Abstract
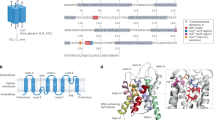
Aquaporins (AQPs) are important in controlling water permeability. As AQP1 is known as a serum-responsive gene, we hypothesized that AQP expression may be involved in the development of human cancer. By reverse transcriptase–polymerase chain reaction analysis, expression of AQPs 1, 3, and 5 was found in seven colon and colorectal cancer cell lines. Western blot analysis confirmed their expression in four of these cell lines. In situ hybridization demonstrated that during colorectal carcinogenesis, the expression of AQPs 1 and 5 was induced in early-stage disease (early dysplasia) and maintained through the late stages of colon cancer development. Expression of AQPs 1 and 5 was maintained even in metastatic lesions in the liver. These findings demonstrate that the expression of several AQPs is found in tumor cells and is associated with an early stage of colorectal cancer development. These novel observations suggest that multiple AQP expression may be advantageous to tumorigenesis, which may lead to a better understanding of colorectal carcinogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Agre P, Bonhivers M and Borgnia MJ . (1998). J. Biol. Chemother., 273, 14659–14662.
Agre P, Brown D and Nielsen S . (1995). Curr. Opin. Cell Biol., 7, 472–483.
Bai C, Fukuda N, Song Y, Ma T, Matthay MA and Verkman AS . (1999). J. Clin. Invest., 103, 555–561.
Calamita G, Kempf B, Bonhivers M, Bishai WR, Bremer E and Agre P . (1998). Proc. Natl. Acad. Sci. USA, 95, 3627–3631.
Deen PMT, Verdijk MAJ, Knoers NVA, Wieringa B, Monnens LAH, van Os CH and van Oost BA . (1994). Science, 264, 92–94.
Fearon ER and Vogelstein B . (1991). Cell, 61, 759–762.
Heymann JB, Agre P and Engel A . (1998). J. Struct. Biol., 121, 191–206.
King LS and Agre P . (1996). Annu. Rev. Physiol., 158, 619–648.
King LS, Nielsen S and Agre P . (1997). Am. J. Physiol., 273, C1541–C1548.
King LS, Yasui M and Agre P . (2000). Mol. Med. Today, 6, 60–65.
Ma T and Verkman AS . (1999). J. Physiol., 517, 317–326.
Marples D . (2000). Lancet, 355, 1571–1572.
Maurel C . (1997). Annu. Rev. Plant Physiol. Plant Mol. Biol., 48, 399–429.
Moon C, King LS and Agre P . (1997). Am. J. Physiol., 273, C1562–C1570.
Moon C, Preston GM, Griffin CA, Jabs EW and Agre P . (1993). J. Biol. Chem., 268, 15772–15778.
Moon C, Williams JB, Preston GM, Copeland NG, Gilbert DJ, Nathans D, Jenkins NA and Agre P . (1995). Genomics, 30, 354–357.
Nakhoul NL, Davis BA, Romero MF and Boron WF . (1998). Am. J. Physiol., 274, C543–C548.
Nielsen S, King LS, Christensen BM and Agre P . (1997). Am. J. Physiol., 273, C1549–C1561.
Preston GM, Carroll TP, Guggino WB and Agre P . (1992). Science, 256, 385–387.
Preston GM, Smith BL, Zeidel ML and Moulds JJ . (1994). Science, 265, 1585–1587.
Saadoun S, Papadopoulos MC, Davies DC, Bell BA and Krishna S . (2002). Br. J. Cancer, 87, 621–623.
Splinter PL, Masyuk AI and LaRusso NF . (2002). J. Biol. Chem., 278, 6268–6274.
Zheng X and Chen X . (2001). FEBS Lett., 489, 4–7.
Acknowledgements
We thank Dr Li K Su, who provided total RNA for the three colon cell lines used in this study. The study was supported in part by NIH/NCI Grant P50 CA96784-01 (to CM), American Cancer Society Grant RPG-98-054 (to LM), Fondation de France, AP-HP and Lilly Fondation Grant (to J-CS), Cancer Center Grant P30 CA 16620 (to MD Anderson Cancer Center), and Tobacco Research Fund from State of Texas (to MD Anderson Cancer Center).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Moon, C., Soria, JC., Jang, S. et al. Involvement of aquaporins in colorectal carcinogenesis. Oncogene 22, 6699–6703 (2003). https://doi.org/10.1038/sj.onc.1206762
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206762
Keywords
This article is cited by
-
The role of Aquaporins in tumorigenesis: implications for therapeutic development
Cell Communication and Signaling (2024)
-
Mesenchymal stem cells- derived exosomes inhibit the expression of Aquaporin-5 and EGFR in HCT-116 human colorectal carcinoma cell line
BMC Molecular and Cell Biology (2022)
-
CTpathway: a CrossTalk-based pathway enrichment analysis method for cancer research
Genome Medicine (2022)
-
Aquaporin ion conductance properties defined by membrane environment, protein structure, and cell physiology
Biophysical Reviews (2022)
-
Transcriptome profiling by combined machine learning and statistical R analysis identifies TMEM236 as a potential novel diagnostic biomarker for colorectal cancer
Scientific Reports (2021)