Abstract
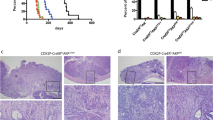
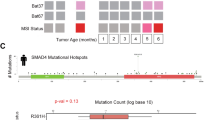
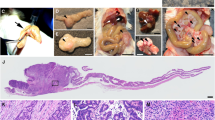
The Smad4+/E6sad mouse carries a null mutation in the endogenous Smad4 gene resulting in serrated adenomas and mixed polyposis of the upper gastrointestinal (GI) tract with 100% penetrance. Here, we show by loss of heterozygosity (LOH) analysis and immunohistochemistry (IHC) that, although the majority of the tumors appear at 9 months of age, somatic loss of the wild-type Smad4 allele occurs only at later stages of tumor progression. Hence, haploinsufficiency underlies Smad4-driven tumor initiation in the GI tract. As both the Apc and Smad4 tumor suppressor genes map to mouse chromosome 18, we have bred Smad4+/E6sad with the Apc+/1638N model to generate two distinct compound heterozygous lines carrying both mutations either in cis (CAS) or in trans (TAS). Strikingly, both models show increased tumor multiplicities when compared with the single mutant littermates, although CAS mice are more severely affected and became moribund at only 5–6 weeks of age. Phenotypic and molecular analyses indicate that Smad4 haploinsufficiency is sufficient to significantly affect tumor initiation and progression both prior to and upon loss of Apc function. Moreover, complete loss of Smad4 strongly enhances Apc-driven tumor formation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Boivin GP, Washington K, Yang K, Ward JM, Pretlow TP, Russell R et al. (2003). Gastroenterology 124: 762–777.
Fearon ER, Vogelstein B . (1990). Cell 61: 759–767.
Fink SP, Mikkola D, Willson JK, Markowitz S . (2003). Oncogene 22: 1317–1323.
Fodde R, Edelmann W, Yang K, van Leeuwen C, Carlson C, Renault B et al. (1994). Proc Natl Acad Sci USA 91: 8969–8973.
Gaspar C, Fodde R . (2004). Int J Dev Biol 48: 377–386.
Haigis KM, Dove WF . (2003). Nat Genet 33: 33–39.
Haramis AP, Begthel H, van den Born M, van Es J, Jonkheer S, Offerhaus GJ et al. (2004). Science 303: 1684–1686.
Hawkins NJ, Bariol C, Ward RL . (2002). Pathology 34: 548–555.
He XC, Zhang J, Tong WG, Tawfik O, Ross J, Scoville DH et al. (2004). Nat Genet 36: 1117–1121.
Hohenstein P, Molenaar L, Elsinga J, Morreau H, van der Klift H, Struijk A et al. (2003). Genes Chromosomes Cancer 36: 273–282.
Howe JR, Roth S, Ringold JC, Summers RW, Jarvinen HJ, Sistonen P et al. (1998). Science 280: 1086–1088.
Itoh S, Itoh F, Goumans MJ, Ten Dijke P . (2000). Eur J Biochem 267: 6954–6967.
Jass JR . (2001). J Pathol 193: 283–285.
Kielman MF, Rindapaa M, Gaspar C, van Poppel N, Breukel C, van Leeuwen S et al. (2002). Nat Genet 32: 594–605.
Koh TJ, Bulitta CJ, Fleming JV, Dockray GJ, Varro A, Wang TC . (2000). J Clin Invest 106: 533–539.
Koyama M, Ito M, Nagai H, Emi M, Moriyama Y . (1999). Mutat Res 406: 71–77.
Lei S, Dubeykovskiy A, Chakladar A, Wojtukiewicz L, Wang TC . (2004). J Biol Chem 279: 42492–42502.
Makinen MJ, George SM, Jernvall P, Makela J, Vihko P, Karttunen TJ . (2001). J Pathol 193: 286–294.
Miyaki M, Iijima T, Konishi M, Sakai K, Ishii A, Yasuno M et al. (1999). Oncogene 18: 3098–3103.
Salovaara R, Roth S, Loukola A, Launonen V, Sistonen P, Avizienyte E et al. (2002). Gut 51: 56–59.
Satterwhite DJ, Neufeld KL . (2004). Cell Cycle 3: 1069–1073.
Shioda T, Lechleider RJ, Dunwoodie SL, Li H, Yahata T, de Caestecker MP et al. (1998). Proc Natl Acad Sci USA 95: 9785–9790.
Sirard C, de la Pompa JL, Elia A, Itie A, Mirtsos C, Cheung A et al. (1998). Genes Dev 12: 107–119.
Smits R, Kartheuser A, Jagmohan-Changur S, Leblanc V, Breukel C, de Vries A et al. (1997). Carcinogenesis 18: 321–327.
Smits R, Kielman MF, Breukel C, Zurcher C, Neufeld K, Jagmohan-Changur S et al. (1999). Genes Dev 13: 1309–1321.
Smits R, van der Houven van Oordt W, Luz A, Zurcher C, Jagmohan-Changur S, Breukel C et al. (1998). Gastroenterology 114: 275–283.
Takagi Y, Kohmura H, Futamura M, Kida H, Tanemura H, Shimokawa K et al. (1996). Gastroenterology 111: 1369–1372.
Takaku K, Miyoshi H, Matsunaga A, Oshima M, Sasaki N, Taketo MM . (1999). Cancer Res 59: 6113–6117.
Takaku K, Oshima M, Miyoshi H, Matsui M, Seldin MF, Taketo MM . (1998). Cell 92: 645–656.
Thiagalingam S, Lengauer C, Leach FS, Schutte M, Hahn SA, Overhauser J et al. (1996). Nat Genet 13: 343–346.
Wilentz RE, Su GH, Dai JL, Sparks AB, Argani P, Sohn TA et al. (2000). Am J Pathol 156: 37–43.
Woodford-Richens K, Williamson J, Bevan S, Young J, Leggett B, Frayling I et al. (2000). Cancer Res 60: 2477–2482.
Xu X, Brodie SG, Yang X, Im YH, Parks WT, Chen L et al. (2000). Oncogene 19: 1868–1874.
Acknowledgements
This work has been made possible by funds from the Dutch Cancer Society, the Dutch Research Council (NWO VICI), and BSIK (ICES/KIS-3). PH is supported by a grant from the Association for International cancer Research (AICR).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alberici, P., Jagmohan-Changur, S., De Pater, E. et al. Smad4 haploinsufficiency in mouse models for intestinal cancer. Oncogene 25, 1841–1851 (2006). https://doi.org/10.1038/sj.onc.1209226
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209226
Keywords
This article is cited by
-
A pan-cancer analysis reveals nonstop extension mutations causing SMAD4 tumour suppressor degradation
Nature Cell Biology (2020)
-
Mitochondrial genome instability resulting from SUV3 haploinsufficiency leads to tumorigenesis and shortened lifespan
Oncogene (2013)
-
Deleted in colorectal carcinoma suppresses metastasis in p53-deficient mammary tumours
Nature (2012)
-
MicroRNAs in colorectal cancer stem cells: new regulators of cancer stemness?
Oncogenesis (2012)