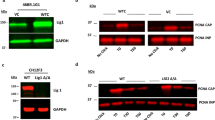
Abstract
The exoRNase Eri1 inhibits RNA interference and trims the 5.8S rRNA 3′ end. It also binds to the stem-loop of histone mRNAs, but the functional importance of this interaction remains elusive. Histone mRNAs are normally degraded at the end of S phase or after pharmacological inhibition of replication. Both processes are impaired in Eri1-deficient mouse cells, which instead accumulate oligouridylated histone mRNAs. Eri1 trims the mature histone mRNAs by two unpaired nucleotides at the 3′ end but stalls close to the double-stranded stem. Upon oligouridylation of the histone mRNA, the Lsm1–7 heteroheptamer recognizes the oligo(U) tail and interacts with Eri1, whose catalytic activity is then able to degrade the stem-loop in a stepwise manner. These data demonstrate how degradation of histone mRNAs is initiated when 3′ oligouridylation creates a cis element that enables Eri1 to process the double-stranded stem-loop structure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Kennedy, S., Wang, D. & Ruvkun, G. A conserved siRNA-degrading RNase negatively regulates RNA interference in C. elegans. Nature 427, 645–649 (2004).
Iida, T., Kawaguchi, R. & Nakayama, J. Conserved ribonuclease, Eri1, negatively regulates heterochromatin assembly in fission yeast. Curr. Biol. 16, 1459–1464 (2006).
Bühler, M., Verdel, A. & Moazed, D. Tethering RITS to a nascent transcript initiates RNAi- and heterochromatin-dependent gene silencing. Cell 125, 873–886 (2006).
Thomas, M.F. et al. Eri1 regulates microRNA homeostasis and mouse lymphocyte development and antiviral function. Blood 120, 130–142 (2012).
Gabel, H.W. & Ruvkun, G. The exonuclease ERI-1 has a conserved dual role in 5.8S rRNA processing and RNAi. Nat. Struct. Mol. Biol. 15, 531–533 (2008).
Ansel, K.M. et al. Mouse Eri1 interacts with the ribosome and catalyzes 5.8S rRNA processing. Nat. Struct. Mol. Biol. 15, 523–530 (2008).
Dominski, Z., Yang, X.C., Kaygun, H., Dadlez, M. & Marzluff, W.F. A 3′ exonuclease that specifically interacts with the 3′ end of histone mRNA. Mol. Cell 12, 295–305 (2003).
Yang, X.C., Purdy, M., Marzluff, W.F. & Dominski, Z. Characterization of 3′hExo, a 3′ exonuclease specifically interacting with the 3′ end of histone mRNA. J. Biol. Chem. 281, 30447–30454 (2006).
Battle, D.J. & Doudna, J.A. The stem-loop binding protein forms a highly stable and specific complex with the 3′ stem-loop of histone mRNAs. RNA 7, 123–132 (2001).
Martin, F., Michel, F., Zenklusen, D., Muller, B. & Schumperli, D. Positive and negative mutant selection in the human histone hairpin-binding protein using the yeast three-hybrid system. Nucleic Acids Res. 28, 1594–1603 (2000).
Williams, A.S. & Marzluff, W.F. The sequence of the stem and flanking sequences at the 3′ end of histone mRNA are critical determinants for the binding of the stem-loop binding protein. Nucleic Acids Res. 23, 654–662 (1995).
Yang, X.C., Torres, M.P., Marzluff, W.F. & Dominski, Z. Three proteins of the U7-specific Sm ring function as the molecular ruler to determine the site of 3′-end processing in mammalian histone pre-mRNA. Mol. Cell. Biol. 29, 4045–4056 (2009).
Marzluff, W.F., Gongidi, P., Woods, K.R., Jin, J. & Maltais, L.J. The human and mouse replication-dependent histone genes. Genomics 80, 487–498 (2002).
Meeks-Wagner, D. & Hartwell, L.H. Normal stoichiometry of histone dimer sets is necessary for high fidelity of mitotic chromosome transmission. Cell 44, 43–52 (1986).
Graves, R.A., Pandey, N.B., Chodchoy, N. & Marzluff, W.F. Translation is required for regulation of histone mRNA degradation. Cell 48, 615–626 (1987).
Pandey, N.B. & Marzluff, W.F. The stem-loop structure at the 3′ end of histone mRNA is necessary and sufficient for regulation of histone mRNA stability. Mol. Cell. Biol. 7, 4557–4559 (1987).
Parker, R. & Song, H. The enzymes and control of eukaryotic mRNA turnover. Nat. Struct. Mol. Biol. 11, 121–127 (2004).
Tharun, S. & Parker, R. Targeting an mRNA for decapping: displacement of translation factors and association of the Lsm1p-7p complex on deadenylated yeast mRNAs. Mol. Cell 8, 1075–1083 (2001).
Wang, Z. & Kiledjian, M. Functional link between the mammalian exosome and mRNA decapping. Cell 107, 751–762 (2001).
Mullen, T.E. & Marzluff, W.F. Degradation of histone mRNA requires oligouridylation followed by decapping and simultaneous degradation of the mRNA both 5′ to 3′ and 3′ to 5′. Genes Dev. 22, 50–65 (2008).
Kaygun, H. & Marzluff, W.F. Regulated degradation of replication-dependent histone mRNAs requires both ATR and Upf1. Nat. Struct. Mol. Biol. 12, 794–800 (2005).
Schmidt, M.J., West, S. & Norbury, C.J. The human cytoplasmic RNA terminal U-transferase ZCCHC11 targets histone mRNAs for degradation. RNA 17, 39–44 (2011).
Sobti, M., Cubeddu, L., Haynes, P.A. & Mabbutt, B.C. Engineered rings of mixed yeast Lsm proteins show differential interactions with translation factors and U-rich RNA. Biochemistry 49, 2335–2345 (2010).
Bonner, W.M., Mannironi, C., Orr, A., Pilch, D.R. & Hatch, C.L. Histone H2A.X gene transcription is regulated differently than transcription of other replication-linked histone genes. Mol. Cell. Biol. 13, 984–992 (1993).
Mannironi, C., Bonner, W.M. & Hatch, C.L. H2A.X. a histone isoprotein with a conserved C-terminal sequence, is encoded by a novel mRNA with both DNA replication type and polyA 3′ processing signals. Nucleic Acids Res. 17, 9113–9126 (1989).
Herrero, A.B. & Moreno, S. Lsm1 promotes genomic stability by controlling histone mRNA decay. EMBO J. 30, 2008–2018 (2011).
Glasmacher, E. et al. Roquin binds inducible costimulator mRNA and effectors of mRNA decay to induce microRNA-independent post-transcriptional repression. Nat. Immunol. 11, 725–733 (2010).
Tharun, S. et al. Yeast Sm-like proteins function in mRNA decapping and decay. Nature 404, 515–518 (2000).
Song, M.G. & Kiledjian, M. 3′ Terminal oligo U-tract-mediated stimulation of decapping. RNA 13, 2356–2365 (2007).
Coller, J. & Parker, R. General translational repression by activators of mRNA decapping. Cell 122, 875–886 (2005).
Hirai, H., Lee, D.I., Natori, S. & Sekimizu, K. Uridylation of U6 RNA in a nuclear extract in Ehrlich ascites tumor cells. J. Biochem. 104, 991–994 (1988).
Heo, I. et al. Lin28 mediates the terminal uridylation of let-7 precursor MicroRNA. Mol. Cell 32, 276–284 (2008).
Jones, M.R. et al. Zcchc11-dependent uridylation of microRNA directs cytokine expression. Nat. Cell Biol. 11, 1157–1163 (2009).
Heo, I. et al. TUT4 in concert with Lin28 suppresses microRNA biogenesis through pre-microRNA uridylation. Cell 138, 696–708 (2009).
Ansel, K.M. et al. Deletion of a conserved Il4 silencer impairs T helper type 1-mediated immunity. Nat. Immunol. 5, 1251–1259 (2004).
Weinmann, L. et al. Importin 8 is a gene silencing factor that targets argonaute proteins to distinct mRNAs. Cell 136, 496–507 (2009).
Ritzi, M. et al. Complex protein-DNA dynamics at the latent origin of DNA replication of Epstein-Barr virus. J. Cell Sci. 116, 3971–3984 (2003).
Lau, N.C., Lim, L.P., Weinstein, E.G. & Bartel, D.P. An abundant class of tiny RNAs with probable regulatory roles in Caenorhabditis elegans. Science 294, 858–862 (2001).
Acknowledgements
We would like to thank W. Pastor and A. Rao (La Jolla Institute of Allergy and Immunology, La Jolla, CA, USA) for providing the FLAG-HA-hERI1 construct. We thank D. Repsilber (Leibniz Institute for Farm Animal Biology, Dummerstorf, Germany) for advice on statistical analyses. R. Lührmann (Max Planck Institute for Biophysical Chemistry, Göttingen, Germany) kindly provided anti-Lsm1 antibodies. This work was supported by the German Research Foundation (DFG HE 3359/3-1 to V.H.).
Author information
Authors and Affiliations
Contributions
V.H. and K.M.A. conceived of the project idea. K.P.H. and V.H. designed and K.P.H. performed most of the experiments with help from C.W., J.D. and A.S. N.R. carried out the reconstitution experiments. N.R. and G.A.H. performed the immunoprecipitation experiments, and E.K. generated the monoclonal antibodies. K.P.H., V.H. and K.M.A. discussed the data, and K.P.H. and V.H. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Tables 1–2 (PDF 5087 kb)
Rights and permissions
About this article
Cite this article
Hoefig, K., Rath, N., Heinz, G. et al. Eri1 degrades the stem-loop of oligouridylated histone mRNAs to induce replication-dependent decay. Nat Struct Mol Biol 20, 73–81 (2013). https://doi.org/10.1038/nsmb.2450
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2450
This article is cited by
-
ISG20L2: an RNA nuclease regulating T cell activation
Cellular and Molecular Life Sciences (2023)
-
Exonuclease requirements for mammalian ribosomal RNA biogenesis and surveillance
Nature Structural & Molecular Biology (2019)
-
Binding of NUFIP2 to Roquin promotes recognition and regulation of ICOS mRNA
Nature Communications (2018)
-
Uridylation and adenylation of RNAs
Science China Life Sciences (2015)
-
Cytoplasmic RNA: a case of the tail wagging the dog
Nature Reviews Molecular Cell Biology (2013)