Abstract
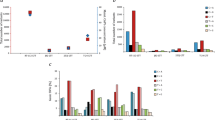
The molecular pathways activated in response to acute cisplatin exposure, as well as the mechanisms involved in the long-term development of cisplatin-resistant cancer cells remain unclear. Using whole genome oligonucleotide microarrays, we have examined the kinetics of gene expression changes in a cisplatin-sensitive cell line, A2780, and its cisplatin-resistant derivative, ACRP. Both sensitive and resistant cell lines exhibited a very similar response of p53-inducible genes as early as 16 h after treatment. This p53 response was further increased at the 24-h time point. These experiments identify p53 as the main pathway producing a large-scale transcriptional response after cisplatin treatment in these cells containing wild-type p53. Consistent with a role for the p53 response in cisplatin sensitivity, knockdown of the p53 protein with small interfering RNA led to a twofold decrease in cell survival in the resistant cells. In addition, our analysis also allowed the identification of several genes that are differentially expressed between sensitive and resistant cells. These genes include GJA1 (encoding connexin 43 (Cx43)) and TWIST1, which are highly upregulated in cisplatin-resistant cells. The importance of Cx43 in drug resistance was demonstrated through functional analyses, although paradoxically, inhibition of Cx43 function in high expressing cells led to an increase in drug resistance. The pathways important in cisplatin response, as well as the genes found differentially expressed between cisplatin-resistant and -sensitive cells, may represent targets for therapy aimed at reversing drug resistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
Accessions
GenBank/EMBL/DDBJ
References
Akervall J, Guo X, Qian CN, Schoumans J, Leeser B, Kort E et al. (2004). Genetic and expression profiles of squamous cell carcinoma of the head and neck correlate with cisplatin sensitivity and resistance in cell lines and patients. Clin Cancer Res 10: 8204–8213.
Amundson SA, Do KT, Vinikoor L, Koch-Paiz CA, Bittner ML, Trent JM et al. (2005). Stress-specific signatures: expression profiling of p53 wild-type and -null human cells. Oncogene 24: 4572–4579.
Azzam EI, de Toledo SM, Gooding T, Little JB . (1998). Intercellular communication is involved in the bystander regulation of gene expression in human cells exposed to very low fluences of alpha particles. Radiat Res 150: 497–504.
Azzam EI, de Toledo SM, Little JB . (2003). Expression of CONNEXIN43 is highly sensitive to ionizing radiation and other environmental stresses. Cancer Res 63: 7128–7135.
Brown R, Clugston C, Burns P, Edlin A, Vasey P, Vojtesek B et al. (1993). Increased accumulation of p53 protein in cisplatin-resistant ovarian cell lines. Int J Cancer 55: 678–684.
Bunz F, Hwang PM, Torrance C, Waldman T, Zhang Y, Dillehay L et al. (1999). Disruption of p53 in human cancer cells alters the responses to therapeutic agents. J Clin Invest 104: 263–269.
Buttitta F, Marchetti A, Gadducci A, Pellegrini S, Morganti M, Carnicelli V et al. (1997). p53 alterations are predictive of chemoresistance and aggressiveness in ovarian carcinomas: a molecular and immunohistochemical study. Br J Cancer 75: 230–235.
Cheng TC, Manorek G, Samimi G, Lin X, Berry CC, Howell SB . (2006). Identification of genes whose expression is associated with cisplatin resistance in human ovarian carcinoma cells. Cancer Chemother Pharmacol 58: 384–395.
Dalton WS . (1999). The tumor microenvironment as a determinant of drug response and resistance. Drug Resist Updat 2: 285–288.
De Feudis P, Debernardis D, Beccaglia P, Valenti M, Graniela Sire E, Arzani D et al. (1997). DDP-induced cytotoxicity is not influenced by p53 in nine human ovarian cancer cell lines with different p53 status. Br J Cancer 76: 474–479.
Dong Y, Walsh MD, McGuckin MA, Cummings MC, Gabrielli BG, Wright GR et al. (1997). Reduced expression of retinoblastoma gene product (pRB) and high expression of p53 are associated with poor prognosis in ovarian cancer. Int J Cancer 74: 407–415.
Einhorn LH . (2002). Curing metastatic testicular cancer. Proc Natl Acad Sci USA 99: 4592–4595.
el-Fouly MH, Trosko JE, Chang CC . (1987). Scrape-loading and dye transfer. A rapid and simple technique to study gap junctional intercellular communication. Exp Cell Res 168: 422–430.
Eliopoulos AG, Kerr DJ, Herod J, Hodgkins L, Krajewski S, Reed JC et al. (1995). The control of apoptosis and drug resistance in ovarian cancer: influence of p53 and Bcl-2. Oncogene 11: 1217–1228.
Fernstrom MJ, Koffler LD, Abou-Rjaily G, Boucher PD, Shewach DS, Ruch RJ . (2002). Neoplastic reversal of human ovarian carcinoma cells transfected with connexin43. Exp Mol Pathol 73: 54–60.
Filmus J . (2001). Glypicans in growth control and cancer. Glycobiology 11: 19R–23R.
Hawkins DS, Demers GW, Galloway DA . (1996). Inactivation of p53 enhances sensitivity to multiple chemotherapeutic agents. Cancer Res 56: 892–898.
Herod JJ, Eliopoulos AG, Warwick J, Niedobitek G, Young LS, Kerr DJ . (1996). The prognostic significance of Bcl-2 and p53 expression in ovarian carcinoma. Cancer Res 56: 2178–2184.
Hough CD, Cho KR, Zonderman AB, Schwartz DR, Morin PJ . (2001). Coordinately up-regulated genes in ovarian cancer. Cancer Res 61: 3869–3876.
Kang HC, Kim IJ, Park JH, Shin Y, Ku JL, Jung MS et al. (2004). Identification of genes with differential expression in acquired drug-resistant gastric cancer cells using high-density oligonucleotide microarrays. Clin Cancer Res 10: 272–284.
Kartalou M, Essigmann JM . (2001). Mechanisms of resistance to cisplatin. Mutat Res 478: 23–43.
Kerley-Hamilton JS, Pike AM, Li N, Direnzo J, Spinella MJ . (2005). A p53-dominant transcriptional response to cisplatin in testicular germ cell tumor-derived human embryonal carcinoma. Oncogene 24: 6090–6100.
Koivusalo R, Krausz E, Ruotsalainen P, Helenius H, Hietanen S . (2002). Chemoradiation of cervical cancer cells: targeting human papillomavirus E6 and p53 leads to either augmented or attenuated apoptosis depending on the platinum carrier ligand. Cancer Res 62: 7364–7371.
Kopf-Maier P, Muhlhausen SK . (1992). Changes in the cytoskeleton pattern of tumor cells by cisplatin in vitro. Chem Biol Interact 82: 295–316.
Li C, Wong WH . (2001). Model-based analysis of oligonucleotide arrays: expression index computation and outlier detection. Proc Natl Acad Sci USA 98: 31–36.
Lin HC, Huber R, Schlessinger D, Morin PJ . (1999). Frequent silencing of the GPC3 gene in ovarian cancer cell lines. Cancer Res 59: 807–810.
Lin JH, Yang J, Liu S, Takano T, Wang X, Gao Q et al. (2003). Connexin mediates gap junction-independent resistance to cellular injury. J Neurosci 23: 430–441.
Livak KJ, Schmittgen TD . (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25: 402–408.
Lowe SW, Ruley HE, Jacks T, Housman DE . (1993). p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74: 957–967.
Maestro R, Dei Tos AP, Hamamori Y, Krasnokutsky S, Sartorelli V, Kedes L et al. (1999). Twist is a potential oncogene that inhibits apoptosis. Genes Dev 13: 2207–2217.
Moorehead RA, Singh G . (2000). Influence of the proto-oncogene c-fos on cisplatin sensitivity. Biochem Pharmacol 59: 337–345.
Morin PJ . (2003). Drug resistance and the microenvironment: nature and nurture. Drug Resist Updat 6: 169–172.
O'Connor PM, Jackman J, Bae I, Myers TG, Fan S, Mutoh M et al. (1997). Characterization of the p53 tumor suppressor pathway in cell lines of the National Cancer Institute anticancer drug screen and correlations with the growth-inhibitory potency of 123 anticancer agents. Cancer Res 57: 4285–4300.
Ozols RF, Young RC . (1984). Chemotherapy of ovarian cancer. Semin Oncol 11: 251–263.
Pestell KE, Hobbs SM, Titley JC, Kelland LR, Walton MI . (2000). Effect of p53 status on sensitivity to platinum complexes in a human ovarian cancer cell line. Mol Pharmacol 57: 503–511.
Pestell KE, Medlow CJ, Titley JC, Kelland LR, Walton MI . (1998). Characterisation of the p53 status, BCL-2 expression and radiation and platinum drug sensitivity of a panel of human ovarian cancer cell lines. Int J Cancer 77: 913–918.
Reles A, Wen WH, Schmider A, Gee C, Runnebaum IB, Kilian U et al. (2001). Correlation of p53 mutations with resistance to platinum-based chemotherapy and shortened survival in ovarian cancer. Clin Cancer Res 7: 2984–2997.
Roberts D, Schick J, Conway S, Biade S, Laub PB, Stevenson JP et al. (2005). Identification of genes associated with platinum drug sensitivity and resistance in human ovarian cancer cells. Br J Cancer 92: 1149–1158.
Ryan KM, Phillips AC, Vousden KH . (2001). Regulation and function of the p53 tumor suppressor protein. Curr Opin Cell Biol 13: 332–337.
Sax JK, El-Deiry WS . (2003). p53 downstream targets and chemosensitivity. Cell Death Differ 10: 413–417.
Shen DW, Liang XJ, Gawinowicz MA, Gottesman MM . (2004). Identification of cytoskeletal [14C]carboplatin-binding proteins reveals reduced expression and disorganization of actin and filamin in cisplatin-resistant cell lines. Mol Pharmacol 66: 789–793.
Sherman-Baust CA, Weeraratna AT, Rangel LBA, Pizer ES, Cho KR, Schwartz DR et al. (2003). Remodeling of the extracellular matrix through overexpression of collagen VI contributes to cisplatin resistance in ovarian cancer cells. Cancer Cell 3: 377–386.
Siddik ZH . (2003). Cisplatin: mode of cytotoxic action and molecular basis of resistance. Oncogene 22: 7265–7279.
Takata R, Katagiri T, Kanehira M, Tsunoda T, Shuin T, Miki T et al. (2005). Predicting response to methotrexate, vinblastine, doxorubicin, and cisplatin neoadjuvant chemotherapy for bladder cancers through genome-wide gene expression profiling. Clin Cancer Res 11: 2625–2636.
Tanaka T, Yamasaki H, Mesnil M . (2001). Induction of a bystander effect in HeLa cells by using a bigenic vector carrying viral thymidine kinase and connexin32 genes. Mol Carcinog 30: 176–180.
Thiel G, Cibelli G . (2002). Regulation of life and death by the zinc finger transcription factor Egr-1. J Cell Physiol 193: 287–292.
Varma RR, Hector SM, Clark K, Greco WR, Hawthorn L, Pendyala L . (2005). Gene expression profiling of a clonal isolate of oxaliplatin-resistant ovarian carcinoma cell line A2780/C10. Oncol Rep 14: 925–932.
Wang X, Ling MT, Guan XY, Tsao SW, Cheung HW, Lee DT et al. (2004). Identification of a novel function of TWIST, a bHLH protein, in the development of acquired taxol resistance in human cancer cells. Oncogene 23: 474–482.
Wernyj RP, Morin PJ . (2004). Molecular mechanisms of platinum resistance: still searching for the Achilles' heel. Drug Resist Updat 7: 227–232.
Whiteside MA, Chen DT, Desmond RA, Abdulkadir SA, Johanning GL . (2004). A novel time-course cDNA microarray analysis method identifies genes associated with the development of cisplatin resistance. Oncogene 23: 744–752.
Wu GS, El-Deiry WS . (1996). Apoptotic death of tumor cells correlates with chemosensitivity, independent of p53 or bcl-2. Clin Cancer Res 2: 623–633.
Acknowledgements
We thank Drs Michael Pazin and Myriam Gorospe, as well as members of our laboratory for useful comments on the manuscript. We thank Dr Kotb Abdelmohsen for help with the scrape-loading/dye transfer assays. This research was supported by the Intramural Research Program of the NIH, National Institute on Aging.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Li, J., Wood, W., Becker, K. et al. Gene expression response to cisplatin treatment in drug-sensitive and drug-resistant ovarian cancer cells. Oncogene 26, 2860–2872 (2007). https://doi.org/10.1038/sj.onc.1210086
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210086
Keywords
This article is cited by
-
Gene networks and expression quantitative trait loci associated with adjuvant chemotherapy response in high-grade serous ovarian cancer
BMC Cancer (2020)
-
Functional and transcriptomic characterization of carboplatin-resistant A2780 ovarian cancer cell line
Biological Research (2019)
-
Akt inhibitor SC66 promotes cell sensitivity to cisplatin in chemoresistant ovarian cancer cells through inhibition of COL11A1 expression
Cell Death & Disease (2019)
-
miRNA-302s may act as oncogenes in human testicular germ cell tumours
Scientific Reports (2019)
-
Concerted changes in transcriptional regulation of genes involved in DNA methylation, demethylation, and folate-mediated one-carbon metabolism pathways in the NCI-60 cancer cell line panel in response to cancer drug treatment
Clinical Epigenetics (2016)