Featured
-
-
Article
| Open Access Bispecific CAR T cell therapy targeting BCMA and CD19 in relapsed/refractory multiple myeloma: a phase I/II trial
Bispecific CAR T cell therapy targeting BCMA and CD19 in relapsed/refractory multiple myeloma: a phase I/II trial
CAR-T cell therapies targeting BCMA have shown promising responses in patients with multiple myeloma (MM), however primary resistance and relapse are frequently observed. Here the authors report the results of a phase I//II study of bispecific CAR T-cells targeting BCMA and CD19 in relapsed/refractory MM.
- Ming Shi
- , Jiaojiao Wang
- & Jiang Cao
-
Article
| Open Access Clinical and biomarker results from a phase II trial of combined cabozantinib and durvalumab in patients with chemotherapy-refractory colorectal cancer (CRC): CAMILLA CRC cohort
Clinical and biomarker results from a phase II trial of combined cabozantinib and durvalumab in patients with chemotherapy-refractory colorectal cancer (CRC): CAMILLA CRC cohort
CAMILLA is a basket trial evaluating cabozantinib plus the ICI durvalumab in chemorefractory gastrointestinal cancer. Here, the authors present the result of the phase II in the colorectal cohort.
- Anwaar Saeed
- , Robin Park
- & Azhar Saeed
-
Article
| Open Access A phase 1/2 clinical trial of invariant natural killer T cell therapy in moderate-severe acute respiratory distress syndrome
A phase 1/2 clinical trial of invariant natural killer T cell therapy in moderate-severe acute respiratory distress syndrome
Invariant natural killer T (iNKT) cells recognize abnormal cells, but their T cell receptor is not variable and kill cancerous or infected target cells without MHC I restriction. Here, the authors show that in a clinical trial, donor-unrestricted allogeneic iNKT cells could be safely administered to human COVID-19 patients suffering from acute respiratory distress syndrome and trigger an anti-inflammatory response.
- Terese C. Hammond
- , Marco A. Purbhoo
- & Mark A. Exley
-
Article
| Open Access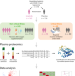 Longitudinal plasma proteomics reveals biomarkers of alveolar-capillary barrier disruption in critically ill COVID-19 patients
Longitudinal plasma proteomics reveals biomarkers of alveolar-capillary barrier disruption in critically ill COVID-19 patients
Using longitudinal profiling of 6385 plasma proteins in hospitalised patients, the authors demonstrate that alveolar capillary barrier disruption in critical COVID-19 is reflected in the plasma proteome, and is attenuated with imatinib treatment.
- Erik Duijvelaar
- , Jack Gisby
- & Jurjan Aman
-
Article
| Open Access Redirecting antibody responses from egg-adapted epitopes following repeat vaccination with recombinant or cell culture-based versus egg-based influenza vaccines
Redirecting antibody responses from egg-adapted epitopes following repeat vaccination with recombinant or cell culture-based versus egg-based influenza vaccines
Here the authors report an exploratory analysis of a clinical trial that tested different influenza virus vaccination platforms. The results show that multiple seasons of recombinant or cell-based influenza vaccinations may be needed to redirect antibody responses away from immune memory to egg-adapted epitopes and refocus on epitopes on the circulating viruses.
- Feng Liu
- , F. Liaini Gross
- & Min Z. Levine
-
Article
| Open Access A pilot study of alternative substrates in the critically Ill subject using a ketogenic feed
A pilot study of alternative substrates in the critically Ill subject using a ketogenic feed
Critical illness leads to altered metabolic states and bioenergetic failure caused by impaired utilisation of glucose, fatty acids and amino acids. Here the authors show ketogenic diets may provide a safe and acceptable alternative metabolic fuel enabling energy production and maintaining tissue homeostasis.
- Angela McNelly
- , Anne Langan
- & Zudin A. Puthucheary
-
Article
| Open Access Tamoxifen for the treatment of myeloproliferative neoplasms: A Phase II clinical trial and exploratory analysis
Tamoxifen for the treatment of myeloproliferative neoplasms: A Phase II clinical trial and exploratory analysis
Preclinical studies indicate that myeloproliferative neoplasms (MPN) may be sensitive to the estrogen receptor modulator, tamoxifen. Here, the authors present a phase II clinical trial reporting the efficacy of tamoxifen in MPN and analysis of peripheral haematopoietic stem cells to identify potential predictive signatures of responders.
- Zijian Fang
- , Giuditta Corbizi Fattori
- & Simón Méndez-Ferrer
-
Article
| Open Access A standardized metric to enhance clinical trial design and outcome interpretation in type 1 diabetes
A standardized metric to enhance clinical trial design and outcome interpretation in type 1 diabetes
The use of a standardized outcome metric enhances clinical trial interpretation and cross-trial comparison. Here, the authors show the implementation of such a metric using type 1 diabetes trial data, reassess and compare results from these trials, and extend its use to define response to therapy.
- Alyssa Ylescupidez
- , Henry T. Bahnson
- & Carla J. Greenbaum
-
Article
| Open Access Nivolumab and ipilimumab in recurrent or refractory cancer of unknown primary: a phase II trial
Nivolumab and ipilimumab in recurrent or refractory cancer of unknown primary: a phase II trial
Standard of care for unfavorable-risk cancer of unknown primary (CUP) comprises platinum-based chemotherapy as first-line treatment, however therapeutic options remain limited. Here the authors report the results of a phase II trial of combined nivolumab (anti-PD1) and ipilimumab (anti-CTLA4) in patients with unfavorable CUP.
- Maria Pouyiourou
- , Bianca N. Kraft
- & Alwin Krämer
-
Article
| Open Access ZSP1601, a novel pan-phosphodiesterase inhibitor for the treatment of NAFLD, A randomized, placebo-controlled phase Ib/IIa trial
ZSP1601, a novel pan-phosphodiesterase inhibitor for the treatment of NAFLD, A randomized, placebo-controlled phase Ib/IIa trial
Non-alcoholic fatty liver disease is a growing health burden with limited treatment options worldwide. Herein the authors report a randomized, double-blind, placebo-controlled, multiple-dose trial of a first-in-class pan-phosphodiesterase inhibitor ZSP1601 in NAFLD patients.
- Yue Hu
- , Haijun Li
- & Yanhua Ding
-
Article
| Open Access Dalpiciclib and pyrotinib in women with HER2-positive advanced breast cancer: a single-arm phase II trial
Dalpiciclib and pyrotinib in women with HER2-positive advanced breast cancer: a single-arm phase II trial
Dalpiciclib is a CDK4/6 inhibitor, recently approved for treatment in advanced breast cancer patients in China. Here, the authors report the results of a phase II trial investigating oral dalpiciclib (CDK4/6 inhibitor) and pyrotinib (pan-HER inhibitor) in patients with HER2-positive metastatic breast cancer.
- Min Yan
- , Limin Niu
- & Huajun Li
-
Article
| Open Access Addressing mechanism bias in model-based impact forecasts of new tuberculosis vaccines
Addressing mechanism bias in model-based impact forecasts of new tuberculosis vaccines
The complex transmission chain of tuberculosis (TB) forces mathematical modelers to make mechanistic assumptions when modelling vaccine effects. Here, authors posit a Bayesian formalism that unlocks mechanism-agnostic impact forecasts for TB vaccines.
- M. Tovar
- , Y. Moreno
- & J. Sanz
-
Article
| Open Access Phase I/II trial of a peptide-based COVID-19 T-cell activator in patients with B-cell deficiency
Phase I/II trial of a peptide-based COVID-19 T-cell activator in patients with B-cell deficiency
Here, Heitmann et al. report results from a Phase I/II trial evaluating CoVac-1, a peptide-based T-cell activator, in patients with B-cell deficiency, demonstrating potent induction of SARS-CoV-2-specific T-cell responses along with a favorable safety profile.
- Jonas S. Heitmann
- , Claudia Tandler
- & Juliane S. Walz
-
Article
| Open Access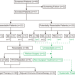 Neoadjuvant Afatinib for stage III EGFR-mutant non-small cell lung cancer: a phase II study
Neoadjuvant Afatinib for stage III EGFR-mutant non-small cell lung cancer: a phase II study
Afatinib is a second-generation EGFR tyrosine kinase inhibitor recommended as the first-line treatment for patients with advanced EGFR mutant non-small cell lung cancer (NSCLC). Here the authors report the results of a phase II clinical trial of neoadjuvant afatinib for stage III EGFR mutant NSCLC.
- Dongliang Bian
- , Liangdong Sun
- & Peng Zhang
-
Article
| Open Access Safety and immunogenicity of a SARS-CoV-2 Gamma variant RBD-based protein adjuvanted vaccine used as booster in healthy adults
Safety and immunogenicity of a SARS-CoV-2 Gamma variant RBD-based protein adjuvanted vaccine used as booster in healthy adults
Here the authors show in a phase 1 trial that a recombinant subunit vaccine based on the gamma variant of SARS-CoV-2 exhibits a satisfactory safety profile, and induces a broad booster response of neutralizing antibodies and a booster effect on T cell immunity in individuals previously immunized with different SARS-CoV-2 vaccine platforms.
- Karina A. Pasquevich
- , Lorena M. Coria
- & Juliana Cassataro
-
Article
| Open Access Safety and immunogenicity of a thermostable ID93 + GLA-SE tuberculosis vaccine candidate in healthy adults
Safety and immunogenicity of a thermostable ID93 + GLA-SE tuberculosis vaccine candidate in healthy adults
Here the authors present results from a randomized, double-blinded Phase 1 clinical trial, testing a thermostable presentation of a clinical-stage adjuvanted subunit tuberculosis vaccine candidate. The vaccine candidate is safe and well tolerated, and elicits comparable or improved immune responses compared to the non-thermostable presentation.
- Zachary K. Sagawa
- , Cristina Goman
- & Christopher B. Fox
-
Perspective
| Open Access Future therapies for cystic fibrosis
Future therapies for cystic fibrosis
The cystic fibrosis landscape has changed dramatically over the last few decades, with improvements in patient quality of life, prognosis and predicted survival. In part, this is related to the availability of novel CFTR modulator drugs, although prior advances in symptom-directed therapies and diagnosis had already led to substantial improvements. However, the authors, part of a national CF focused group, recognize that more needs to be done and outline their considerations on research priorities in this perspective.
- Lucy Allen
- , Lorna Allen
- & Jane C. Davies
-
Article
| Open Access Immunogenicity and efficacy of fourth BNT162b2 and mRNA1273 COVID-19 vaccine doses; three months follow-up
Immunogenicity and efficacy of fourth BNT162b2 and mRNA1273 COVID-19 vaccine doses; three months follow-up
Here the authors provide immunogenicity and efficacy data at 3-month follow-up for vaccinees who have received a fourth dose of either mRNA1273 or BNT162b2. Both vaccines were highly effective against substantial symptomatic disease, but had little effect against SARS-CoV-2 infection.
- Michal Canetti
- , Noam Barda
- & Gili Regev-Yochay
-
Article
| Open Access First-in-human phase Ia study of the PI3Kα inhibitor CYH33 in patients with solid tumors
First-in-human phase Ia study of the PI3Kα inhibitor CYH33 in patients with solid tumors
PIK3CA is a commonly mutated cancer-associated gene, making it an attractive therapeutic target. Here, the authors report the results of a first-in-human phase Ia trial to assess the safety and recommended phase II dose of CYH33, a PI3Kα inhibitor, in patients with advanced solid tumors.
- Xiao-Li Wei
- , Fu-Rong Liu
- & Rui-Hua Xu
-
Article
| Open Access A phase II trial of weekly nab-paclitaxel for progressive and symptomatic desmoid tumors
A phase II trial of weekly nab-paclitaxel for progressive and symptomatic desmoid tumors
In patients with progressive and symptomatic desmoid fibromatosis (DF) treatment with chemotherapy is typically prolonged, while surgery carries a high risk of relapse. Here, the authors report the results of a phase II clinical trial of short-course nab-paclitaxel in patients with progressive and symptomatic DF.
- Javier Martin-Broto
- , Andres Redondo
- & Nadia Hindi
-
Article
| Open Access The metabolic effects of adding exenatide to basal insulin therapy when targeting remission in early type 2 diabetes in a randomized clinical trial
The metabolic effects of adding exenatide to basal insulin therapy when targeting remission in early type 2 diabetes in a randomized clinical trial
Combining a GLP-1 receptor agonist, to increase endogenous beta cell function, with exogenous insulin is a possible therapeutic option for the treatment of type 2 diabetes. Here, the authors show adding exenatide to basal insulin in early type 2 diabetes does not further enhance underlying pancreatic beta-cell function or the capacity to achieve diabetes remission, but may yield on-treatment glycemic benefits.
- Ravi Retnakaran
- , Chang Ye
- & Bernard Zinman
-
Article
| Open Access Heterologous vector versus homologous mRNA COVID-19 booster vaccination in non-seroconverted immunosuppressed patients: a randomized controlled trial
Heterologous vector versus homologous mRNA COVID-19 booster vaccination in non-seroconverted immunosuppressed patients: a randomized controlled trial
Optimizing COVID-19 vaccination strategies for patients under immunosuppressive medication is of high importance. In this clinical trial including non-seroconverted immunosuppressed patients, a homologous mRNA booster vaccination resulted in higher seroconversion rate than a switch to a vector-based vaccine.
- Daniel Mrak
- , Daniela Sieghart
- & Michael Bonelli
-
Article
| Open Access Results of the phase I CCTG IND.231 trial of CX-5461 in patients with advanced solid tumors enriched for DNA-repair deficiencies
Results of the phase I CCTG IND.231 trial of CX-5461 in patients with advanced solid tumors enriched for DNA-repair deficiencies
G-quadruplex stabilizers, including CX-5461, exhibit synthetic lethality with loss of BRCA1/2 in preclinical models. Here the authors report the results of a phase I study of CX-5461 in patients with solid tumors enriched for DNA-repair deficiencies.
- John Hilton
- , Karen Gelmon
- & David W. Cescon
-
Article
| Open Access Safety and serum distribution of anti-SARS-CoV-2 monoclonal antibody MAD0004J08 after intramuscular injection
Safety and serum distribution of anti-SARS-CoV-2 monoclonal antibody MAD0004J08 after intramuscular injection
Monoclonal antibodies (mAbs) are a potential therapeutic option for treatment of COVID-19. Here, the authors report safety and pharmacokinetics of an intramuscularly injected mAb (MAD0004J08) during the first 30 days of a phase 1 clinical study.
- Simone Lanini
- , Stefano Milleri
- & Rino Rappuoli
-
Article
| Open Access Phase II trial of cytarabine and mitoxantrone with devimistat in acute myeloid leukemia
Phase II trial of cytarabine and mitoxantrone with devimistat in acute myeloid leukemia
Combining cytarabine and mitoxantrone with the tricarboxylic acid cycle inhibitor devimistat has been reported in a phase I clinical trial with relapsed or refractory acute myeloid leukaemia (AML). Here, the authors report the outcomes of a phase II study, analyse samples from both phases and perform preclinical analyses that show mitochondrial fission or autophagy inhibition sensitizes AML cells to devimistat.
- Rebecca Anderson
- , Lance D. Miller
- & Timothy S. Pardee
-
Article
| Open Access Tolerogenic nanoparticles mitigate the formation of anti-drug antibodies against pegylated uricase in patients with hyperuricemia
Tolerogenic nanoparticles mitigate the formation of anti-drug antibodies against pegylated uricase in patients with hyperuricemia
Anti-drug antibodies (ADA) induced by biologic drugs may hamper the efficacy of treatment, so inhibiting ADA induction is desirable. Here, in two clinical trials, the authors show that ImmTOR, previously reported to reduce drug immunogenicity in animal studies, helps mitigate ADA induced by pegylated uricase for treating patients with hyperuricemia.
- Earl Sands
- , Alan Kivitz
- & Takashi Kei Kishimoto
-
Article
| Open Access Neoadjuvant immunotherapy with nivolumab and ipilimumab induces major pathological responses in patients with head and neck squamous cell carcinoma
Neoadjuvant immunotherapy with nivolumab and ipilimumab induces major pathological responses in patients with head and neck squamous cell carcinoma
Immune checkpoint blockade has become standard care for patients with recurrent metastatic head and neck squamous cell carcinoma (HNSCC). Here the authors present the results of a non-randomized phase Ib/IIa trial, reporting safety and efficacy of neoadjuvant nivolumab monotherapy and nivolumab plus ipilimumab prior to standard-of-care surgery in patients with HNSCC. .
- Joris L. Vos
- , Joris B. W. Elbers
- & Charlotte L. Zuur
-
Article
| Open Access A randomized study to evaluate safety and immunogenicity of the BNT162b2 COVID-19 vaccine in healthy Japanese adults
A randomized study to evaluate safety and immunogenicity of the BNT162b2 COVID-19 vaccine in healthy Japanese adults
Here the authors provide the interim analysis of an ongoing phase 1/2 study of the BNT162b2 vaccine in healthy Japanese adults. They report mainly mild to moderate local reactions and no serious adverse events as well as good antibody induction one month after the second dose.
- Miwa Haranaka
- , James Baber
- & Stephen Lockhart
-
Article
| Open Access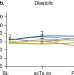 Noninvasive spinal stimulation safely enables upright posture in children with spinal cord injury
Noninvasive spinal stimulation safely enables upright posture in children with spinal cord injury
Scoliosis due to trunk muscle paralysis frequently requires surgical treatment in children with spinal cord injury. The authors demonstrate the safety and efficacy of transcutaneous spinal stimulation to enable upright sitting posture in 7/8 children with trunk control impairment in a within-subjects, repeated measures pilot clinical trial.
- Anastasia Keller
- , Goutam Singh
- & Andrea L. Behrman
-
Article
| Open Access The PEMDAC phase 2 study of pembrolizumab and entinostat in patients with metastatic uveal melanoma
The PEMDAC phase 2 study of pembrolizumab and entinostat in patients with metastatic uveal melanoma
The authors report the results of the phase II PEMDAC clinical study testing the combination of the HDAC inhibitor entinostat with the anti- PD-1 antibody pembrolizumab in uveal melanoma. Low tumor burden, a wildtype BAP1 gene in the tumor or iris melanoma correlates with response and longer survival.
- Lars Ny
- , Henrik Jespersen
- & Jonas A. Nilsson
-
Article
| Open Access Lack of consideration of sex and gender in COVID-19 clinical studies
Lack of consideration of sex and gender in COVID-19 clinical studies
Sex and gender have been associated with differences in SARS-CoV-2 incidence and clinical outcomes and therefore warrant consideration in study designs. Here, the authors assess registered and published clinical COVID-19 studies and find that sex-disaggregated analyses are infrequently presented or planned.
- Emer Brady
- , Mathias Wullum Nielsen
- & Sabine Oertelt-Prigione
-
Article
| Open Access In-silico trial of intracranial flow diverters replicates and expands insights from conventional clinical trials
In-silico trial of intracranial flow diverters replicates and expands insights from conventional clinical trials
In-silico trials rely on virtual populations and interventions simulated using patient-specific models and may offer a solution to lower costs. Here, the authors present the flow diverter performance assessment in-silico trial, which models the treatment of intracranial aneurysms with a flow-diverting stent.
- Ali Sarrami-Foroushani
- , Toni Lassila
- & Alejandro F. Frangi
-
Article
| Open Access Gut microbiota diversity after autologous fecal microbiota transfer in acute myeloid leukemia patients
Gut microbiota diversity after autologous fecal microbiota transfer in acute myeloid leukemia patients
The combination of chemotherapy and broad-spectrum antibiotics induces gut microbiota (GM) dysbiosis in acute myeloid leukaemia (AML) leading to additional complications. Here, the authors report the efficacy in GM restoration and safety of autologous faecal microbiota transfer in treated AML patients in a phase II clinical trial.
- Florent Malard
- , Anne Vekhoff
- & Mohamad Mohty
-
Article
| Open Access Heterologous protection against malaria by a simple chemoattenuated PfSPZ vaccine regimen in a randomized trial
Heterologous protection against malaria by a simple chemoattenuated PfSPZ vaccine regimen in a randomized trial
In this placebo-controlled trial, 10/13 malaria naïve subjects immunized with a simplified regimen of chemoattenuated P. falciparum sporozoites, PfSPZ-CVac, show sterile protection from heterologous malaria challenge. Immunization was well tolerated and induced high levels of anti-PfCSP antibodies.
- Zita Sulyok
- , Rolf Fendel
- & Peter G. Kremsner
-
Matters Arising
| Open AccessHypothermic machine perfusion before viability testing of previously discarded human livers
- Otto B. van Leeuwen
- , Yvonne de Vries
- & Robert J. Porte
-
Article
| Open Access The effects of releasing early results from ongoing clinical trials
The effects of releasing early results from ongoing clinical trials
Sharing early evidence, as a trial is ongoing, is fundamental for both physicians and patients to make enrollment decisions. Here, the authors report the results of a simulation study evaluating the potential effects of early release of interim efficacy and safety data on the duration and validity of an ongoing clinical trial and demonstrate that positive interim results may shorten trial duration through increased enrollment.
- Steffen Ventz
- , Sergio Bacallado
- & Lorenzo Trippa
-
Article
| Open Access Co-administered antibody improves penetration of antibody–dye conjugate into human cancers with implications for antibody–drug conjugates
Co-administered antibody improves penetration of antibody–dye conjugate into human cancers with implications for antibody–drug conjugates
Antibody-drug conjugates targeting high expression receptors can suffer from poor tumour penetration. Here, the authors use unconjugated antibody to improve the penetration of an antibody-dye conjugate in a clinical study, supporting further clinical investigation of the co-administration strategy.
- Guolan Lu
- , Naoki Nishio
- & Eben L. Rosenthal
-
Article
| Open Access Phase I clinical trial repurposing all-trans retinoic acid as a stromal targeting agent for pancreatic cancer
Phase I clinical trial repurposing all-trans retinoic acid as a stromal targeting agent for pancreatic cancer
All-trans retinoic acid - ATRA- is known to remodulate the stroma of pancreatic cancer in mice. Here, the authors carried out a Phase Ib trial in pancreatic patients and show that ATRA in combination with chemotherapy is a safe potential treatment for patients with advanced pancreatic cancer, and demonstrate a stromal modulatory effect.
- Hemant M. Kocher
- , Bristi Basu
- & David J. Propper
-
Article
| Open Access A nonrandomized open-label phase 2 trial of nonischemic heart preservation for human heart transplantation
A nonrandomized open-label phase 2 trial of nonischemic heart preservation for human heart transplantation
Ischemia and reperfusion damage contribute to early graft dysfunction and recipient’s death. Here the authors show the feasibility and safety of a non-ischemic heart preservation method for heart transplantation in a non-randomized trial.
- Johan Nilsson
- , Victoria Jernryd
- & Stig Steen
-
Article
| Open Access Clonal kinetics and single-cell transcriptional profiling of CAR-T cells in patients undergoing CD19 CAR-T immunotherapy
Clonal kinetics and single-cell transcriptional profiling of CAR-T cells in patients undergoing CD19 CAR-T immunotherapy
Understanding factors that impact CAR T cell expansion in the clinic is crucial to improving its therapeutic success. Here the authors document heterogeneity in the clonal dynamics of CAR-T cells by tracking individual clones using the endogenous TCR and integration sites and provide further insights into the role of transcriptional states in clonal kinetics.
- Alyssa Sheih
- , Valentin Voillet
- & Cameron J. Turtle
-
Article
| Open Access A zwitterionic near-infrared fluorophore for real-time ureter identification during laparoscopic abdominopelvic surgery
A zwitterionic near-infrared fluorophore for real-time ureter identification during laparoscopic abdominopelvic surgery
Iatrogenic injury of the ureters is a feared complication of laparoscopic abdominal surgery. Here the authors present the NIR fluorophore ZW800-1 as an intraoperative imaging agent for ureter mapping, showing its safety, pharmacokinetic properties, and efficacy in healthy volunteers and patients undergoing abdominopelvic surgery.
- Kim S. de Valk
- , Henricus J. Handgraaf
- & Alexander L. Vahrmeijer
-
Article
| Open Access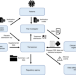 Prototype of running clinical trials in an untrustworthy environment using blockchain
Prototype of running clinical trials in an untrustworthy environment using blockchain
Ensuring the integrity of clinical trial data is crucial to securing trust in the process. Here, the authors present a prototype of a blockchain-based clinical trial management system that ensures immutability and traceability of trial data, and demonstrate a proof of concept web portal service.
- Daniel R. Wong
- , Sanchita Bhattacharya
- & Atul J. Butte
-
Article
| Open Access Implementation and benchmarking of a novel analytical framework to clinically evaluate tumor-specific fluorescent tracers
Implementation and benchmarking of a novel analytical framework to clinically evaluate tumor-specific fluorescent tracers
Fluorescent tracers are being tested in clinical trials to improve detection of tumor margins, but procedures are not standardised. Here, the authors develop an analytical framework that is compatible with the workflow in the operating theatre, and show that it leads to an 88% increase in intraoperative detection of tumor margins in patients with breast cancer.
- Marjory Koller
- , Si-Qi Qiu
- & Gooitzen M. van Dam
-
Article
| Open Access Refined efficacy estimates of the Sanofi Pasteur dengue vaccine CYD-TDV using machine learning
Refined efficacy estimates of the Sanofi Pasteur dengue vaccine CYD-TDV using machine learning
Clinical trials for the CYD-TDV dengue vaccine showed that vaccine efficacy varies with prior dengue exposure, but baseline serostatus is only known for 12% of subjects. Here, Dorigatti et al. use machine learning to impute baseline serostatus and determine vaccine efficacy by baseline serostatus, age and dengue serotype.
- I. Dorigatti
- , C. A. Donnelly
- & N. M. Ferguson
-
Article
| Open Access Treating cat allergy with monoclonal IgG antibodies that bind allergen and prevent IgE engagement
Treating cat allergy with monoclonal IgG antibodies that bind allergen and prevent IgE engagement
Allergen-specific immunotherapy is used to treat patients affected by acute immunoglobulin E (IgE) responses, but the function mechanism is unclear. Here the authors show that the administration of two cat allergen-specific IgGs reduces allergic responses in mouse models and helps ameliorate clinical symptoms in a phase 1b clinical trial.
- J. M. Orengo
- , A. R. Radin
- & G. D. Yancopoulos
-
Article
| Open Access Fasoracetam in adolescents with ADHD and glutamatergic gene network variants disrupting mGluR neurotransmitter signaling
Fasoracetam in adolescents with ADHD and glutamatergic gene network variants disrupting mGluR neurotransmitter signaling
Stimulant drugs are most commonly used to treat ADHD. Here, the authors demonstrate that in adolescents with ADHD who also have genetic variation in genes impacting metabotropic glutamate signaling, the non-stimulant mGluR activator fasoracetam is well tolerated and may be beneficial in alleviating symptoms of this disease.
- Josephine Elia
- , Grace Ungal
- & Hakon Hakonarson
-
Article
| Open Access Atezolizumab in combination with bevacizumab enhances antigen-specific T-cell migration in metastatic renal cell carcinoma
Atezolizumab in combination with bevacizumab enhances antigen-specific T-cell migration in metastatic renal cell carcinoma
Cancer immunotherapy can be used in combination with other therapies for a better response. Here, the authors conduct a phase Ib clinical study and report the clinical activity and the immune response of the anti-PDL1 agent, atezolizumab, in combination with bevacizumab in ten patients with metastatic renal cell carcinoma.
- Jeffrey J. Wallin
- , Johanna C. Bendell
- & David F. McDermott
-
Article
| Open Access Vertical suppression of the EGFR pathway prevents onset of resistance in colorectal cancers
Vertical suppression of the EGFR pathway prevents onset of resistance in colorectal cancers
Cancer patients often respond well to primary treatment but then develop resistance. Here, Misale et al. show that dual treatment with EGFR and MEK inhibitors block resistance in mice containing patient-derived xenografts and provide a mathematical model that describes the temporal development of resistant tumour clones.
- Sandra Misale
- , Ivana Bozic
- & Alberto Bardelli
-
Article |
 Glycopeptide analogues of PSGL-1 inhibit P-selectin in vitro and in vivo
Glycopeptide analogues of PSGL-1 inhibit P-selectin in vitro and in vivo
Inhibiting the interaction between the membrane protein P-selectin and its ligand PSGL-1 is thought to block inflammation. Here the authors report an efficient stereoselective synthesis for PSGL-1 glycopeptide mimics and show that these compounds inhibit PSGL-1/P-selectin in vitro and in vivo.
- Venkata R. Krishnamurthy
- , Mohammed Y. R. Sardar
- & Elliot L. Chaikof

 Original COVID-19 priming regimen impacts the immunogenicity of bivalent BA.1 and BA.5 boosters
Original COVID-19 priming regimen impacts the immunogenicity of bivalent BA.1 and BA.5 boosters