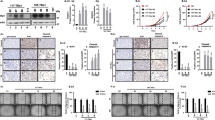
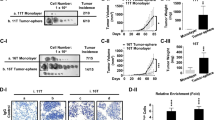
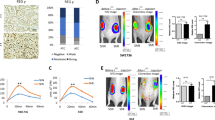
Abstract
Medullary thyroid carcinoma (MTC) is a malignancy derived from the calcitonin-producing C-cells of the thyroid gland. Oncogenic mutations of the Ret proto-oncogene are found in all heritable forms of MTC and roughly one half of the sporadic cases. However, several lines of evidence argue for the existence of additional genetic lesions necessary for the development of MTC. Sprouty (Spry) family of genes is composed of four members in mammals (Spry1-4). Some Spry family members have been proposed as candidate tumor-suppressor genes in a variety of cancerous pathologies. In this work, we show that targeted deletion of Spry1 causes C-cell hyperplasia, a precancerous lesion preceding MTC, in young adult mice. Expression of Spry1 restrains proliferation of the MTC-derived cell line, TT. Finally, we found that the Spry1 promoter is frequently methylated in MTC and that Spry1 expression is consequently decreased. These findings identify Spry1 as a candidate tumor-suppressor gene in MTC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Acton DS, Velthuyzen D, Lips CJ, Hoppener JW . (2000). Multiple endocrine neoplasia type 2B mutation in human RET oncogene induces medullary thyroid carcinoma in transgenic mice. Oncogene 19: 3121–3125.
Airaksinen MS, Saarma M . (2002). The GDNF family: signalling, biological functions and therapeutic value. Nat Rev Neurosci 3: 383–394.
Akeno-Stuart N, Croyle M, Knauf JA, Malaguarnera R, Vitagliano D, Santoro M et al. (2007). The RET kinase inhibitor NVP-AST487 blocks growth and calcitonin gene expression through distinct mechanisms in medullary thyroid cancer cells. Cancer Res 67: 6956–6964.
Aranda S, Alvarez M, Turro S, Laguna A, de la Luna S . (2008). Sprouty2-mediated inhibition of fibroblast growth factor signaling is modulated by the protein kinase DYRK1A. Mol Cell Biol 28: 5899–5911.
Asai N, Jijiwa M, Enomoto A, Kawai K, Maeda K, Ichiahara M et al. (2006). RET receptor signaling: dysfunction in thyroid cancer and Hirschsprung's disease. Pathol Int 56: 164–172.
Baloh RH, Enomoto H, Johnson Jr EM, Milbrandt J . (2000). The GDNF family ligands and receptors - implications for neural development. Curr Opin Neurobiol 10: 103–110.
Basson MA, Akbulut S, Watson-Johnson J, Simon R, Carroll TJ, Shakya R et al. (2005). Sprouty1 is a critical regulator of GDNF/RET-mediated kidney induction. Dev Cell 8: 229–239.
Basson MA, Watson-Johnson J, Shakya R, Akbulut S, Hyink D, Costantini FD et al. (2006). Branching morphogenesis of the ureteric epithelium during kidney development is coordinated by the opposing functions of GDNF and Sprouty1. Dev Biol 299: 466–477.
Cabrita MA, Christofori G . (2008). Sprouty proteins, masterminds of receptor tyrosine kinase signaling. Angiogenesis 11: 53–62.
Cerrato A, De Falco V, Santoro M . (2009). Molecular genetics of medullary thyroid carcinoma: the quest for novel therapeutic targets. J Mol Endocrinol 43: 143–155.
Collado M, Serrano M . (2010). Senescence in tumours: evidence from mice and humans. Nat Rev Cancer 10: 51–57.
Courtois-Cox S, Genther Williams SM, Reczek EE, Johnson BW, McGillicuddy LT, Johannessen CM et al. (2006). A negative feedback signaling network underlies oncogene-induced senescence. Cancer Cell 10: 459–472.
Darimipourain M, Wang S, Ittmann M, Kwabi-Addo B . (2011). Transcriptional and post-transcriptional regulation of Sprouty1, a receptor tyrosine kinase inhibitor in prostate cancer. Prostate Cancer Prostatic Dis 14: 279–285.
Edwin F, Anderson K, Ying C, Patel TB . (2009). Intermolecular interactions of Sprouty proteins and their implications in development and disease. Mol Pharmacol 76: 679–691.
Encinas M, Rozen EJ, Dolcet X, Jain S, Comella JX, Milbrandt J et al. (2008). Analysis of Ret knockin mice reveals a critical role for IKKs, but not PI 3-K, in neurotrophic factor-induced survival of sympathetic neurons. Cell Death Differ 15: 1510–1521.
Eritja N, Llobet D, Domingo M, Santacana M, Yeramian A, Matias-Guiu X et al. (2010). A novel three-dimensional culture system of polarized epithelial cells to study endometrial carcinogenesis. Am J Pathol 176: 2722–2731.
Fagman H, Nilsson M . (2010). Morphogenesis of the thyroid gland. Mol Cell Endocrinol 323: 35–54.
Fong CW, Chua MS, McKie AB, Ling SH, Mason V, Li R et al. (2006). Sprouty 2, an inhibitor of mitogen-activated protein kinase signaling, is down-regulated in hepatocellular carcinoma. Cancer Res 66: 2048–2058.
Frank MJ, Dawson DW, Bensinger SJ, Hong JS, Knosp WM, Xu L et al. (2009). Expression of sprouty2 inhibits B-cell proliferation and is epigenetically silenced in mouse and human B-cell lymphomas. Blood 113: 2478–2487.
Fritzsche S, Kenzelmann M, Hoffmann MJ, Muller M, Engers R, Grone HJ et al. (2006). Concomitant down-regulation of SPRY1 and SPRY2 in prostate carcinoma. Endocr Relat Cancer 13: 839–849.
Gagel RF, Marx SJ . (2003). Multiple endocrine neoplasia. In: Larsen PR, H Kronenberg, S Melmed & K Polonsky (eds). Williams Textbook of Endocrinology 10th edn. Saunders, Elsevier Science: Philadelphia, pp 1717–1762.
Gallel P, Pallares J, Dolcet X, Llobet D, Eritja N, Santacana M et al. (2008). Nuclear factor-kappaB activation is associated with somatic and germ line RET mutations in medullary thyroid carcinoma. Hum Pathol 39: 994–1001.
Guy GR, Jackson RA, Yusoff P, Chow SY . (2009). Sprouty proteins: modified modulators, matchmakers or missing links? J Endocrinol 203: 191–202.
Hacohen N, Kramer S, Sutherland D, Hiromi Y, Krasnow MA . (1998). Sprouty encodes a novel antagonist of FGF signaling that patterns apical branching of the Drosophila airways. Cell 92: 253–263.
Ishida M, Ichihara M, Mii S, Jijiwa M, Asai N, Enomoto A et al. (2007). Sprouty2 regulates growth and differentiation of human neuroblastoma cells through RET tyrosine kinase. Cancer Sci 98: 815–821.
Kawai K, Iwashita T, Murakami H, Hiraiwa N, Yoshiki A, Kusakabe M et al. (2000). Tissue-specific carcinogenesis in transgenic mice expressing the RET proto-oncogene with a multiple endocrine neoplasia type 2A mutation. Cancer Res 60: 5254–5260.
Kwabi-Addo B, Ren C, Ittmann M . (2009). DNA methylation and aberrant expression of Sprouty1 in human prostate cancer. Epigenetics 4: 54–61.
Kwabi-Addo B, Wang J, Erdem H, Vaid A, Castro P, Ayala G et al. (2004). The expression of Sprouty1, an inhibitor of fibroblast growth factor signal transduction, is decreased in human prostate cancer. Cancer Res 64: 4728–4735.
Lanigan F, Geraghty JG, Bracken AP . (2011). Transcriptional regulation of cellular senescence. Oncogene 30: 2901–2911.
Li LC, Dahiya R . (2002). MethPrimer: designing primers for methylation PCRs. Bioinformatics 18: 1427–1431.
Lin AW, Barradas M, Stone JC, van Aelst L, Serrano M, Lowe SW . (1998). Premature senescence involving p53 and p16 is activated in response to constitutive MEK/MAPK mitogenic signaling. Genes Dev 12: 3008–3019.
Litovchick L, Florens LA, Swanson SK, Washburn MP, DeCaprio JA . (2011). DYRK1A protein kinase promotes quiescence and senescence through DREAM complex assembly. Genes Dev 25: 801–813.
Llobet D, Eritja N, Encinas M, Llecha N, Yeramian A, Pallares J et al. (2008). CK2 controls TRAIL and Fas sensitivity by regulating FLIP levels in endometrial carcinoma cells. Oncogene 27: 2513–2524.
Majumdar A, Vainio S, Kispert A, McMahon J, McMahon AP . (2003). Wnt11 and Ret/Gdnf pathways cooperate in regulating ureteric branching during metanephric kidney development. Development 130: 3175–3185.
Mason JM, Morrison DJ, Basson MA, Licht JD . (2006). Sprouty proteins: multifaceted negative-feedback regulators of receptor tyrosine kinase signaling. Trends Cell Biol 16: 45–54.
Matias-Guiu X, DeLellis G, Moley JF, Gagel RF, Albores-Saavedra J, Bussolati G et al. (2004). Medullary thyroid carcinoma. In: DeLellis RA, Lloyd RV, Heitz PU, Eng C (eds). World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of Endocrine Organs, vol. 8. IARC Press: Lyon, pp 86–91.
McKie AB, Douglas DA, Olijslagers S, Graham J, Omar MM, Heer R et al. (2005). Epigenetic inactivation of the human sprouty2 (hSPRY2) homologue in prostate cancer. Oncogene 24: 2166–2174.
Michiels FM, Chappuis S, Caillou B, Pasini A, Talbot M, Monier R et al. (1997). Development of medullary thyroid carcinoma in transgenic mice expressing the RET protooncogene altered by a multiple endocrine neoplasia type 2A mutation. Proc Natl Acad Sci USA 94: 3330–3335.
Michos O, Cebrian C, Hyink D, Grieshammer U, Williams L, D'Agati V et al. (2010). Kidney development in the absence of Gdnf and Spry1 requires Fgf10. PLoS Genet 6: e1000809.
Plaza-Menacho I, Burzynski GM, de Groot JW, Eggen BJ, Hofstra RM . (2006). Current concepts in RET-related genetics, signaling and therapeutics. Trends Genet 22: 627–636.
Prieur A, Peeper DS . (2008). Cellular senescence in vivo: a barrier to tumorigenesis. Curr Opin Cell Biol 20: 150–155.
Reynolds L, Jones K, Winton DJ, Cranston A, Houghton C, Howard L et al. (2001). C-cell and thyroid epithelial tumours and altered follicular development in transgenic mice expressing the long isoform of MEN 2A RET. Oncogene 20: 3986–3994.
Rozen EJ, Schmidt H, Dolcet X, Basson MA, Jain S, Encinas M . (2009). Loss of Sprouty1 rescues renal agenesis caused by Ret mutation. J Am Soc Nephrol 20: 255–259.
Santarpia L, Ye L, Gagel RF . (2009). Beyond RET: potential therapeutic approaches for advanced and metastatic medullary thyroid carcinoma. J Intern Med 266: 99–113.
Shamma A, Takegami Y, Miki T, Kitajima S, Noda M, Obara T et al. (2009). Rb Regulates DNA damage response and cellular senescence through E2F-dependent suppression of N-ras isoprenylation. Cancer Cell 15: 255–269.
Smith-Hicks CL, Sizer KC, Powers JF, Tischler AS, Costantini F . (2000). C-cell hyperplasia, pheochromocytoma and sympathoadrenal malformation in a mouse model of multiple endocrine neoplasia type 2B. Embo J 19: 612–622.
Takahashi M, Ritz J, Cooper GM . (1985). Activation of a novel human transforming gene, ret, by DNA rearrangement. Cell 42: 581–588.
Taketomi T, Yoshiga D, Taniguchi K, Kobayashi T, Nonami A, Kato R et al. (2005). Loss of mammalian Sprouty2 leads to enteric neuronal hyperplasia and esophageal achalasia. Nat Neurosci 8: 855–857.
Thum T, Gross C, Fiedler J, Fischer T, Kissler S, Bussen M et al. (2008). MicroRNA-21 contributes to myocardial disease by stimulating MAP kinase signalling in fibroblasts. Nature 456: 980–984.
Velasco A, Pallares J, Santacana M, Gatius S, Fernandez M, Domingo M et al. (2010). Promoter hypermethylation and expression of sprouty 2 in endometrial carcinoma. Hum Pathol 42: 185–193.
Yaguchi Y, Yu T, Ahmed MU, Berry M, Mason I, Basson MA . (2009). Fibroblast growth factor (FGF) gene expression in the developing cerebellum suggests multiple roles for FGF signaling during cerebellar morphogenesis and development. Dev Dyn 238: 2058–2072.
Ye L, Santarpia L, Cote GJ, El-Naggar AK, Gagel RF . (2008). High resolution array-comparative genomic hybridization profiling reveals deoxyribonucleic acid copy number alterations associated with medullary thyroid carcinoma. J Clin Endocrinol Metab 93: 4367–4372.
Yin L, Puliti A, Bonora E, Evangelisti C, Conti V, Tong WM et al. (2007). C620R mutation of the murine ret proto-oncogene: loss of function effect in homozygotes and possible gain of function effect in heterozygotes. Int J Cancer 121: 292–300.
Zaremba A, Schmuecker U, Esche H . (2011). Sprouty is a cytoplasmic target of adenoviral E1A oncoproteins to regulate the receptor tyrosine kinase signalling pathway. Virol J 8: 192.
Zhu J, Woods D, McMahon M, Bishop JM . (1998). Senescence of human fibroblasts induced by oncogenic Raf. Genes Dev 12: 2997–3007.
Acknowledgements
We are grateful to Esteban J Rozen for his initial contribution to the development of this work. We also thank Ana Velasco and Carme Guerris for technical assistance. This work was supported by grants from Ministerio de Educación y Ciencia (BFU2007-67619 and BFU2010-17628) and Fundació La Marató de TV3, and funding from Suport als Grups de Recerca (2009 SGR 740) from Generalitat de Catalunya to ME; grants 2009SGR794, RD06/0020/1034 and programa de intensificación de la investigación, Instituto Carlos III to XM-G; grants from Fondo de Investigaciones Sanitarias (PI080883) and Fundación Mutua Madrileña (AP2775/2008) to MR; and from the Medical Research Council (G0601104) to MAB Tumor samples were obtained with the support of Xarxa catalana de Bancs de Tumors, the Tumor Banc Platform of RTICC and RD09/0076/00059. AM is supported by a predoctoral fellowship from Universitat de Lleida and was supported by a fellowship from a Fundació Alícia Cuello de Merigó. MV was recipient of a ‘beca d’introducció a la recerca’ fellowship from Universitat de Lleida.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Macià, A., Gallel, P., Vaquero, M. et al. Sprouty1 is a candidate tumor-suppressor gene in medullary thyroid carcinoma. Oncogene 31, 3961–3972 (2012). https://doi.org/10.1038/onc.2011.556
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.556
Keywords
This article is cited by
-
EWS-FLI1-mediated suppression of the RAS-antagonist Sprouty 1 (SPRY1) confers aggressiveness to Ewing sarcoma
Oncogene (2017)
-
KRAS-mediated oncogenic transformation of thyroid follicular cells requires long-term TSH stimulation and is regulated by SPRY1
Laboratory Investigation (2015)
-
The expression of the Sprouty 1 protein inversely correlates with growth, proliferation, migration and invasion of ovarian cancer cells
Journal of Ovarian Research (2014)
-
Sprouty1 induces a senescence-associated secretory phenotype by regulating NFκB activity: implications for tumorigenesis
Cell Death & Differentiation (2014)
-
Medullary Thyroid Carcinoma: a 25-Year Perspective
Endocrine Pathology (2014)