Featured
-
-
Article |
 Tunable molecular editing of indoles with fluoroalkyl carbenes
Tunable molecular editing of indoles with fluoroalkyl carbenes
The rapid generation of molecular complexity from a given molecular scaffold is crucial to drug discovery and development. Now the chemodivergent molecular editing of indoles using fluoroalkyl carbenes has been developed to modularly access four different types of fluorine-containing N-heterocyclic compound with high molecular complexity.
- Shaopeng Liu
- , Yong Yang
- & Xihe Bi
-
Article
| Open Access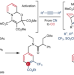 Skeletal editing of pyridines through atom-pair swap from CN to CC
Skeletal editing of pyridines through atom-pair swap from CN to CC
Skeletal editing enables diversification of compounds not possible by applying peripheral editing strategies. Now, a catalyst-free atom-pair swap strategy for pyridine editing has been developed via one-pot sequential dearomatization, cycloaddition and rearomative retrocyclization. Benzenes and naphthalenes with precisely installed functional groups are produced, and the mild conditions enable late-stage skeletal diversification of pyridine cores.
- Qiang Cheng
- , Debkanta Bhattacharya
- & Armido Studer
-
News & Views |
 Simple start for complex products
Simple start for complex products
Natural products often provide lead scaffolds for the development of therapeutics, but complexity of their synthesis can limit the discovery of improved analogues. Pharmacophore-directed retrosynthesis aims to accelerate the building of a structure–activity relationship profile of a natural product, aiming to identifying a simplified lead.
- Jason R. Hudlicky
- & Gary A. Sulikowski
-
Article |
 Simplified immunosuppressive and neuroprotective agents based on gracilin A
Simplified immunosuppressive and neuroprotective agents based on gracilin A
Pharmacophore-directed retrosynthesis targets a potential pharmacophore from early on in a natural product synthesis and incremental increases in the complexity of this minimal structure enable a SAR profile to develop over the course of the campaign. The method is applied to gracilin A, finding simplified derivatives displaying potent immunosuppressive effects or selective neuroprotective effects in cell-based assays.
- Mikail E. Abbasov
- , Rebeca Alvariño
- & Daniel Romo
-
News & Views |
 Taming reactive benzynes
Taming reactive benzynes
Natural products often serve as sources of new drugs, either directly or after synthetic modification, but site-selective functionalization of complex small molecules is challenging. Now, a method has been developed that enables selective modification of a wide range of natural products by engaging a benzyne intermediate in a variety of reaction modes.
- Sarah Z. Tasker
- & Paul J. Hergenrother
-
Article |
 Unified biomimetic assembly of voacalgine A and bipleiophylline via divergent oxidative couplings
Unified biomimetic assembly of voacalgine A and bipleiophylline via divergent oxidative couplings
The biomimetic syntheses of bipleiophylline, one of the most complex monoterpene indole alkaloids, and voacalgine A, its biosynthetic precursor, have been achieved from pleiocarpamine starting material. The development of a divergent oxidative coupling for the formation of the benzofuro[2,3-b]indolenine and isochromano[3,4-b]indolenine moieties was key to this accomplishment.
- David Lachkar
- , Natacha Denizot
- & Guillaume Vincent
-
Article |
 Synthesis and characterization of hexaarylbenzenes with five or six different substituents enabled by programmed synthesis
Synthesis and characterization of hexaarylbenzenes with five or six different substituents enabled by programmed synthesis
A lack of general methods for making multisubstituted benzene derivatives means that only a small fraction of the huge number of possible structures based on this ubiquitous building block have been explored. Now, a programmed synthesis of hexaarylbenzenes using C–H activation, cross-coupling and [4 + 2] cycloaddition reactions has been developed that can also be applied to tetraarylnaphthalenes and pentaarylpyridines.
- Shin Suzuki
- , Yasutomo Segawa
- & Junichiro Yamaguchi
-
Interview |
 Parallels with nature
Parallels with nature
Adam Nelson and Stuart Warriner, from the University of Leeds, talk with Nature Chemistry about their work to develop viable synthetic strategies for preparing new chemical structures in parallel with the identification of desirable biological activity.
- Stephen Davey
-
Article |
 Efficient discovery of bioactive scaffolds by activity-directed synthesis
Efficient discovery of bioactive scaffolds by activity-directed synthesis
A discovery approach termed activity-directed synthesis is described; it exploits arrays of reactions whose outcome is critically dependent on the conditions used, and prioritizes reactions that yield bioactive product mixtures. The discovery of both bioactive small molecules and associated synthetic routes thus occurs in parallel.
- George Karageorgis
- , Stuart Warriner
- & Adam Nelson
-
Article |
 Biogenetically inspired synthesis and skeletal diversification of indole alkaloids
Biogenetically inspired synthesis and skeletal diversification of indole alkaloids
Emulating the biogenesis of natural products, a synthetic strategy is described in which an achiral multipotent intermediate reacts through three distinct [4 + 2] cyclizations and two types of redox-mediated annulation. This results in divergent access to natural product-like scaffolds in 6–9 steps. The efficiency of this approach is highlighted in the total syntheses of three natural products.
- Haruki Mizoguchi
- , Hideaki Oikawa
- & Hiroki Oguri
-
Article |
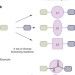 A strategy for the diversity-oriented synthesis of macrocyclic scaffolds using multidimensional coupling
A strategy for the diversity-oriented synthesis of macrocyclic scaffolds using multidimensional coupling
Diversity oriented synthesis (DOS) aims to build structurally diverse compound libraries — potentially useful in drug discovery — from a small number of starting materials. Here, the build/couple/pair algorithm — commonly used in DOS — is extended to incorporate variations in the coupling step as well as in the starting materials. This produces a compound library with exceptionally high diversity in fewer than five steps from a common precursor.
- Henning S. G. Beckmann
- , Feilin Nie
- & David R. Spring
-
Article |
 Remodelling of the natural product fumagillol employing a reaction discovery approach
Remodelling of the natural product fumagillol employing a reaction discovery approach
The natural product fumagillol has been exploited as a stereochemically rich scaffold for the synthesis of a structurally unique, chemically diverse library with chemotypes distinctly different from the parent structure. Thus, fumagillol has been remodelled into a diverse array of isoindoles, isoquinolines, furans, mopholinones and benzoxazepines.
- Bradley R. Balthaser
- , Meghan C. Maloney
- & John K. Snyder

 Persistent organonickel complexes as general platforms for Csp2–Csp3 coupling reactions
Persistent organonickel complexes as general platforms for Csp2–Csp3 coupling reactions