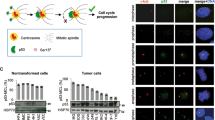
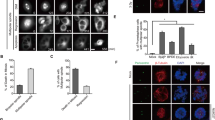
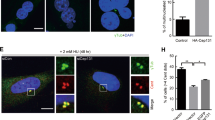
Abstract
Aberrant centrosome numbers are detected in virtually all human cancers where they can contribute to chromosomal instability by promoting mitotic spindle abnormalities. Despite their widespread occurrence, the molecular mechanisms that underlie centrosome amplification are only beginning to emerge. Here, we present evidence for a novel regulatory circuit involved in centrosome overduplication that centers on RNA polymerase II (pol II). We found that human papillomavirus type 16 E7 (HPV-16 E7)- and hydroxyurea (HU)-induced centriole overduplication are abrogated by α-amanitin, a potent and specific RNA pol II inhibitor. In contrast, normal centriole duplication proceeded undisturbed in α-amanitin-treated cells. Centriole overduplication was significantly reduced by siRNA-mediated knock down of CREB-binding protein (CBP), a transcriptional co-activator. We identified cyclin A2 as a key transcriptional target of RNA pol II during HU-induced centriole overduplication. Collectively, our results show that ongoing RNA pol II transcription is required for centriole overduplication whereas it may be dispensable for normal centriole duplication. Given that many chemotherapeutic agents function through inhibition of transcription, our results may help to develop strategies to target centrosome-mediated chromosomal instability for cancer therapy and prevention.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bai VU, Cifuentes E, Menon M, Barrack ER, Reddy GP . (2005). J Cell Physiol 204: 381–387.
Balczon R, Bao L, Zimmer WE, Brown K, Zinkowski RP, Brinkley BR . (1995). J Cell Biol 130: 105–115.
Bornens M . (2002). Curr Opin Cell Biol 14: 25–34.
Braude PR . (1979). Dev Biol 68: 440–452.
Bunz F, Dutriaux A, Lengauer C, Waldman T, Zhou S, Brown JP et al. (1998). Science 282: 1497–1501.
Bushnell DA, Cramer P, Kornberg RD . (2002). Proc Natl Acad Sci USA 99: 1218–1222.
Caldeira S, de Villiers EM, Tommasino M . (2000). Oncogene 19: 821–826.
D'Assoro AB, Busby R, Suino K, Delva E, Almodovar-Mercado GJ, Johnson H et al. (2004). Oncogene 23: 4068–4075.
Derheimer FA, Chang CW, Ljungman M . (2005). Eur J Cancer 41: 2569–2576.
Dodson H, Bourke E, Jeffers LJ, Vagnarelli P, Sonoda E, Takeda S et al. (2004). EMBO J 23: 3864–3873.
Doxsey S . (2002). Mol Cell 10: 439–440.
Duensing A, Liu Y, Tseng M, Malumbres M, Barbacid M, Duensing S . (2006). Oncogene 25: 2943–2949.
Duensing S . (2005). Cell Biol Int 29: 352–359.
Duensing S, Duensing A, Crum CP, Munger K . (2001). Cancer Res 61: 2356–2360.
Duensing S, Duensing A, Lee DC, Edwards KM, Piboonniyom S, Manuel E et al. (2004). Oncogene 23: 8206–8215.
Duensing S, Lee LY, Duensing A, Basile J, Piboonniyom S, Gonzalez S et al. (2000). Proc Natl Acad Sci USA 97: 10002–10007.
Duensing S, Munger K . (2001). Biochim Biophys Acta 2: M81–88.
Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T . (2001). Nature 411: 494–498.
Faivre J, Frank-Vaillant M, Poulhe R, Mouly H, Jessus C, Brechot C et al. (2002). Oncogene 21: 1493–1500.
Fukasawa K . (2005). Cancer Lett 230: 6–19.
Girdham CH, Glover DM . (1991). Genes Dev 5: 1786–1799.
Gong XQ, Nedialkov YA, Burton ZF . (2004). J Biol Chem 279: 27422–27427.
Guarguaglini G, Duncan PI, Stierhof YD, Holmstrom T, Duensing S, Nigg EA . (2005). Mol Biol Cell 16: 1095–1107.
Hahn S . (2004). Nat Struct Mol Biol 11: 394–403.
Hinchcliffe EH, Li C, Thompson EA, Maller JL, Sluder G . (1999). Science 283: 851–854.
Hinchcliffe EH, Sluder G . (2001). Genes Dev 15: 1167–1181.
Hoffelder DR, Luo L, Burke NA, Watkins SC, Gollin SM, Saunders WS . (2004). Chromosoma 112: 389–397.
Janknecht R, Hunter T . (1996). Nature 383: 22–23.
Jiang Y, Liu M, Spencer CA, Price DH . (2004). Mol Cell 14: 375–385.
Kuriyama R, Borisy GG . (1981). J Cell Biol 91: 814–821.
Malumbres M, Barbacid M . (2001). Nat Rev Cancer 1: 222–231.
Mantel C, Braun SE, Reid S, Henegariu O, Liu L, Hangoc G et al. (1999). Blood 93: 1390–1398.
Martin LG, Demers GW, Galloway DA . (1998). J Virol 72: 975–985.
Matsumoto Y, Hayashi K, Nishida E . (1999). Curr Biol 9: 429–432.
McDermott KM, Zhang J, Holst CR, Kozakiewicz BK, Singla V, Tlsty TD . (2006). PLoS Biol 4: e51.
Meraldi P, Lukas J, Fry AM, Bartek J, Nigg EA . (1999). Nat Cell Biol 1: 88–93.
Mihaylov IS, Kondo T, Jones L, Ryzhikov S, Tanaka J, Zheng J et al. (2002). Mol Cell Biol 22: 1868–1880.
Munger K, Basile JR, Duensing S, Eichten A, Gonzalez SL, Grace M et al. (2001). Oncogene 20: 7888–7898.
Munger K, Howley PM . (2002). Virus Res 89: 213–228.
Mussman JG, Horn HF, Carroll PE, Okuda M, Tarapore P, Donehower LA et al. (2000). Oncogene 19: 1635–1646.
Nigg EA . (2002). Nat Rev Cancer 2: 1–11.
Parvin JD, Young RA . (1998). Curr Opin Genet Dev 8: 565–570.
Phillips SG, Rattner JB . (1976). J Cell Biol 70: 9–19.
Salisbury JL, Whitehead CM, Lingle WL, Barrett SL . (1999). Biol Cell 91: 451–460.
Sluder G, Miller FJ, Rieder CL . (1986). J Cell Biol 103: 1873–1881.
Tommasino M, Adamczewski JP, Carlotti F, Barth CF, Manetti R, Contorni M et al. (1993). Oncogene 8: 195–202.
Urbani L, Stearns T . (1999). Curr Biol 9: R315–317.
Uzawa M, Grams J, Madden B, Toft D, Salisbury JL . (1995). Dev Biol 171: 51–59.
Wong C, Stearns T . (2003). Nat Cell Biol 5: 539–544.
Yam CH, Fung TK, Poon RY . (2002). Cell Mol Life Sci 59: 1317–1326.
Acknowledgements
We are grateful to Michel Bornens, Karl Münger and Jeffrey L Salisbury for sharing important reagents and William Saunders for helpful suggestions. This work was supported by Public Health Service Grant NIH/NCI R01 CA112598 (to SD).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duensing, A., Liu, Y., Spardy, N. et al. RNA polymerase II transcription is required for human papillomavirus type 16 E7- and hydroxyurea-induced centriole overduplication. Oncogene 26, 215–223 (2007). https://doi.org/10.1038/sj.onc.1209782
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209782
Keywords
This article is cited by
-
MPS1 is involved in the HPV16-E7-mediated centrosomes amplification
Cell Division (2021)
-
Centrosome amplification: a quantifiable cancer cell trait with prognostic value in solid malignancies
Cancer and Metastasis Reviews (2021)
-
Drosophila, destroying angels, and deathcaps! Oh my! A review of mycotoxin tolerance in the genus Drosophila
Frontiers in Biology (2018)
-
The G1 phase Cdks regulate the centrosome cycle and mediate oncogene-dependent centrosome amplification
Cell Division (2011)
-
The HPV-16 E7 oncoprotein induces centriole multiplication through deregulation of Polo-like kinase 4 expression
Molecular Cancer (2011)