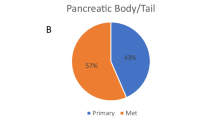
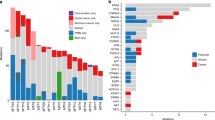
Abstract
Pancreatic ductal adenocarcinoma (PDAC) carries the most dismal prognosis of all solid tumours. Both the late clinical presentation of patients, due to lack of early symptoms, as well as the rapid and aggressive course of the disease contribute to the extremely high mortality of this malignancy. Recently, a multistep progression model for PDAC integrating morphological, clinical and molecular evidence has been proposed. Putative precursor lesions, termed pancreatic intraepithelial neoplasia (PanIN), are classified into three different grades (PanIN-1 through -3) based on the degree of cellular atypia they display. We have conducted large-scale expression profiling analyses of microdissected cells from normal pancreatic ducts, PanINs of different grades and PDACs using whole-genome oligonucleotide microarrays. Verification of hybridisation results for selected genes was performed using quantitative real-time PCR and immunohistochemical analyses on PanIN tissue microarrays. Comparison of the expression profiles demonstrated that the greatest changes in gene expression occur between PanIN stages 1B and 2, suggesting that PanIN-2 may represent the first truly preneoplastic stage in PDAC progression. Our results identify a large number of potential target genes for the development of novel molecular diagnostic and therapeutic tools for the prevention and early diagnosis of PDAC and provide novel insights into the pathophysiological mechanisms involved in tumour progression in the pancreas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bramhall SR, Neoptolemos JP, Stamp GW and Lemoine NR . (1997). J. Pathol., 182, 347–355.
Brentnall TA, Bronner MP, Byrd DR, Haggitt RC and Kimmey MB . (1999). Ann. Intern. Med., 131, 247–255.
Bubendorf L, Nocito A, Moch H and Sauter G . (2001). J. Pathol., 195, 72–79.
Buchholz M, Biebl A, Neesse A, Wagner M, Iwamura T, Leder G, Adler G and Gress TM . (2003). Cancer Res., 63, 4945–4951.
Canto MI, Goggins M, Yeo CJ, Griffin C, Axilbund JE, Brune K, Ali SZ, Jagannath S, Petersen GM, Fishman EK, Piantadosi S, Giardiello FM and Hruban RH . (2004). Clin. Gastroenterol. Hepatol., 2, 606–621.
Conlon KC, Klimstra DS and Brennan MF . (1996). Ann. Surg., 223, 273–279.
Crnogorac-Jurcevic T, Missiaglia E, Blaveri E, Gangeswaran R, Jones M, Terris B, Costello E, Neoptolemos JP and Lemoine NR . (2003). J. Pathol., 201, 63–74.
Ellenrieder V, Alber B, Lacher U, Hendler SF, Menke A, Boeck W, Wagner M, Wilda M, Friess H, Buchler M, Adler G and Gress TM . (2000). Int. J. Cancer, 85, 14–20.
Fearon ER and Vogelstein B . (1990). Cell, 61, 759–767.
Goggins M, Hruban RH and Kern SE . (2000). Am. J. Pathol., 156, 1767–1771.
Gronborg M, Bunkenborg J, Kristiansen TZ, Jensen ON, Yeo CJ, Hruban RH, Maitra A, Goggins MG and Pandey A . (2004). J. Proteome Res., 3, 1042–1055.
Heinmoller E, Dietmaier W, Zirngibl H, Heinmoller P, Scaringe W, Jauch KW, Hofstadter F and Ruschoff J . (2000). Am. J. Pathol., 157, 83–92.
Hruban RH, Adsay NV, Albores-Saavedra J, Compton C, Garrett ES, Goodman SN, Kern SE, Klimstra DS, Kloppel G, Longnecker DS, Luttges J and Offerhaus GJ . (2001a). Am. J. Surg. Pathol., 25, 579–586.
Hruban RH, Adsay NV, Albores-Saavedra J, Compton C, Garrett ES, Goodman SN, Kern SE, Klimstra DS, Kloppel G, Longnecker DS, Luttges J and Offerhaus GJ . (2001b). Am. J. Surg. Pathol., 25, 579–586.
Hruban RH, Canto MI and Yeo CJ . (2001c). Dig. Dis., 19, 76–84.
Hruban RH, Takaori K, Klimstra DS, Adsay NV, Albores-Saavedra J, Biankin AV, Biankin SA, Compton C, Fukushima N, Furukawa T, Goggins M, Kato Y, Kloppel G, Longnecker DS, Luttges J, Maitra A, Offerhaus GJ, Shimizu M and Yonezawa S . (2004). Am. J. Surg. Pathol., 28, 977–987.
Hruban RH, Wilentz RE and Kern SE . (2000). Am. J. Pathol., 156, 1821–1825.
Huflejt ME and Leffler H . (2004). Glycoconj. J., 20, 247–255.
Kawakami K, Kawakami M, Joshi BH and Puri RK . (2001). Cancer Res., 61, 6194–6200.
Kloppel G and Luttges J . (2004). Curr. Gastroenterol. Rep., 6, 111–118.
Kos J, Werle B, Lah T and Brunner N . (2000). Int. J. Biol. Markers, 15, 84–89.
Kristiansen G, Pilarsky C, Wissmann C, Kaiser S, Bruemmendorf T, Roepcke S, Dahl E, Hinzmann B, Specht T, Pervan J, Stephan C, Loening S, Dietel M and Rosenthal A . (2004). J. Pathol., 205, 359–376.
Liotta LA and Kohn EC . (2001). Nature, 411, 375–379.
Luttges J, Diederichs A, Menke MA, Vogel I, Kremer B and Kloppel G . (2000). Cancer, 88, 2495–2504.
Luttges J, Galehdari H, Brocker V, Schwarte-Waldhoff I, Henne-Bruns D, Kloppel G, Schmiegel W and Hahn SA . (2001). Am. J. Pathol., 158, 1677–1683.
Luttges J, Reinecke-Luthge A, Mollmann B, Menke MA, Clemens A, Klimpfinger M, Sipos B and Kloppel G . (1999). Virchows. Arch., 435, 461–468.
Luttges J, Stigge C, Pacena M and Kloppel G . (2004). Cancer, 100, 173–182.
Lynch HT . (1994). Arch. Surg., 129, 266–268.
Lynch HT, Fitzsimmons ML, Smyrk TC, Lanspa SJ, Watson P, McClellan J and Lynch JF . (1990). Am. J. Gastroenterol., 85, 54–60.
Lynch HT, Smyrk T, Kern SE, Hruban RH, Lightdale CJ, Lemon SJ, Lynch JF, Fusaro LR, Fusaro RM and Ghadirian P . (1996). Semin. Oncol., 23, 251–275.
Maitra A, Adsay NV, Argani P, Iacobuzio-Donahue C, De Marzo A, Cameron JL, Yeo CJ and Hruban RH . (2003). Mod. Pathol., 16, 902–912.
Neoptolemos JP, Stocken DD, Friess H, Bassi C, Dunn JA, Hickey H, Beger H, Fernandez-Cruz L, Dervenis C, Lacaine F, Falconi M, Pederzoli P, Pap A, Spooner D, Kerr DJ and Buchler MW . (2004). N. Engl. J. Med., 350, 1200–1210.
Parkin DM, Pisani P and Ferlay J . (1999). CA Cancer J. Clin., 49, 33–64 1.
Pupa SM, Menard S, Forti S and Tagliabue E . (2002). J. Cell Physiol., 192, 259–267.
Smyth GK . (2004). Statistical Applications in Genetics and Molecular Biology, 3, Article 3.
Sperti C, Pasquali C, Piccoli A and Pedrazzoli S . (1997). World J. Surg., 21, 195–200.
Stepanova L, Yang G, DeMayo F, Wheeler TM, Finegold M, Thompson TC and Harper JW . (2000). Oncogene, 19, 2186–2193.
Strojan P, Svetic B, Smid L and Kos J . (2004). Clin. Chim. Acta, 344, 155–161.
Sturn A, Quackenbush J and Trajanoski Z . (2002). Bioinformatics, 18, 207–208.
Suomela S, Cao L, Bowcock A and Saarialho-Kere U . (2004). J. Invest. Dermatol., 122, 717–721.
Terris B, Blaveri E, Crnogorac-Jurcevic T, Jones M, Missiaglia E, Ruszniewski P, Sauvanet A and Lemoine NR . (2002). Am. J. Pathol., 160, 1745–1754.
Trede M, Richter A and Wendl K . (2001). Surg. Clin. N Am., 81, 595–610.
Tusher VG, Tibshirani R and Chu G . (2001). Proc. Natl. Acad. Sci. USA, 98, 5116–5121.
Vimalachandran D, Ghaneh P, Costello E and Neoptolemos JP . (2004). Cancer Control, 11, 6–14.
Wilentz RE, Geradts J, Maynard R, Offerhaus GJ, Kang M, Goggins M, Yeo CJ, Kern SE and Hruban RH . (1998). Cancer Res., 58, 4740–4744.
Wilentz RE, Iacobuzio-Donahue CA, Argani P, McCarthy DM, Parsons JL, Yeo CJ, Kern SE and Hruban RH . (2000). Cancer Res., 60, 2002–2006.
Yamakawa T, Miyata S, Ogawa N, Koshikawa N, Yasumitsu H, Kanamori T and Miyazaki K . (1998). Biochim. Biophys. Acta, 1395, 202–208.
Yeo CJ and Cameron JL . (1998). Langenbecks Arch. Surg., 383, 129–133.
Zeeberg BR, Feng W, Wang G, Wang MD, Fojo AT, Sunshine M, Narasimhan S, Kane DW, Reinhold WC, Lababidi S, Bussey KJ, Riss J, Barrett JC and Weinstein JN . (2003). Genome Biol., 4, R28.
Acknowledgements
We thank Susanne Braun, Karin Lanz, Claudia Ruhland, Pat Schreiter and Britta Redeker for excellent technical assistance. The work of the GPCN is supported by a multicentre grant form the Deutsche Krebshilfe, Bonn, Germany.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Buchholz, M., Braun, M., Heidenblut, A. et al. Transcriptome analysis of microdissected pancreatic intraepithelial neoplastic lesions. Oncogene 24, 6626–6636 (2005). https://doi.org/10.1038/sj.onc.1208804
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208804
Keywords
This article is cited by
-
Comprehensive analysis of expression profile and prognostic significance of interferon regulatory factors in pancreatic cancer
BMC Genomic Data (2022)
-
HELLS serves as a poor prognostic biomarker and its downregulation reserves the malignant phenotype in pancreatic cancer
BMC Medical Genomics (2021)
-
Apoptosis-associated speck-like protein containing a CARD regulates the growth of pancreatic ductal adenocarcinoma
Scientific Reports (2021)
-
Molecular profiling of stroma highlights stratifin as a novel biomarker of poor prognosis in pancreatic ductal adenocarcinoma
British Journal of Cancer (2020)
-
Transient receptor potential ion-channel subfamily V member 4: a potential target for cancer treatment
Cell Death & Disease (2019)