932). https://doi.org/10.1038/130401a0
Issue Date:
DOI: https://doi.org/10.1038/130401a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.