Featured
-
-
Article
| Open Access Genetic and functional diversity of β-N-acetylgalactosamine-targeting glycosidases expanded by deep-sea metagenome analysis
Genetic and functional diversity of β-N-acetylgalactosamine-targeting glycosidases expanded by deep-sea metagenome analysis
Four β-N-acetylgalactosaminidase gene families were identified via deep-sea metagenome analysis. The biochemical and structural characterization of the aforementioned enzymes revealed their functional diversity and monophyletic evolutionary history.
- Tomomi Sumida
- , Satoshi Hiraoka
- & Takuro Nunoura
-
Article
| Open Access Remote loop evolution reveals a complex biological function for chitinase enzymes beyond the active site
Remote loop evolution reveals a complex biological function for chitinase enzymes beyond the active site
Loop regions play a key role in protein evolution. Herein the authors demonstrate how GH19 chitinase acquired additional antifungal activity by introducing remote loops, without compromising its original function. This work offers an innovative approach to expand enzyme function.
- Dan Kozome
- , Adnan Sljoka
- & Paola Laurino
-
Article
| Open Access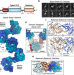 Architecture and regulation of filamentous human cystathionine beta-synthase
Architecture and regulation of filamentous human cystathionine beta-synthase
Cystathionine beta-synthase is a conserved essential enzyme of one-carbon metabolism. Here, the authors show that the enzyme oligomerises to form filaments that undergo conformational and morphological changes in response to its activator S-adenosyl-L-methionine, the global methyl donor.
- Thomas J. McCorvie
- , Douglas Adamoski
- & Wyatt W. Yue
-
Article
| Open Access Structural basis for phage-mediated activation and repression of bacterial DSR2 anti-phage defense system
Structural basis for phage-mediated activation and repression of bacterial DSR2 anti-phage defense system
The bacterial DSR2 defense system counters phage invasion by depleting NAD+. Here, Zhang et al. reveal molecular mechanisms underlying phage-mediated activation and repression of DSR2, enhancing our understanding of the bacterial-phage arms race.
- Jun-Tao Zhang
- , Xiao-Yu Liu
- & Ning Jia
-
Article
| Open Access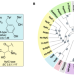 Natural diversity screening, assay development, and characterization of nylon-6 enzymatic depolymerization
Natural diversity screening, assay development, and characterization of nylon-6 enzymatic depolymerization
Polyamides (PAs) or nylons are types of plastics with wide applications, but due to their accumulation in the environment, strategies for their deconstruction are of interest. Here, the authors screen 40 potential nylon-hydrolyzing enzymes (nylonases) using a mass spectrometry-based approach and identify a thermostabilized N-terminal nucleophile hydrolase as the most promising for further development, as well as crucial targets for progressing PA6 enzymatic depolymerization.
- Elizabeth L. Bell
- , Gloria Rosetto
- & Gregg T. Beckham
-
Article
| Open Access Molecular basis for the catalytic mechanism of human neutral sphingomyelinases 1 (hSMPD2)
Molecular basis for the catalytic mechanism of human neutral sphingomyelinases 1 (hSMPD2)
Neutral sphingomyelinases play pivotal roles in ceramide-related signaling transduction. Here, the authors solve the structure of human neutral sphingomyelinase SMPD2 and propose a catalytic mechanism, potentially enhancing understanding of ceramide in disease and cancer treatment.
- Jingbo Yi
- , Boya Qi
- & Maojun Yang
-
Article
| Open Access Catalytically inactive long prokaryotic Argonaute systems employ distinct effectors to confer immunity via abortive infection
Catalytically inactive long prokaryotic Argonaute systems employ distinct effectors to confer immunity via abortive infection
Here, Song et al. show that catalytically inactive long prokaryotic Argonaute proteins are equipped with distinct effectors that are activated upon recognition of invading genetic elements to trigger cell death and confer abortive infection immunity.
- Xinmi Song
- , Sheng Lei
- & Wenyuan Han
-
Article
| Open Access Structural basis for the unique molecular properties of broad-range phospholipase C from Listeria monocytogenes
Structural basis for the unique molecular properties of broad-range phospholipase C from Listeria monocytogenes
The bacterium Listeria monocytogenes possesses two major virulence factors, broad-range phospholipase C (LmPC-PLC) and the pore-forming toxin listeriolysin O (LLO). Here, authors perform structural and biochemical analysis of LmPC-PLC and show that unique structural features enable self-regulation of its enzymatic activity and positive synergy with the pore-forming toxin listeriolysin O.
- Nejc Petrišič
- , Maksimiljan Adamek
- & Marjetka Podobnik
-
Article
| Open Access Discovery and rational engineering of PET hydrolase with both mesophilic and thermophilic PET hydrolase properties
Discovery and rational engineering of PET hydrolase with both mesophilic and thermophilic PET hydrolase properties
Extensive research efforts have been directed towards the development of PET hydrolases with improved activity, but template enzymes used are limited. Here, the authors report a PET hydrolase from Cryptosporangium aurantiacum (CaPETase) that exhibits high thermostability and PET degradation activity at ambient temperatures and determine its crystal structure.
- Hwaseok Hong
- , Dongwoo Ki
- & Kyung-Jin Kim
-
Article
| Open Access The structural pathology for hypophosphatasia caused by malfunctional tissue non-specific alkaline phosphatase
The structural pathology for hypophosphatasia caused by malfunctional tissue non-specific alkaline phosphatase
Hypophosphatasia (HPP) is a bone disease caused by mutations in tissue non-specific alkaline phosphatase (TNAP). Here, authors solved the crystal and cryoEM structures of TNAP, shedding light on the molecular mechanisms underlying HPP.
- Yating Yu
- , Kewei Rong
- & An Qin
-
Article
| Open Access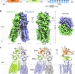 Structural insight into the human SID1 transmembrane family member 2 reveals its lipid hydrolytic activity
Structural insight into the human SID1 transmembrane family member 2 reveals its lipid hydrolytic activity
The SID-1 family is a putative channel/transporter for transporting nucleic acids. Here, Qian et al. report the dimeric structure of human SIDT2 suggesting that it may act as a transporter but not a channel. In addition, it has a ceramidase activity.
- Dandan Qian
- , Ye Cong
- & Deshun Gong
-
Article
| Open Access A multicentric consortium study demonstrates that dimethylarginine dimethylaminohydrolase 2 is not a dimethylarginine dimethylaminohydrolase
A multicentric consortium study demonstrates that dimethylarginine dimethylaminohydrolase 2 is not a dimethylarginine dimethylaminohydrolase
While dimethylarginine dimethylaminohydrolase 1 (DDAH1) is known to metabolize the endogenous inhibitor of nitric oxide synthases, asymmetric dimethylarginine (ADMA), the function of DDAH2 has remained controversial. Here, the authors present several lines of evidence that DDAH2 does not hydrolyze ADMA.
- Vinitha N. Ragavan
- , Pramod C. Nair
- & Roman N. Rodionov
-
Article
| Open Access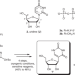 A selective and atom-economic rearrangement of uridine by cascade biocatalysis for production of pseudouridine
A selective and atom-economic rearrangement of uridine by cascade biocatalysis for production of pseudouridine
Pseudouridine (Ψ), the C-nucleoside isomer of uridine, and its 1-N-methyl derivative, are incorporated in mRNA vaccines and essential for their efficiency, but difficult to synthetically access. Here, the authors report on selective and atom-economic 1N-5C rearrangement of β-d-ribosyl on uracil to obtain Ψ from unprotected U in quantitative yield
- Martin Pfeiffer
- , Andrej Ribar
- & Bernd Nidetzky
-
Article
| Open Access Structure and mechanism of the alkane-oxidizing enzyme AlkB
Structure and mechanism of the alkane-oxidizing enzyme AlkB
Alkane monooxygenase (AlkB) plays a key role in the global carbon cycle and remediation of oil spills. Here, the authors report the cryo-EM structure of AlkB to provide insight into the catalytic mechanism and substrate selectivity.
- Xue Guo
- , Jianxiu Zhang
- & Liang Feng
-
Article
| Open Access Inhibitor-3 inhibits Protein Phosphatase 1 via a metal binding dynamic protein–protein interaction
Inhibitor-3 inhibits Protein Phosphatase 1 via a metal binding dynamic protein–protein interaction
Protein phosphatase 1 (PP1) is regulated by intrinsically disordered proteins like inhibitor-3, I3. The authors show that I3 does not inhibit PP1 by forming a rigid complex but instead by binding dynamically with its active site metals, showing how flexibility is used in biology.
- Gautam Srivastava
- , Meng S. Choy
- & Wolfgang Peti
-
Article
| Open Access Mechanism of antibody-specific deglycosylation and immune evasion by Streptococcal IgG-specific endoglycosidases
Mechanism of antibody-specific deglycosylation and immune evasion by Streptococcal IgG-specific endoglycosidases
Bacterial pathogens have evolved intricate mechanisms to evade the human immune system, including the production of immunomodulatory enzymes. Here, the authors establish the mechanisms of recognition and specific deglycosylation of IgG antibodies by the multi-modular enzymes EndoS and EndoS2
- Beatriz Trastoy
- , Jonathan J. Du
- & Marcelo E. Guerin
-
Article
| Open Access Complete bio-degradation of poly(butylene adipate-co-terephthalate) via engineered cutinases
Complete bio-degradation of poly(butylene adipate-co-terephthalate) via engineered cutinases
Bio-degradation of poly(butylene adipate-co-terephthalate) is an attractive tactic but requires an effective hydrolytic enzyme. Here, the authors demonstrate that cutinases are highly potent PBAT-decomposing enzymes and their mechanism is proposed based on substrate-binding mode.
- Yu Yang
- , Jian Min
- & Rey-Ting Guo
-
Article
| Open Access Sourcing thermotolerant poly(ethylene terephthalate) hydrolase scaffolds from natural diversity
Sourcing thermotolerant poly(ethylene terephthalate) hydrolase scaffolds from natural diversity
Enzymes have potential for recycling plastics such as PET, a polyester used in textiles and single-use packaging. Here, the authors identify and characterize additional PET-active biocatalysts and expand the number and diversity of thermotolerant scaffolds for enzymatic PET deconstruction.
- Erika Erickson
- , Japheth E. Gado
- & John E. McGeehan
-
Article
| Open Access Rational enzyme design for enabling biocatalytic Baldwin cyclization and asymmetric synthesis of chiral heterocycles
Rational enzyme design for enabling biocatalytic Baldwin cyclization and asymmetric synthesis of chiral heterocycles
Chiral heterocyclic compounds are privileged structures in medicinal chemistry. Here, the authors report an in silico strategy for the enzymatic synthesis of pharmaceutically significant chiral N- and O-heterocycles via Baldwin cyclization of hydroxy- and amino-substituted epoxides and oxetanes using epoxide hydrolase mutants.
- Jun-Kuan Li
- , Ge Qu
- & Zhoutong Sun
-
Article
| Open Access Biodegradation of highly crystallized poly(ethylene terephthalate) through cell surface codisplay of bacterial PETase and hydrophobin
Biodegradation of highly crystallized poly(ethylene terephthalate) through cell surface codisplay of bacterial PETase and hydrophobin
High-crystallinity poly(ethylene terephthalate) is a major recycling challenge. Here, the authors show an engineered whole-cell biocatalyst showing adhesive hydrophobin and PETase on the surface of cells, for biodegradation of PET.
- Zhuozhi Chen
- , Rongdi Duan
- & Zefang Wang
-
Article
| Open Access Design and directed evolution of noncanonical β-stereoselective metalloglycosidases
Design and directed evolution of noncanonical β-stereoselective metalloglycosidases
Metallohydrolases are ubiquitous enzymes. Here the authors show the design and biochemical characterisation of metalloglycosidase by constructing a hydrolytically active Zn-binding site within a barrel-shaped outer membrane protein OmpF.
- Woo Jae Jeong
- & Woon Ju Song
-
Article
| Open Access Cooperative assembly of p97 complexes involved in replication termination
Cooperative assembly of p97 complexes involved in replication termination
This study describes how p97Ufd1-Npl4 and the UBA-UBX protein Ubxn7 disassemble vertebrate replisomes during replication termination, and it provides novel insights into how p97 complexes assemble with UBA-UBX proteins on ubiquitylated substrates
- Olga V. Kochenova
- , Sirisha Mukkavalli
- & Johannes C. Walter
-
Article
| Open Access Structural characterization of a soil viral auxiliary metabolic gene product – a functional chitosanase
Structural characterization of a soil viral auxiliary metabolic gene product – a functional chitosanase
Metagenomics is revealing auxiliary metabolic genes (AMGs) in soil viral genomes. Here, authors solve the crystal structure for a soil viral AMG product, free and ligand bound, and show the protein can decompose chitin, a common carbon polymer.
- Ruonan Wu
- , Clyde A. Smith
- & Janet K. Jansson
-
Article
| Open Access Structural insights into the pSer/pThr dependent regulation of the SHP2 tyrosine phosphatase in insulin and CD28 signaling
Structural insights into the pSer/pThr dependent regulation of the SHP2 tyrosine phosphatase in insulin and CD28 signaling
SHP2 is an important human tyrosine phosphatase with key roles in cancer, immune responses and insulin signaling. Here, the authors explore its substrate recognition mechanism in molecular detail and uncover a complex regulatory mechanism for this enzyme that marks specific target sites for dephosphorylation.
- András Zeke
- , Tamás Takács
- & Attila Reményi
-
Article
| Open Access Flavin-enabled reductive and oxidative epoxide ring opening reactions
Flavin-enabled reductive and oxidative epoxide ring opening reactions
Epoxide ring opening reactions are important in both biological processes and synthetic applications. Here, the authors show that flavin cofactors can catalyze reductive and oxidative epoxide ring opening reactions and propose the underlying mechanisms.
- Bidhan Chandra De
- , Wenjun Zhang
- & Changsheng Zhang
-
Article
| Open Access Golgi stress induces SIRT2 to counteract Shigella infection via defatty-acylation
Golgi stress induces SIRT2 to counteract Shigella infection via defatty-acylation
Here the authors revealed a role for the protein deacetylase SIRT2 in Golgi stress, particularly induced by bacterial infection. Shigella secrete effector proteins such as IcsB, which transfers fatty acyl groups to modify host proteins to evade host immune surveillance. The upregulated SIRT2 counteracts this function by removing the fatty acyl groups and enhancing the killing of Shigella.
- Miao Wang
- , Yugang Zhang
- & Hening Lin
-
Article
| Open Access A cryptic third active site in cyanophycin synthetase creates primers for polymerization
A cryptic third active site in cyanophycin synthetase creates primers for polymerization
Cyanophycin synthetase CphA1 polymerizes Asp and Arg into the nitrogen reserve polymer cyanophycin using two active sites. Sharon et al. show CphA1 has a cryptic 3rd active site that cleaves cyanophycin into primers for self-sufficient biosynthesis.
- Itai Sharon
- , Sharon Pinus
- & T. Martin Schmeing
-
Article
| Open Access Engineering substrate specificity of HAD phosphatases and multienzyme systems development for the thermodynamic-driven manufacturing sugars
Engineering substrate specificity of HAD phosphatases and multienzyme systems development for the thermodynamic-driven manufacturing sugars
Haloacid dehalogenase-like phosphatases are widespread across all domains of life and play a crucial role in the regulation of levels of sugar phosphate metabolites in cells. The authors report on the structure-guided engineering of phosphatases for dedicated substrate specificity for the conversion of sucrose and starch into fructose and mannose.
- Chaoyu Tian
- , Jiangang Yang
- & Yanhe Ma
-
Article
| Open Access A structural exposé of noncanonical molecular reactivity within the protein tyrosine phosphatase WPD loop
A structural exposé of noncanonical molecular reactivity within the protein tyrosine phosphatase WPD loop
Inositol pyrophosphates (PP-IPs) regulate immunity and phosphate homeostasis. Here, the authors’ structural dissection of an Arabidopsis thaliana PP-IP phosphatase reveals that substrates drive their own hydrolysis and identifies a highly elusive metaphosphate-like reaction intermediate.
- Huanchen Wang
- , Lalith Perera
- & Stephen B. Shears
-
Article
| Open Access Mechanism and biomass association of glucuronoyl esterase: an α/β hydrolase with potential in biomass conversion
Mechanism and biomass association of glucuronoyl esterase: an α/β hydrolase with potential in biomass conversion
Zong and coworkers combine computational and experimental methods to decipher in detail the mechanism of action of glucuronoyl esterases, enzymes with significant biotechnological potential for decoupling lignin from polysaccharides in biomass.
- Zhiyou Zong
- , Scott Mazurkewich
- & Leila Lo Leggio
-
Article
| Open Access The catalytic activity of TCPTP is auto-regulated by its intrinsically disordered tail and activated by Integrin alpha-1
The catalytic activity of TCPTP is auto-regulated by its intrinsically disordered tail and activated by Integrin alpha-1
TCPTP is a non-receptor type protein tyrosine phosphatase involved in various signalling pathways. Here, the authors provide structural insights into TCPTP activation, showing that TCPTP is inhibited by its C-terminal tail, which can be displaced by the cytosolic tail of integrin-α1, leading to activation.
- Jai Prakash Singh
- , Yang Li
- & Tzu-Ching Meng
-
Article
| Open Access Crystal structures of the elusive Rhizobium etli l-asparaginase reveal a peculiar active site
Crystal structures of the elusive Rhizobium etli l-asparaginase reveal a peculiar active site
L-asparaginases catalyse the hydrolysis of L-asparagine to L-aspartic acid and ammonia. Here, the authors present high resolution crystal structures of Rhizobium etli L-asparaginase that contains a Zn2+ binding site without a catalytic role and discuss the catalytic mechanism of the enzyme.
- Joanna I. Loch
- , Barbara Imiolczyk
- & Mariusz Jaskolski
-
Article
| Open Access Palmitoylation targets the calcineurin phosphatase to the phosphatidylinositol 4-kinase complex at the plasma membrane
Palmitoylation targets the calcineurin phosphatase to the phosphatidylinositol 4-kinase complex at the plasma membrane
Calcineurin — the Ca2+ regulated phosphatase and target of immunosuppressants — regulates GPCR-mediated phospholipid signaling at the plasma membrane. Here the authors show that CNAβ1 (a poorly studied isoform of the calcineurin catalytic subunit) is targeted to the plasma membrane through palmitoylation to dephosphorylate and promote PI4KA complex activity.
- Idil Ulengin-Talkish
- , Matthew A. H. Parson
- & Martha S. Cyert
-
Article
| Open Access Mechanism of genome instability mediated by human DNA polymerase mu misincorporation
Mechanism of genome instability mediated by human DNA polymerase mu misincorporation
Pol μ performs gap-filling repair synthesis in the nonhomologous end joining (NHEJ) pathway. Here the authors provide crystal structures and kinetics of human Pol μ to reveal insights into the molecular mechanism of Pol μ during the process of dGTP misincorporation.
- Miao Guo
- , Yina Wang
- & Ye Zhao
-
Article
| Open Access Engineering the protein dynamics of an ancestral luciferase
Engineering the protein dynamics of an ancestral luciferase
Directed evolution commonly relies on point mutations but InDels frequently occur in evolution. Here the authors report a protein-engineering framework based on InDel mutagenesis and fragment transplantation resulting in greater catalysis and longer glow-type bioluminescence of the ancestral luciferase.
- Andrea Schenkmayerova
- , Gaspar P. Pinto
- & Jiri Damborsky
-
Article
| Open Access Structural and functional insights into esterase-mediated macrolide resistance
Structural and functional insights into esterase-mediated macrolide resistance
Erythromycin esterases (Eres) cleave the macrolactone ring of macrolides, a class of widely used antibiotics. Structures of EreC, in silico flexible docking studies and previous mutagenesis data lead to the proposal of a detailed catalytic mechanism for the Ere family of enzymes.
- Michał Zieliński
- , Jaeok Park
- & Albert M. Berghuis
-
Article
| Open Access Discovery of fungal surface NADases predominantly present in pathogenic species
Discovery of fungal surface NADases predominantly present in pathogenic species
Some bacterial pathogens release NADase enzymes into the host cell that deplete the host’s NAD+ pool, thereby causing rapid cell death. Here, Strømland et al. identify NADases on the surface of fungal spores, and show that the enzymes display unique biochemical and structural properties.
- Øyvind Strømland
- , Juha P. Kallio
- & Mathias Ziegler
-
Article
| Open Access Cryo-EM structure of Helicobacter pylori urease with an inhibitor in the active site at 2.0 Å resolution
Cryo-EM structure of Helicobacter pylori urease with an inhibitor in the active site at 2.0 Å resolution
Infection by Helicobacter pylori is associated with peptic ulcers and gastric cancer. H. pylori urease is required for colonization of the stomach and thus an attractive antimicrobial drug target. Cryo-EM analyses of the H. pylori urease with inhibitors bound reveal structural details useful in rational drug design.
- Eva S. Cunha
- , Xiaorui Chen
- & Hartmut Luecke
-
Article
| Open Access Site-specific incorporation of citrulline into proteins in mammalian cells
Site-specific incorporation of citrulline into proteins in mammalian cells
Citrullination of arginine is crucial for several physiological processes. Here the authors report the site-specific incorporation of citrulline into proteins in mammalian cells using an engineered tRNA synthetase/tRNA pair and a photocaged-citrulline.
- Santanu Mondal
- , Shu Wang
- & Paul R. Thompson
-
Article
| Open Access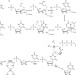 Reverse C-glycosidase reaction provides C-nucleotide building blocks of xenobiotic nucleic acids
Reverse C-glycosidase reaction provides C-nucleotide building blocks of xenobiotic nucleic acids
C-nucleosides are analogues of the canonical N-nucleosides and, despite their synthetic value, biocatalysis has not targeted them yet. Here, the authors report a pseudouridine monophosphate C-glycosidase enzyme for selective 5-β-C-glycosylation of uracil and its derivatives from pentose 5- phosphate substrates.
- Martin Pfeiffer
- & Bernd Nidetzky
-
Article
| Open Access Structure and dynamics of an α-fucosidase reveal a mechanism for highly efficient IgG transfucosylation
Structure and dynamics of an α-fucosidase reveal a mechanism for highly efficient IgG transfucosylation
AlfC transfucosidase is used to modulate fucosylation of glycans decorating monoclonal antibodies. Herein, structural and biophysical characterization reveals the enzymatic mechanism of AlfC and a blueprint for the design of AlfC mutants with novel specificities and functions.
- Erik H. Klontz
- , Chao Li
- & Eric J. Sundberg
-
Article
| Open Access Structural asymmetry governs the assembly and GTPase activity of McrBC restriction complexes
Structural asymmetry governs the assembly and GTPase activity of McrBC restriction complexes
The bacterial defense system McrBC is a two-component motor-driven nuclease complex that cleaves foreign DNA. Here, the authors present the structures of the GTP-specific AAA + motor protein McrB and two McrBC complexes and discuss the molecular mechanism of how McrC binding stimulates McrB GTP hydrolysis.
- Yiming Niu
- , Hiroshi Suzuki
- & Joshua S. Chappie
-
Article
| Open Access A catalogue of biochemically diverse CRISPR-Cas9 orthologs
A catalogue of biochemically diverse CRISPR-Cas9 orthologs
A few bacterial Cas9 nucleases have been repurposed as genome editing tools. Here, the authors use bioinformatic and biochemical analyses to characterize 79 Cas9 proteins, revealing substantial functional diversity and thus expanding the available toolbox of RNA-programmable CRISPR-associated nucleases.
- Giedrius Gasiunas
- , Joshua K. Young
- & Virginijus Siksnys
-
Article
| Open Access Thioglycoligase derived from fungal GH3 β-xylosidase is a multi-glycoligase with broad acceptor tolerance
Thioglycoligase derived from fungal GH3 β-xylosidase is a multi-glycoligase with broad acceptor tolerance
Thioglycoligases have proved useful for bonding carbohydrates to non-sugar acceptors, however, the scope of these biocatalysts is usually limited. Here, the authors engineer a xylosidase into a thioglycoligase with the ability to form O-, N-, S- and Se- glycosides together with sugar esters and phosphoesters.
- Manuel Nieto-Domínguez
- , Beatriz Fernández de Toro
- & María Jesús Martínez
-
Article
| Open Access ABHD4-dependent developmental anoikis safeguards the embryonic brain
ABHD4-dependent developmental anoikis safeguards the embryonic brain
During embryonic development, neural progenitor cells undergo numerous cell divisions. Here, the authors show that ABHD4-mediated developmental anoikis distinguishes the physiological delamination and the pathological detachment of progenitor cells with relevance to fetal alcohol-induced apoptosis.
- Zsófia I. László
- , Zsolt Lele
- & István Katona
-
Article
| Open Access ABHD11 maintains 2-oxoglutarate metabolism by preserving functional lipoylation of the 2-oxoglutarate dehydrogenase complex
ABHD11 maintains 2-oxoglutarate metabolism by preserving functional lipoylation of the 2-oxoglutarate dehydrogenase complex
2-oxoglutarate links mitochondrial metabolism to oxygen sensing, gene transcription and epigenetic modifications. Here, the authors describe a mutagenesis screen to find genes that control 2-oxoglutarate levels, and identify ABHD11 as a mitochondrial regulator of 2-oxoglutarate abundance.
- Peter S. J. Bailey
- , Brian M. Ortmann
- & James A. Nathan
-
Article
| Open Access Dissecting the sequence determinants for dephosphorylation by the catalytic subunits of phosphatases PP1 and PP2A
Dissecting the sequence determinants for dephosphorylation by the catalytic subunits of phosphatases PP1 and PP2A
The substrate specificity of phosphoprotein phosphatases PP1 and PP2A depends on their catalytic and regulatory subunits. Using proteomics approaches, the authors here provide insights into the sequence specificity of the catalytic subunits and their distinct contributions to PP1 and PP2A selectivity.
- Bernhard Hoermann
- , Thomas Kokot
- & Maja Köhn
-
Article
| Open Access The receptor PTPRU is a redox sensitive pseudophosphatase
The receptor PTPRU is a redox sensitive pseudophosphatase
Receptor-linked protein tyrosine phosphatases (RPTPs) usually contain an active membrane proximal domain and an inactive pseudophosphatase domain. Here, the authors characterize an RPTP with two pseudophosphatase domains, providing evidence that it may act as a decoy receptor for substrate sequestration.
- Iain M. Hay
- , Gareth W. Fearnley
- & Janet E. Deane
-
Article
| Open Access Structural plasticity of SARS-CoV-2 3CL Mpro active site cavity revealed by room temperature X-ray crystallography
Structural plasticity of SARS-CoV-2 3CL Mpro active site cavity revealed by room temperature X-ray crystallography
The SARS-CoV-2 3CL main protease (3CL Mpro) is a chymotrypsin-like protease that facilitates the production of non-structural proteins, which are essential for viral replication and is therefore of great interest as a drug target. Here, the authors present the 2.30 Å room temperature crystal structure of ligand-free 3CL Mpro and compare it with the earlier determined low-temperature ligand-free and inhibitor-bound crystal structures.
- Daniel W. Kneller
- , Gwyndalyn Phillips
- & Andrey Kovalevsky

 Contractile injection systems facilitate sporogenic differentiation of Streptomyces davawensis through the action of a phage tapemeasure protein-related effector
Contractile injection systems facilitate sporogenic differentiation of Streptomyces davawensis through the action of a phage tapemeasure protein-related effector