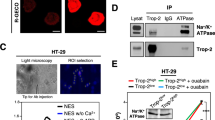
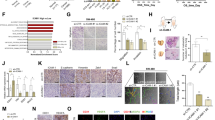
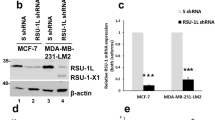
Abstract
The nonreceptor tyrosine kinases of the Src family (SFK) are frequently deregulated in human colorectal cancer (CRC), and they have been implicated in tumour growth and metastasis. How SFK are activated in this cancer has not been clearly established. Here, we show that the SFK-dependent invasion is induced by inactivation of the negative regulator C-terminal Src kinase, Csk. While the level of Csk was inconsistent with SFK activity in colon cancer cells, its membrane translocation, needed for efficient regulation of membrane-localized SFK activity, was impaired. Accordingly, Csk downregulation did not affect SFK oncogenic activity in these cells, whereas expression of a membrane-localized form of this kinase affected their invasive activity. Downregulation of the transmembrane and rafts-localized Csk-binding protein/phosphoprotein associated with glycosphingolipid-enriched microdomain (PAG), was instrumental for the cytoplasmic accumulation of Csk. Re-expression of PAG in cells from late-stage CRC inhibited SFK invasive activity in a Csk-dependent manner. Conversely, inactivation of its residual expression in early-stage CRC cells promoted SFK invasive activity. Finally, this mechanism was specific to CRC as Csk coupling to SFK was readily detected in breast cancer cells. Therefore, Csk mis-localization defines a novel mechanism for SFK oncogenic activation in CRC cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Baumeister U, Funke R, Ebnet K, Vorschmitt H, Koch S, Vestweber D . (2005). Association of Csk to VE-cadherin and inhibition of cell proliferation. EMBO J 24: 1686–1695.
Bénistant C, Bourgaux J, Chapuis H, Mottet N, Roche S, Bali J . (2001). The C-terminal Src kinase is a tumour antigen in human carcinoma. Cancer Res 61: 1415–1420.
Boggon TJ, Eck MJ . (2004). Structure and regulation of Src family kinases. Oncogene 23: 7918–7927.
Boureux A, Furstoss O, Simon V, Roche S . (2005). c-Abl tyrosine kinase regulates a Rac/JNK and a Rac/Nox pathway for DNA synthesis and c-myc expression induced by growth factors. J Cell Sci 118: 3717–3726.
Brdicka T, Pavlistova D, Leo A, Bruyns E, Korinek V, Angelisova P et al. (2000). Phosphoprotein associated with glycosphingolipid-enriched microdomains (PAG), a novel ubiquitously expressed transmembrane adaptor protein, binds the protein tyrosine kinase csk and is involved in regulation of T cell activation. J Exp Med 191: 1591–1604.
Cam WR, Masaki T, Shiratori Y, Kato N, Ikenoue T, Okamoto M et al. (2001). Reduced C-terminal Src kinase activity is correlated inversely with pp60(c-src) activity in colorectal carcinoma. Cancer 92: 61–70.
Causeret M, Taulet N, Comunale F, Favard C, Gauthier-Rouviere C . (2005). N-cadherin association with lipid rafts regulates its dynamic assembly at cell-cell junctions in C2C12 myoblasts. Mol Biol Cell 16: 2168–2180.
Collin G, Franco M, Simon V, Benistant C, Roche S . (2007). The Tom1L1-clathrin heavy chain complex regulates membrane partitioning of the tyrosine kinase Src required for mitogenic and transforming activities. Mol Cell Biol 27: 7631–7640.
Emaduddin M, Bicknell DC, Bodmer WF, Feller SM . (2008). Cell growth, global phosphotyrosine elevation, and c-Met phosphorylation through Src family kinases in colorectal cancer cells. Proc Natl Acad Sci USA 105: 2358–2362.
Frame MC . (2004). Newest findings on the oldest oncogene; how activated src does it. J Cell Sci 117: 989–998.
Horejsi V, Zhang W, Schraven B . (2004). Transmembrane adaptor proteins: organizers of immunoreceptor signalling. Nat Rev Immunol 4: 603–616.
Imamoto A, Soriano P . (1993). Disruption of the Csk gene, encoding a negative regulator of Src family tyrosine kinases, leads to neural tube defects and embryonic lethality in mice. Cell 73: 1117–1124.
Ingley E, Schneider JR, Payne CJ, McCarthy DJ, Harder KW, Hibbs ML et al. (2006). Csk-binding protein mediates sequential enzymatic down-regulation and degradation of Lyn in erythropoietin-stimulated cells. J Biol Chem 281: 31920–31929.
Irby RB, Mao W, Coppola D, Kang J, Loubeau JM, Trudeau W et al. (1999). Activating SRC mutation in a subset of advanced human colon cancers. Nat Genet 21: 187–190.
Ishizawar RC, Tice DA, Karaoli T, Parsons SJ . (2004). The C terminus of c-Src inhibits breast tumor cell growth by a kinase-independent mechanism. J Biol Chem 279: 23773–23781.
Kawabuchi M, Satomi Y, Takao T, Shimonishi Y, Nada S, Nagai K et al. (2000). Transmembrane phosphoprotein Cbp regulates the activities of Src-family tyrosine kinases. Nature 404: 999–1003.
Koegl M, Kypta RM, Bergman M, Alitalo K, Courtneidge SA . (1994). Rapid and efficient purification of Src homology 2 domain-containing proteins: Fyn, Csk and phosphatidylinositol 3-kinase p85. Biochem J 302: 737–744.
Kunte DP, Wali RK, Koetsier JL, Hart J, Kostjukova MN, Kilimnik AY et al. (2005). Down-regulation of the tumor suppressor gene C-terminal Src kinase: an early event during premalignant colonic epithelial hyperproliferation. FEBS Lett 579: 3497–3502.
Lee H, Volonte D, Galbiati F, Iyengar P, Lublin DM, Bregman DB et al. (2000). Constitutive and growth factor-regulated phosphorylation of caveolin-1 occurs at the same site (Tyr-14) in vivo: identification of a c-Src/Cav-1/Grb7 signaling cassette. Mol Endocrinol 14: 1750–1775.
Leroy C, Fialin C, Sirvent A, Simon V, Urbach S, Poncet J et al. (2009). Quantitative phosphoproteomics reveals a cluster of tyrosine kinases that mediates Src invasive activity in advanced colon carcinoma cells. Cancer Res 69: 2279–2286.
Liang F, Liang J, Wang WQ, Sun JP, Udho E, Zhang ZY . (2007). PRL3 promotes cell invasion and proliferation by down-regulation of Csk leading to Src activation. J Biol Chem 282: 5413–5419.
Mao W, Irby R, Coppola D, Fu L, Wloch M, Turner J et al. (1997). Activation of c-Src by receptor tyrosine kinases in human colon cancer cells with high metastatic potential. Oncogene 15: 3083–3090.
Miyagi T, Wada T, Yamaguchi K . (2008). Roles of plasma membrane-associated sialidase NEU3 in human cancers. Biochim Biophys Acta 1780: 532–537.
Nada S, Yagi T, Takeda H, Tokunaga T, Nakagawa H, Ikawa Y et al. (1993). Constitutive activation of Src family kinases in mouse embryos that lack Csk. Cell 73: 1125–1135.
Nusrat A, Parkos CA, Verkade P, Foley CS, Liang TW, Innis-Whitehouse W et al. (2000). Tight junctions are membrane microdomains. J Cell Sci 113: 1771–1781.
Oneyama C, Hikita T, Enya K, Dobenecker MW, Saito K, Nada S et al. (2008). The lipid raft-anchored adaptor protein Cbp controls the oncogenic potential of c-Src. Mol Cell 30: 426–436.
Pannequin J, Delaunay N, Buchert M, Surrel F, Bourgaux JF, Ryan J et al. (2007). Beta-catenin/Tcf-4 inhibition after progastrin targeting reduces growth and drives differentiation of intestinal tumors. Gastroenterology 133: 1554–1568.
Read RD, Bach EA, Cagan RL . (2004). Drosophila C-terminal Src kinase negatively regulates organ growth and cell proliferation through inhibition of the Src, Jun N-terminal kinase, and STAT pathways. Mol Cell Biol 24: 6676–6689.
Resh MD . (1999). Fatty acylation of proteins: new insights into membrane targeting of myristoylated and palmitoylated proteins. Biochim Biophys Acta 1451: 1–16.
Saito K, Enya K, Oneyama C, Hikita T, Okada M . (2008). Proteomic identification of ZO-1/2 as a novel scaffold for Src/Csk regulatory circuit. Biochem Biophys Res Commun 366: 969–975.
Schaller MD, Parsons JT . (1995). pp125FAK-dependent tyrosine phosphorylation of paxillin creates a high-affinity binding site for Crk. Mol Cell Biol 15: 2635–2645.
Sirvent A, Boureux A, Simon V, Leroy C, Roche S . (2007). The tyrosine kinase Abl is required for Src-transforming activity in mouse fibroblasts and human breast cancer cells. Oncogene 26: 7313–7323.
Smida M, Posevitz-Fejfar A, Horejsi V, Schraven B, Lindquist JA . (2007). A novel negative regulatory function of PAG: blocking Ras activation. Blood 110: 596–615.
Solheim SA, Torgersen KM, Tasken K, Berge T . (2008). Regulation of FynT function by dual domain docking on PAG/Cbp. J Biol Chem 283: 2773–2783.
Stewart RA, Li DM, Huang H, Xu T . (2003). A genetic screen for modifiers of the lats tumor suppressor gene identifies C-terminal Src kinase as a regulator of cell proliferation in Drosophila. Oncogene 22: 6436–6444.
Summy JM, Gallick GE . (2003). Src family kinases in tumor progression and metastasis. Cancer Metastasis Rev 22: 337–358.
Tan M, Li P, Klos KS, Lu J, Lan KH, Nagata Y et al. (2005). ErbB2 promotes Src synthesis and stability: novel mechanisms of Src activation that confer breast cancer metastasis. Cancer Res 65: 1858–1867.
Tauzin S, Ding H, Khatib K, Ahmad I, Burdevet D, van Echten-Deckert G et al. (2008). Oncogenic association of the Cbp/PAG adaptor protein with the Lyn tyrosine kinase in human B-NHL rafts. Blood 111: 2310–2320.
Thomas SM, Brugge JS . (1997). Cellular functions regulated by Src family kinases. Annu Rev Cell Dev Biol 13: 513–609.
Veracini L, Franco M, Boureux A, Simon V, Roche S, Benistant C . (2006). Two distinct pools of Src family tyrosine kinases regulate PDGF-induced DNA synthesis and actin dorsal ruffles. J Cell Sci 20: 2921–2934.
Veracini L, Simon V, Richard V, Schraven B, Horejsi V, Roche S et al. (2008). The Csk-binding protein PAG regulates PDGF-induced Src mitogenic signaling via GM1. J Cell Biol 182: 603–614.
Yeatman TJ . (2004). A renaissance for SRC. Nat Rev Cancer 4: 470–480.
Zhang SQ, Yang W, Kontaridis MI, Bivona TG, Wen G, Araki T et al. (2004). Shp2 regulates SRC family kinase activity and Ras/Erk activation by controlling Csk recruitment. Mol Cell 13: 341–355.
Zhao M, Janas JA, Niki M, Pandolfi PP, Van Aelst L . (2006). Dok-1 independently attenuates Ras/mitogen-activated protein kinase and Src/c-myc pathways to inhibit platelet-derived growth factor-induced mitogenesis. Mol Cell Biol 26: 2479–2489.
Zheng X, Resnick RJ, Shalloway D . (2008). Apoptosis of estrogen-receptor negative breast cancer and colon cancer cell lines by PTP alpha and src RNAi. Int J Cancer 122: 1999–2007.
Zhu S, Bjorge JD, Cheng HC, Fujita DJ . (2008). Decreased CHK protein levels are associated with Src activation in colon cancer cells. Oncogene 27: 2027–2034.
Zhu S, Bjorge JD, Fujita DJ . (2007). PTP1B contributes to the oncogenic properties of colon cancer cells through Src activation. Cancer Res 67: 10129–10137.
Acknowledgements
We thank M Okada, S Parsons, P Mangeat and N Morin for various reagents; C Naudin, G Collin and C Leroy for technical assistance; Sylvain De Rossi and Virginie Georget (Montpellier RIO Imaging platform) for confocal microscopy analysis; and J Hickmann for helpful discussion. This work was supported by Servier Laboratories, la Ligue Nationale Contre le Cancer, the CNRS, the University of Montpellier 1 and 2 and l’Association pour la Recherche contre le Cancer (ARC, n° 4025). AS was supported by Servier Laboratories and by the INCa. SR and CB are INSERM investigators.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Sirvent, A., Bénistant, C., Pannequin, J. et al. Src family tyrosine kinases-driven colon cancer cell invasion is induced by Csk membrane delocalization. Oncogene 29, 1303–1315 (2010). https://doi.org/10.1038/onc.2009.450
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.450
Keywords
This article is cited by
-
A renaissance for YES in cancer
Oncogene (2023)
-
Regulation of Src tumor activity by its N-terminal intrinsically disordered region
Oncogene (2022)
-
XIAP Interaction with E2F1 and Sp1 via its BIR2 and BIR3 domains specific activated MMP2 to promote bladder cancer invasion
Oncogenesis (2019)
-
Genome-wide methylation profiling of ovarian cancer patient-derived xenografts treated with the demethylating agent decitabine identifies novel epigenetically regulated genes and pathways
Genome Medicine (2016)
-
CUB-domain-containing protein 1 overexpression in solid cancers promotes cancer cell growth by activating Src family kinases
Oncogene (2015)