Abstract
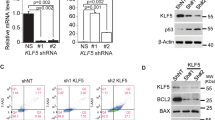
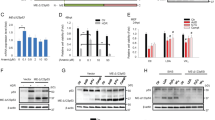
Following genotoxic stress, p53 either rescues a damaged cell or promotes its elimination. The parameters determining a specific outcome of the p53 response are largely unknown. In mouse fibroblasts treated with different irradiation schemes, we monitored transcriptional and non-transcriptional p53 activities and identified determinants that initiate an anti- or a pro-apoptotic p53 response within the context of p53-independent stress signaling. The primary, transcription-mediated p53 response in these cells is anti-apoptotic, while induction of p53-dependent apoptosis requires an additional, transcription-independent p53 activity, provided by high intracellular levels of activated p53. High intracellular levels of p53 were selectively generated after apoptosis-inducing high-dose UV-irradiation, and correlated with a strongly delayed upregulation of Mdm2. Following high-dose UV-irradiation, p53 accumulated in the cytoplasm and led to activation of the pro-apoptotic protein Bax. As p53-dependent Bax-activation is transcription-independent, we postulated that certain transcription-deficient mutant p53 proteins might also exert this activity. Indeed we found an endogenous, transcription-inactive mutant p53 that upon genotoxic stress induced Bax-activation in vivo. Our results demonstrate the impact and in vivo relevance of non-transcriptional mechanisms for wild-type and mutant p53-mediated apoptosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Chen X, Ko LJ, Jayaraman L, Prives C . (1996). Genes Dev 10: 2438–2451.
Chen X, Zhang W, Gao YF, Su XQ, Zhai ZH . (2002). Cell Res 12: 229–233.
Chipuk JE, Maurer U, Green DR, Schuler M . (2003). Cancer Cell 4: 371–381.
Chipuk JE, Kuwana T, Bouchier-Hayes L, Droin NM, Newmeyer DD, Schuler M et al. (2004). Science 303: 1010–1014.
Cory S, Adams JM . (2002). Nat Rev Cancer 2: 647–656.
Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C et al. (1995). Proc Natl Acad Sci USA 92: 9363–9367.
Dumont P, Leu JIJ, Della Pietra III AC, George DL, Murphy M . (2003). Nat Genet 33: 357–365.
El-Deiry WS . (2003). Oncogene 22: 7486–7495.
El-Deiry WS, Tokino T, Velculescu VE, Levy DB, Parsons R, Trent JM et al. (1993). Cell 75: 817–825.
Fridman JS, Lowe SW . (2003). Oncogene 22: 9030–9040.
Gartel AL, Tyner AL . (2002). Mol Cancer Ther 1: 639–649.
Harvey DM, Levine AJ . (1991). Genes Dev 5: 2375–2385.
Hermeking H . (2003). Nat Rev Cancer 2: 594–604.
Hermeking H, Lengauer C, Polyak K, Tong-Chuan H, Zhang L, Thiagalingam S et al. (1997). Mol Cell 1: 3–11.
Inga A, Storici F, Darden TA, Resnick MA . (2002). Mol Cell Biol 22: 8612–8625.
Kaeser MD, Iggo RD . (2002). Proc Natl Acad Sci USA 99: 95–100.
Kim E, Rohaly G, Heinrichs S, Gimnopoulos D, Meissner H, Deppert W . (1999). Oncogene 18: 7310–7318.
Leu JIJ, Dumont P, Hafey M, Murphy ME, George DL . (2004). Nat Cell Biol. 6: 443–450.
Li M, Brooks CL, Wu-Baer F, Chen D, Baer R, Gu W . (2003). Science 302: 1972–1975.
Marchenko ND, Zaika A, Moll UM . (2000). J Biol Chem 275: 16202–16212.
Michael D, Oren M . (2003). Semin Cancer Biol 13: 49–58.
Michalovitz D, Eliyahu D, Oren M . (1990). Cell 62: 671–680.
Mihara M, Erster S, Zaika A, Petrenko O, Chittenden T, Pancoska P et al. (2003). Mol Cell 11: 577–590.
Milner J, Chan YS, Medcalf EA, Wang Y, Eckhart W . (1993). Oncogene 8: 2001–2008.
Miyashita T, Reed JC . (1995). Cell 80: 293–299.
Nakano K, Vousden KH . (2001). Mol Cell 7: 683–694.
Pelizon C, d'Adda di Fagagna F, Farrace L, Laskey RA . (2002). EMBO Rep 3: 780–784.
Polyak K, Xia Y, Zweier JL, Kinzler B, Vogelstein B . (1997). Nature 389: 300–305.
Ronen D, Schwartz D, Teitz Y, Goldfinger N, Rotter V . (1996). Cell Growth Differ 7: 21–30.
Roninson IB . (2003). Cancer Res 63: 2705–2715.
Vousden KH, Lu X . (2002). Nat Rev Cancer 2: 594–604.
Yu J, Zhang L, Hwang PM, Kinzler KW, Vogelstein B . (2001). Mol Cell 7: 673–682.
Ziegler C . (1998). Der Tumorsuppressor p53 als auxiliäres Onkogen in der Simian Virus 40-induzierten Zelltransformation. Dissertation, Universität Hamburg: Hamburg, Germany.
Acknowledgements
We thank H Lother for invaluable support, J Milner, AJ Levine, F Fackelmayer, E Kim, I Dornreiter and A Hermannstädter for providing cell lines and reagents, C Stocking and F Fackelmayer for critically reading the manuscript, E Kim for insightful discussions and M Hintz-Malchow for her competent help in editing this manuscript. This work was supported by Deutsche Krebshilfe/Förderungsschwerpunkt ‘Apoptosedefizienz und ihre Modulation bei malignen Erkrankungen’, by the Fonds der Chemischen Industrie, and by EC FP6 funding. This publication reflects the author's views and not necessarily those of the EC. The community is not liable for any use that may be made of the information contained herein. The Heinrich-Pette-Institut is financially supported by Freie und Hansestadt Hamburg, and by Bundesministerium für Gesundheit und Soziales.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary information accompanies the paper on Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Speidel, D., Helmbold, H. & Deppert, W. Dissection of transcriptional and non-transcriptional p53 activities in the response to genotoxic stress. Oncogene 25, 940–953 (2006). https://doi.org/10.1038/sj.onc.1209126
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209126
Keywords
This article is cited by
-
Spatial regulation and generation of diversity in signaling pathways
Journal of Biosciences (2021)
-
IFN-gamma-induced PD-L1 expression in melanoma depends on p53 expression
Journal of Experimental & Clinical Cancer Research (2019)
-
Etoposide induces cell death via mitochondrial-dependent actions of p53
Cancer Cell International (2015)
-
Hyperglycemia promotes p53-Mdm2 interaction but reduces p53 ubiquitination in RINm5F cells
Molecular and Cellular Biochemistry (2015)
-
The role of DNA damage responses in p53 biology
Archives of Toxicology (2015)