Abstract
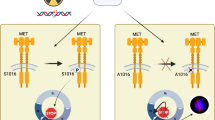
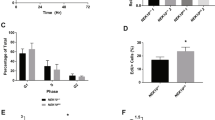
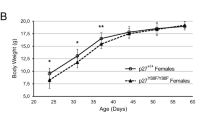
The c-Abl protein tyrosine kinase is activated by ionizing radiation (IR) and certain other DNA-damaging agents. The present studies demonstrate that c-Abl associates constitutively with protein kinase C δ (PKCδ). The results show that the SH3 domain of c-Abl interacts directly with PKCδ. c-Abl phosphorylates and activates PKCδ in vitro. We also show that IR treatment of cells is associated with c-Abl-dependent phosphorylation of PKCδ and translocation of PKCδ to the nucleus. These findings support a functional interaction between c-Abl and PKCδ in the cellular response to genotoxic stress.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yuan, ZM., Utsugisawa, T., Ishiko, T. et al. Activation of protein kinase C δ by the c-Abl tyrosine kinase in response to ionizing radiation. Oncogene 16, 1643–1648 (1998). https://doi.org/10.1038/sj.onc.1201698
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1201698
Keywords
This article is cited by
-
Identification and characterisation of lamprey protein kinase C delta-like gene
Scientific Reports (2017)
-
A balancing act: orchestrating amino-truncated and full-length p73 variants as decisive factors in cancer progression
Oncogene (2015)
-
Bryostatin-1 causes radiosensitization of BMG-1 malignant glioma cells through differential activation of protein kinase-Cδ not evident in the non-malignant AA8 fibroblasts
Molecular and Cellular Biochemistry (2015)
-
MiR-181a confers resistance of cervical cancer to radiation therapy through targeting the pro-apoptotic PRKCD gene
Oncogene (2013)
-
Regulation of p73 activity by post-translational modifications
Cell Death & Disease (2012)