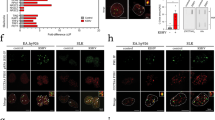
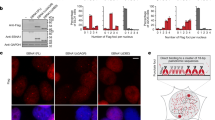
Abstract
Epstein–Barr virus (EBV) has been implicated in the pathogenesis of human malignancies but the mechanisms of oncogenesis remain largely unknown. Genomic instability and chromosomal aberrations are hallmarks of malignant transformation. We report that EBV carriage promotes genomic instability in Burkitt's lymphoma (BL). Cytogenetic analysis of EBV− and EBV+ BL lines and their sublines derived by EBV conversion or spontaneous loss of the viral genome revealed a significant increase in dicentric chromosomes, chromosome fragments and chromatid gaps in EBV-carrying cells. Expression of EBV latency I was sufficient for this effect, whereas a stronger effect was observed in cells expressing latency III. Telomere analysis by fluorescent in situ hybridization revealed an overall increase of telomere size and prevalence of telomere fusion and double strand-break fusion in dicentric chromosomes from EBV+ cells. Phosphorylated H2AX, a reporter of DNA damage and ongoing repair, was increased in virus-carrying cells in the absence of exogenous stimuli, whereas efficient activation of DNA repair was observed in both EBV+ and EBV− cells following treatment with etoposide. These findings point to induction of telomere dysfunction and DNA damage as important mechanisms for EBV oncogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- BL:
-
Burkitt's lymphoma
- EBV:
-
Epstein–Barr virus
- LCL EBV:
-
immortalized lymphoblastoid cell line
- CIN:
-
chromosomal instability
- DSB:
-
double-strand DNA breaks
References
Bernheim A, Berger R, Lenoir G . (1981). Cytogenetic studies on African Burkitt's lymphoma cell lines: t(8;14), t(2;8) and t(8;22) translocations. Cancer Genet Cytogenet 3: 307–315.
Cao J, Liu Y, Sun H, Cheng G, Pang X, Zhou Z . (2002). Chromosomal aberrations, DNA strand breaks and gene mutations in nasopharyngeal cancer patients undergoing radiation therapy. Mutat Res 504: 85–90.
Chan SW, Blackburn EH . (2003). Telomerase and ATM/Tel1p protect telomeres from nonhomologous end joining. Mol Cell 11: 1379–1387.
Chan WY, Chan AB, Liu AY, Chow JH, Ng EK, Chung SS . (2001). Chromosome 11 copy number gains and Epstein–Barr virus-associated malignancies. Diagn Mol Pathol 10: 223–227.
Chan WY, Liu Y, Li CY, Ng EK, Chow JH, Li KK et al. (2002). Recurrent genomic aberrations in gastric carcinomas associated with H. pylori and Epstein–Barr virus. Diagn Mol Pathol 11: 127–134.
Chen F, Liu C, Lindvall C, Xu D, Ernberg I . (2005). Epstein–Barr virus latent membrane 2A (LMP2A) down-regulates telomerase reverse transcriptase (hTERT) in epithelial cell lines. Int J Cancer 113: 284–289.
Dolcetti R, Masucci MG . (2003). Epstein–Barr virus: induction and control of cell transformation. J Cell Physiol 196: 207–218.
Dolcetti R, Serriano D, Masucci MG . (2005). EBV associated malignancies: epidemiology, pathogenesis and therapeutic perspective. In: Thebault S (ed). Progress in virus research Nova Science Publisher Inc: New York, pp 191–226.
Gualandi G, Giselico L, Carloni M, Palitti F, Mosesso P, Alfonsi AM . (2001). Enhancement of genetic instability in human B cells by Epstein–Barr virus latent infection. Mutagenesis 16: 203–208.
Hammerschmidt W, Sugden B . (2004). Epstein–Barr virus sustains Burkitt's lymphomas and Hodgkin's disease. Trends Mol Med 10: 331–336.
Jox A, Rohen C, Belge G, Bartnitzke S, Pawlita M, Diehl V et al. (1997). Integration of Epstein–Barr virus in Burkitt's lymphoma cells leads to a region of enhanced chromosome instability. Ann Oncol 8 (Suppl 2): 131–135.
Karpova MB, Schoumans J, Blennow E, Ernberg I, Henter JI, Smirnov AF et al. (2006). Combined spectral karyotyping, comparative genomic hybridization, and in vitro apoptyping of a panel of Burkitt's lymphoma-derived B cell lines reveals an unexpected complexity of chromosomal aberrations and a recurrence of specific abnormalities in chemoresistant cell lines. Int J Oncol 28: 605–617.
Klein G . (1986). Constitutive activation of oncogenes by chromosomal translocations in B-cell derived tumors. AIDS Res 2 (Suppl 1): S167–176.
Kuhn-Hallek I, Sage DR, Stein L, Groelle H, Fingeroth JD . (1995). Expression of recombination activating genes (RAG-1 and RAG-2) in Epstein–Barr virus-bearing B cells. Blood 85: 1289–1299.
Liu MT, Chang YT, Chen SC, Chuang YC, Chen YR, Lin CS et al. (2005). Epstein–Barr virus latent membrane protein 1 represses p53-mediated DNA repair and transcriptional activity. Oncogene 24: 2635–2646.
Liu MT, Chen YR, Chen SC, Hu CY, Lin CS, Chang YT et al. (2004). Epstein–Barr virus latent membrane protein 1 induces micronucleus formation, represses DNA repair and enhances sensitivity to DNA-damaging agents in human epithelial cells. Oncogene 23: 2531–2539.
Lowndes NF, Toh GW . (2005). DNA repair: the importance of phosphorylating histone H2AX. Curr Biol 15: R99–R102.
Mochida A, Gotoh E, Senpuku H, Harada S, Kitamura R, Takahashi T et al. (2005). Telomere size and telomerase activity in Epstein–Barr virus (EBV)-positive and EBV-negative Burkitt's lymphoma cell lines. Arch Virol 150: 2139–2150.
O'Nions J, Allday MJ . (2003). Epstein–Barr virus can inhibit genotoxin-induced G1 arrest downstream of p53 by preventing the inactivation of CDK2. Oncogene 22: 7181–7191.
Obe G, Pfeiffer P, Savage JR, Johannes C, Goedecke W, Jeppesen P et al. (2002). Chromosomal aberrations: formation, identification and distribution. Mutat Res 504: 17–36.
Pfeffer S, Zavolan M, Grasser FA, Chien M, Russo JJ, Ju J et al. (2004). Identification of virus-encoded microRNAs. Science 304: 734–736.
Potter M, Marcu KB . (1997). The c-myc story: where we've been, where we seem to be going. Curr Top Microbiol Immunol 224: 1–17.
Raptis S, Bapat B . (2006). Genetic instability in human tumors. Exs 96: 303–320.
Rickinson AB, Kieff E . (1996). Epstein–Barr virus. In: Fields BN, Knipe DM, Howley PM (eds). Virology. Lippincott-Raven: Philadelphia, pp 2397–2446.
Rowe M, Rowe DT, Gregory CD, Young LS, Farrell PJ, Rupani H et al. (1987). Differences in B cell growth phenotype reflect novel patterns of Epstein–Barr virus latent gene expression in Burkitt's lymphoma cells. EMBO J 6: 2743–2751.
Schuhmacher M, Staege MS, Pajic A, Polack A, Weidle UH, Bornkamm GW et al. (1999). Control of cell growth by c-Myc in the absence of cell division. Curr Biol 9: 1255–1258.
Shao JY, Huang XM, Yu XJ, Huang LX, Wu QL, Xia JC et al. (2001). Loss of heterozygosity and its correlation with clinical outcome and Epstein–Barr virus infection in nasopharyngeal carcinoma. Anticancer Res 21: 3021–3029.
Smogorzewska A, Karlseder J, Holtgreve-Grez H, Jauch A, de Lange T . (2002). DNA ligase IV-dependent NHEJ of deprotected mammalian telomeres in G1 and G2. Curr Biol 12: 1635–1644.
Stollmann B, Fonatsch C, Havers W . (1985). Persistent Epstein–Barr virus infection associated with monosomy 7 or chromosome 3 abnormality in childhood myeloproliferative disorders. Br J Haematol 60: 183–196.
Thorley-Lawson DA . (2001). Epstein–Barr virus: exploiting the immune system. Nat Rev Immunol 1: 75–82.
Urushibara A, Kodama S, Suzuki K, Desa MB, Suzuki F, Tsutsui T et al. (2004). Involvement of telomere dysfunction in the induction of genomic instability by radiation in SCID mouse cells. Biochem Biophys Res Commun 313: 1037–1043.
Yates JL, Warren N, Sugden B . (1985). Stable replication of plasmids derived from Epstein–Barr virus in various mammalian cells. Nature 313: 812–815.
Young LS, Murray PG . (2003). Epstein–Barr virus and oncogenesis: from latent genes to tumours. Oncogene 22: 5108–5121.
Zattara-Cannoni H, Horshowski N, Figarella-Branger D, Dufour H, Grisoli F, Vagner-Capodano AM . (1996). Unusual chromosome abnormalities in primary central nervous system lymphoma. Leuk Lymphoma 21: 515–517.
Zech L, Haglund U, Nilsson K, Klein G . (1976). Characteristic chromosomal abnormalities in biopsies and lymphoid-cell lines from patients with Burkitt and non-Burkitt lymphomas. Int J Cancer 17: 47–56.
Zimonjic DB, Keck-Waggoner C, Popescu NC . (2001). Novel genomic imbalances and chromosome translocations involving c-myc gene in Burkitt's lymphoma. Leukemia 15: 1582–1588.
Acknowledgements
This study was supported by grants from the Swedish Cancer Society, the Swedish Medical Research Council and the Karolinska Institute, Stockholm, Sweden. SAK is supported by a PhD fellowship awarded by the Iranian Government. AS was supported by personal fellowship awarded by the Cancer Research Institute/Cancer Foundation, New York, USA.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Kamranvar, S., Gruhne, B., Szeles, A. et al. Epstein–Barr virus promotes genomic instability in Burkitt's lymphoma. Oncogene 26, 5115–5123 (2007). https://doi.org/10.1038/sj.onc.1210324
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210324
Keywords
This article is cited by
-
The Epstein–Barr virus nuclear antigen-1 upregulates the cellular antioxidant defense to enable B-cell growth transformation and immortalization
Oncogene (2020)
-
miR-18a reactivates the Epstein-Barr virus through defective DNA damage response and promotes genomic instability in EBV-associated lymphomas
BMC Cancer (2018)
-
Detection of nasopharyngeal carcinoma in Morocco (North Africa) using a multiplex methylation-specific PCR biomarker assay
Clinical Epigenetics (2015)
-
Telomere dysfunction and activation of alternative lengthening of telomeres in B-lymphocytes infected by Epstein–Barr virus
Oncogene (2013)
-
An update on viral association of human cancers
Archives of Virology (2013)