Abstract
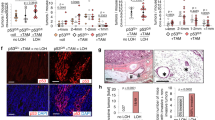
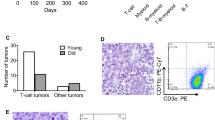
A hallmark in the pathogenesis of cancer is the increased expression of heat shock proteins (Hsps) and other molecular chaperones observed in many tumor types, which is considered to be an adaptive response to enhance tumor cell survival. Heat shock transcription factor 1 (Hsf1) is a major transactivator of Hsp induction and has been proposed to affect tumor initiation and progression, regulating expression of Hsps and other molecular targets. In this report, we provide direct in vivo evidence that Hsf1 plays a critical role in the evolution of spontaneous tumors arising in p53−/− mice. Thus, loss of Hsf1 function did not prolong tumor-free survival, but surprisingly altered the spectrum of tumors that arose in p53−/− mice. Tumor development is rapid in p53−/− mice, which predominantly (about 70%) succumb to lymphomas. In contrast, hsf1−/−p53−/− mice rarely develop lymphomas (<8%), but succumb to other tumor types including testicular carcinomas and soft tissue sarcomas. Our findings suggest that an increase in p53-independent apoptotic cell death in association with altered cytokine signaling and suppressed production of inflammatory factors in hsf1−/− mice may contribute to selective lymphoma suppression. In conclusion, the data presented here link the loss of Hsf1-dependent function to decreased susceptibility to spontaneous lymphomagenesis, which may have implications for cancer prevention and therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bartkova J, Horejsi Z, Koed K, Kramer A, Tort F, Zieger K et al. (2005). DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434: 864–870.
Bukau B, Weissman J, Horwich A . (2006). Molecular chaperones and protein quality control. Cell 125: 443–451.
Ciocca DR, Calderwood SK . (2005). Heat shock proteins in cancer: diagnostic, prognostic, predictive, and treatment implications. Cell Stress Chaperones 10: 86–103.
Clarke AR, Purdie CA, Harrison DJ, Morris RG, Bird CC, Hooper ML et al. (1993). Thymocyte apoptosis induced by p53-dependent and independent pathways. Nature 362: 849–852.
Donehower LA . (1997). Genetic instability in animal tumorigenesis models. Cancer Surv 29: 329–352.
Flohe SB, Bruggemann J, Lendemans S, Nikulina M, Meierhoff G, Flohe S et al. (2003). Human heat shock protein 60 induces maturation of dendritic cells versus a Th1-promoting phenotype. J Immunol 170: 2340–2348.
Fukasawa K, Wiener F, Vande Woude GF, Mai S . (1997). Genomic instability and apoptosis are frequent in p53 deficient young mice. Oncogene 15: 1295–1302.
Hakem R, Hakem A, Duncan GS, Henderson JT, Woo M, Soengas MS et al. (1998). Differential requirement for caspase 9 in apoptotic pathways in vivo. Cell 94: 339–352.
Hanahan D, Weinberg RA . (2000). The hallmarks of cancer. Cell 100: 57–70.
Harvey M, McArthur MJ, Montgomery Jr CA, Butel JS, Bradley A, Donehower LA . (1993). Spontaneous and carcinogen-induced tumorigenesis in p53-deficient mice. Nat Genet 5: 225–229.
Hill R, Song Y, Cardiff RD, Van Dyke T . (2005). Selective evolution of stromal mesenchyme with p53 loss in response to epithelial tumorigenesis. Cell 123: 1001–1011.
Inouye S, Izu H, Takaki E, Suzuki H, Shirai M, Yokota Y et al. (2004). Impaired IgG production in mice deficient for heat shock transcription factor 1. J Biol Chem 279: 38701–38709.
Jacks T, Remington L, Williams BO, Schmitt EM, Halachmi S, Bronson RT et al. (1994). Tumor spectrum analysis in p53-mutant mice. Curr Biol 4: 1–7.
Jiang LI, Nadeau JH . (2001). 129/Sv mice – a model system for studying germ cell biology and testicular cancer. Mamm Genome 12: 89–94.
Jolly C, Morimoto RI . (2000). Role of the heat shock response and molecular chaperones in oncogenesis and cell death. J Natl Cancer Inst 92: 1564–1572.
Karin M, Lawrence T, Nizet V . (2006). Innate immunity gone awry: linking microbial infections to chronic inflammation and cancer. Cell 124: 823–835.
Knowlton AA . (2006). NFkappaB, heat shock proteins, HSF-1, and inflammation. Cardiovasc Res 69: 7–8.
Leonard WJ, Spolski R . (2005). Interleukin-21: a modulator of lymphoid proliferation, apoptosis and differentiation. Nat Rev Immunol 5: 688–698.
Li ZW, Dalton WS . (2006). Tumor microenvironment and drug resistance in hematologic malignancies. Blood Rev 20: 333–342.
Liu G, Parant JM, Lang G, Chau P, Chavez-Reyes A, El-Naggar AK et al. (2004). Chromosome stability, in the absence of apoptosis, is critical for suppression of tumorigenesis in Trp53 mutant mice. Nat Genet 36: 63–68.
Lowe SW, Ruley HE, Jacks T, Housman DE . (1993). p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74: 957–967.
Morimoto RI . (1998). Regulation of the heat shock transcriptional response: cross talk between a family of heat shock factors, molecular chaperones, and negative regulators. Genes Dev 12: 3788–3796.
Oosterhuis JW, Looijenga LH . (2005). Testicular germ-cell tumours in a broader perspective. Nat Rev Cancer 5: 210–222.
Renauld JC, Vink A, Louahed J, Van Snick J . (1995). Interleukin-9 is a major anti-apoptotic factor for thymic lymphomas. Blood 85: 1300–1305.
Takaki E, Fujimoto M, Sugahara K, Nakahari T, Yonemura S, Tanaka Y et al. (2006). Maintenance of olfactory neurogenesis requires HSF1, a major heat shock transcription factor in mice. J Biol Chem 281: 4931–4937.
Wang Y, Theriault JR, He H, Gong J, Calderwood SK . (2004). Expression of a dominant negative heat shock factor-1 construct inhibits aneuploidy in prostate carcinoma cells. J Biol Chem 279: 32651–32659.
Whitesell L, Lindquist SL . (2005). HSP90 and the chaperoning of cancer. Nat Rev Cancer 5: 761–772.
Xiao X, Zuo X, Davis AA, McMillan DR, Curry BB, Richardson JA et al. (1999). HSF1 is required for extra-embryonic development, postnatal growth and protection during inflammatory responses in mice. EMBO J 18: 5943–5952.
Zhang Y, Huang L, Zhang J, Moskophidis D, Mivechi NF . (2002). Targeted disruption of hsf1 leads to lack of thermotolerance and defines tissue-specific regulation for stress-inducible Hsp molecular chaperones. J Cell Biochem 86: 376–393.
Zimonjic DB, Pollock JL, Westervelt P, Popescu NC, Ley TJ . (2000). Acquired, nonrandom chromosomal abnormalities associated with the development of acute promyelocytic leukemia in transgenic mice. Proc Natl Acad Sci USA 97: 13306–13311.
Acknowledgements
This work was supported by NIH grants GM070451 and CA62130 (to NFM) and CA121951 (to DM) and Intramural Research Program of the NIH-NCI, Center for Cancer Research (DBZ).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Min, JN., Huang, L., Zimonjic, D. et al. Selective suppression of lymphomas by functional loss of Hsf1 in a p53-deficient mouse model for spontaneous tumors. Oncogene 26, 5086–5097 (2007). https://doi.org/10.1038/sj.onc.1210317
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210317
Keywords
This article is cited by
-
Heat shock factor 1 inhibition enhances the effects of modulated electro hyperthermia in a triple negative breast cancer mouse model
Scientific Reports (2024)
-
The proteostasis guardian HSF1 directs the transcription of its paralog and interactor HSF2 during proteasome dysfunction
Cellular and Molecular Life Sciences (2021)
-
Interplay between HSF1 and p53 signaling pathways in cancer initiation and progression: non-oncogene and oncogene addiction
Cellular Oncology (2019)
-
Regulation of heat shock transcription factors and their roles in physiology and disease
Nature Reviews Molecular Cell Biology (2018)
-
Cooperative interaction among BMAL1, HSF1, and p53 protects mammalian cells from UV stress
Communications Biology (2018)