Abstract
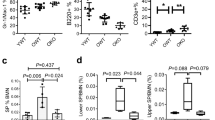
Somatic stem cells are ultimately responsible for mediating appropriate organ homeostasis and have therefore been proposed to represent a cellular origin of the ageing process—a state often characterized by inappropriate homeostasis. Specifically, it has been suggested that ageing stem cells might succumb to replicative senescence by a mechanism involving the cyclin-dependent kinase inhibitor p16INK4A. Here, we tested multiple functional and molecular parameters indicative of p16INK4A activity in primary aged murine hematopoietic stem cells (HSCs). We found no evidence that replicative senescence accompanies stem cell ageing in vivo, and in line with p16INK4A being a critical determinant of such processes, most aged HSCs (>99%) failed to express p16INK4A at the mRNA level. Moreover, whereas loss of epigenetically guided repression of the INK4A/ARF locus accompanied replicative senescent murine embryonic fibroblasts, such repression was maintained in aged stem cells. Taken together, these studies indicate that increased senescence as mediated by the p16INK4A tumor suppressor has only a minor function as an intrinsic regulator of steady-state HSC ageing in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Attema JL, Papathanasiou P, Forsberg EC, Xu J, Smale ST, Weissman IL . (2007). Epigenetic characterization of hematopoietic stem cell differentiation using miniChIP and bisulfite sequencing analysis. Proc Natl Acad Sci USA 104: 12371–12376.
Bracken AP, Kleine-Kohlbrecher D, Dietrich N, Pasini D, Gargiulo G, Beekman C et al. (2007). The Polycomb group proteins bind throughout the INK4A-ARF locus and are disassociated in senescent cells. Genes Dev 21: 525–530.
Bryder D, Rossi DJ, Weissman IL . (2006). Hematopoietic stem cells: the paradigmatic tissue-specific stem cell. Am J Pathol 169: 338–346.
Jacobs JJ, Kieboom K, Marino S, DePinho RA, van Lohuizen M . (1999). The oncogene and Polycomb-group gene bmi-1 regulates cell proliferation and senescence through the ink4a locus. Nature 397: 164–168.
Janzen V, Forkert R, Fleming HE, Saito Y, Waring MT, Dombkowski DM et al. (2006). Stem-cell ageing modified by the cyclin-dependent kinase inhibitor p16INK4a. Nature 443: 421–426.
Kiel MJ, Yilmaz OH, Iwashita T, Yilmaz OH, Terhorst C, Morrison SJ . (2005). SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 121: 1109–1121.
Kotake Y, Cao R, Viatour P, Sage J, Zhang Y, Xiong Y . (2007). pRB family proteins are required for H3K27 trimethylation and Polycomb repression complexes binding to and silencing p16INK4alpha tumor suppressor gene. Genes Dev 21: 49–54.
Mansson R, Hultquist A, Luc S, Yang L, Anderson K, Kharazi S et al. (2007). Molecular evidence for hierarchical transcriptional lineage priming in fetal and adult stem cells and multipotent progenitors. Immunity 26: 407–419.
Merlo A, Herman JG, Mao L, Lee DJ, Gabrielson E, Burger PC et al. (1995). 5′ CpG island methylation is associated with transcriptional silencing of the tumour suppressor p16/CDKN2/MTS1 in human cancers. Nat Med 1: 686–692.
Nygren JM, Bryder D . (2008). A novel assay to trace proliferation history in vivo reveals that enhanced divisional kinetics accompany loss of hematopoietic stem cell self-renewal. PLoS ONE 3: e3710.
Oguro H, Iwama A, Morita Y, Kamijo T, van Lohuizen M, Nakauchi H . (2006). Differential impact of Ink4a and Arf on hematopoietic stem cells and their bone marrow microenvironment in Bmi1-deficient mice. J Exp Med 203: 2247–2253.
Ohtani N, Zebedee Z, Huot TJ, Stinson JA, Sugimoto M, Ohashi Y et al. (2001). Opposing effects of Ets and Id proteins on p16INK4a expression during cellular senescence. Nature 409: 1067–1070.
Park IK, Qian D, Kiel M, Becker MW, Pihalja M, Weissman IL et al. (2003). Bmi-1 is required for maintenance of adult self-renewing haematopoietic stem cells. Nature 423: 302–305.
Patel AC, Anna CH, Foley JF, Stockton PS, Tyson FL, Barrett JC et al. (2000). Hypermethylation of the p16 (Ink4a) promoter in B6C3F1 mouse primary lung adenocarcinomas and mouse lung cell lines. Carcinogenesis 21: 1691–1700.
Pearce DJ, Anjos-Afonso F, Ridler CM, Eddaoudi A, Bonnet D . (2007). Age-dependent increase in side population distribution within hematopoiesis: implications for our understanding of the mechanism of aging. Stem Cells 25: 828–835.
Pronk CJ, Rossi DJ, Mansson R, Attema JL, Norddahl GL, Chan CK et al. (2007). Elucidation of the phenotypic, functional, and molecular topography of a myeloerythroid progenitor cell hierarchy. Cell Stem Cell 1: 428–442.
Randall TD, Weissman IL . (1998). Characterization of a population of cells in the bone marrow that phenotypically mimics hematopoietic stem cells: resting stem cells or mystery population? Stem Cells 16: 38–48.
Reik W . (2007). Stability and flexibility of epigenetic gene regulation in mammalian development. Nature 447: 425–432.
Ringrose L, Paro R . (2007). Polycomb/Trithorax response elements and epigenetic memory of cell identity. Development 134: 223–232.
Rossi DJ, Bryder D, Zahn JM, Ahlenius H, Sonu R, Wagers AJ et al. (2005). Cell intrinsic alterations underlie hematopoietic stem cell aging. Proc Natl Acad Sci USA 102: 9194–9199.
Rossi DJ, Jamieson CH, Weissman IL . (2008). Stems cells and the pathways to aging and cancer. Cell 132: 681–696.
Schneider R, Bannister AJ, Myers FA, Thorne AW, Crane-Robinson C, Kouzarides T . (2004). Histone H3 lysine 4 methylation patterns in higher eukaryotic genes. Nat Cell Biol 6: 73–77.
Sharpless NE, DePinho RA . (2007). How stem cells age and why this makes us grow old. Nat Rev Mol Cell Biol 8: 703–713.
Signer RA, Montecino-Rodriguez E, Witte ON, Dorshkind K . (2008). Aging and cancer resistance in lymphoid progenitors are linked processes conferred by p16Ink4a and Arf. Genes Dev 22: 3115–3120.
Sudo K, Ema H, Morita Y, Nakauchi H . (2000). Age-associated characteristics of murine hematopoietic stem cells. J Exp Med 192: 1273–1280.
Zindy F, Quelle DE, Roussel MF, Sherr CJ . (1997). Expression of the p16INK4a tumor suppressor versus other INK4 family members during mouse development and aging. Oncogene 15: 203–211.
Acknowledgements
We thank William Agace for critical review of the paper. The expert technical support from Gerd Sten is greatly appreciated. These studies were generously supported by grants to DB from the Swedish Medical Research Council, the Swedish Strategic Research Foundation, The Swedish Cancer Society, The Crafoord foundation and the Medical Faculty at Lund University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Attema, J., Pronk, C., Norddahl, G. et al. Hematopoietic stem cell ageing is uncoupled from p16INK4A-mediated senescence. Oncogene 28, 2238–2243 (2009). https://doi.org/10.1038/onc.2009.94
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.94
Keywords
This article is cited by
-
Loss of SIRT1 inhibits hematopoietic stem cell aging and age-dependent mixed phenotype acute leukemia
Communications Biology (2022)
-
Ageing and atherosclerosis: vascular intrinsic and extrinsic factors and potential role of IL-6
Nature Reviews Cardiology (2021)
-
Peroxiredoxin 4 protects against ovarian ageing by ameliorating d-galactose-induced oxidative damage in mice
Cell Death & Disease (2020)
-
Signaling Pathways Regulating Hematopoietic Stem Cell and Progenitor Aging
Current Stem Cell Reports (2018)
-
Stem cell aging: mechanisms, regulators and therapeutic opportunities
Nature Medicine (2014)