Abstract
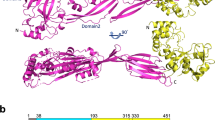
The first structure of a pneumococcal autolysin, that of the LytC lysozyme, has been solved in ternary complex with choline and a pneumococcal peptidoglycan (PG) fragment. The active site of the hydrolase module is not fully exposed but is oriented toward the choline-binding module, which accounts for its unique in vivo features in PG hydrolysis, its activation and its regulatory mechanisms. Because of the unusual hook-shaped conformation of the multimodular protein, it is only able to hydrolyze non–cross-linked PG chains, an assertion validated by additional experiments. These results explain the activation of LytC by choline-binding protein D (CbpD) in fratricide, a competence-programmed mechanism of predation of noncompetent sister cells. The results provide the first structural insights to our knowledge into the critical and central function that LytC plays in pneumococcal virulence and explain a long-standing puzzle of how murein hydrolases can be controlled to avoid self-lysis during bacterial growth and division.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Höltje, J.V. Growth of the stress-bearing and shape-maintaining murein sacculus of Escherichia coli. Microbiol. Mol. Biol. Rev. 62, 181–203 (1998).
Vollmer, W., Joris, B., Charlier, P. & Foster, S. peptidoglycan (murein) hydrolases. FEMS Microbiol. Rev. 32, 259–286 (2008).
López, R. & García, E. Recent trends on the molecular biology of pneumococcal capsules, lytic enzymes, and bacteriophage. FEMS Microbiol. Rev. 28, 553–580 (2004).
Tomasz, A. Choline in the cell wall of a bacterium: novel type of polymer-linked choline in pneumococcus. Science 157, 694–697 (1967).
García, J.L., Sánchez-Beato, A.R., Medrano, F.J. & López, R. Versatility of choline-binding domain. in Streptococcus pneumoniae: Molecular Biology & Mechanisms of Disease (ed. Tomasz, A.) 231–244 (Mary Ann Liebert, Inc., Larchmont, New York, USA, 2000).
López, R., García, E., García, P. & García, J.L. The pneumococcal cell wall degrading enzymes: a modular design to create new lysins? Microb. Drug Resist. 3, 199–211 (1997).
García, P., González, M.P., García, E., García, J.L. & López, R. The molecular characterization of the first autolytic lysozyme of Streptococcus pneumoniae reveals evolutionary mobile domains. Mol. Microbiol. 33, 128–138 (1999).
Claverys, J.P. & Håvarstein, L.S. Cannibalism and fratricide: mechanisms and raisons d'être. Nat. Rev. Microbiol. 5, 219–229 (2007).
Claverys, J.P., Prudhomme, M. & Martin, B. Induction of competence regulons as general stress responses in Gram-positive bacteria. Annu. Rev. Microbiol. 60, 451–475 (2006).
Johnsborg, O., Eldholm, V., Bjørnstad, M.L. & Håvarstein, L.S. A predatory mechanism dramatically increases the efficiency of lateral gene transfer in Streptococcus pneumoniae and related commensal species. Mol. Microbiol. 69, 245–253 (2008).
Eldholm, V., Johnsborg, O., Haugen, K., Ohnstad, H.S. & Håvarstein, L.S. Fratricide in Streptococcus pneumoniae: contributions and role of the cell wall hydrolases CbpD, LytA and LytC. Microbiology 155, 2223–2234 (2009).
Johnsborg, O. & Håvarstein, L.S. Regulation of natural genetic transformation and acquisition of transforming DNA in Streptococcus pneumoniae. FEMS Microbiol. Rev. 33, 627–642 (2009).
Molina, R. et al. Crystal structure of CbpF, a bifunctional choline-binding protein and autolysis regulator from Streptococcus pneumoniae. EMBO Rep. 10, 246–251 (2009).
Gosink, K.K., Mann, E.R., Guglielmo, C., Tuomanen, E.I. & Masure, H.R. Role of novel choline binding proteins in virulence of Streptococcus pneumoniae. Infect. Immun. 68, 5690–5695 (2000).
Klein, R.A., Hartman, R., Egge, H., Behr, T. & Fischer, W. The aqueous solution of a lipoteichoic acid from Streptococcus pneumoniae strain R6 containing 2,4-diamino-2,4,6-trideoxy-galactose: evidence for conformational mobility of the galactopyranose ring. Carbohydr. Res. 281, 79–98 (1996).
Hermoso, J.A. et al. Insights into pneumococcal pathogenesis from the crystal structure of the modular teichoic acid phosphorylcholine esterase Pce. Nat. Struct. Mol. Biol. 12, 533–538 (2005).
Jones, G., Willett, P., Glen, R.C., Leach, A.R. & Taylor, R. Development and validation of a genetic algorithm for flexible docking. J. Mol. Biol. 267, 727–748 (1997).
De Las Rivas, B., García, J.L., López, R. & García, P. Purification and polar localization of the pneumococcal LytB, a putative endo-β-N-acetylglucosaminidase: the chain-dispersing murein hydrolase. J. Bacteriol. 184, 4988–5000 (2002).
Hermoso, J.A. et al. Structural basis for selective recognition of pneumococcal cell wall by modular endolysin from phage Cp-1. Structure 11, 1239–1249 (2003).
Pérez-Dorado, I. et al. Elucidation of the molecular recognition of bacterial cell wall by modular pneumococcal phage endolysin CPL-1. J. Biol. Chem. 282, 24990–24999 (2007).
Monterroso, B., Sáiz, J.L., García, P., García, J.L. & Menéndez, M. Insights into the structure-function relationships of pneumococcal cell wall lysozymes, LytC and Cpl-1. J. Biol. Chem. 283, 28618–28628 (2008).
Meroueh, S.O. et al. Three-dimensional structure of the bacterial cell wall peptidoglycan. Proc. Natl. Acad. Sci. USA 103, 4404–4409 (2006).
Hesek, D., Lee, M., Morio, K. & Mobashery, S. Synthesis of a fragment of bacterial cell wall. J. Org. Chem. 69, 2137–2146 (2004).
Hesek, D. et al. Synthetic peptidoglycan substrates for penicillin-binding protein 5 of Gram-negative bacteria. J. Org. Chem. 69, 778–784 (2004).
Ottolenghi, E. & Hotchkiss, R.D. Appearance of genetic transforming activity in pneumococcal cultures. Science 132, 1257–1258 (1960).
Horne, D.S. & Tomasz, A. Possible role of a choline-containing teichoic acid in the maintenance of normal cell shape and physiology in Streptococcus oralis. J. Bacteriol. 175, 1717–1722 (1993).
Vollmer, W. Preparation and analysis of pneumococcal murein (peptidoglycan). in Molecular Biology of Streptococci (eds. Hakenbeck, R. & Chhatwal, S.) 531–535 (Horizon Scientific Press, Norfolk, UK, 2007).
Severin, A. & Tomasz, A. Naturally occuring peptidoglycan variants of Streptococcus pneumoniae. J. Bacteriol. 178, 168–174 (1996).
Sánchez-Puelles, J.M., Sanz, J.M., García, J.L. & García, E. Cloning and expression of gene fragments encoding the choline-binding domain of pneumococcal murein hydrolases. Gene 89, 69–75 (1990).
Mosser, J.L. & Tomasz, A. Choline-containing teichoic acid as a structural component of pneumococcal cell wall and its role in sensitivity to lysis by an autolytic enzyme. J. Biol. Chem. 245, 287–298 (1970).
Leslie, A.G.W. Profile fitting. in Proceedings of the CCP4 Study Weekend (eds. Machin, J.R. & Papiz, M.Z.) 39–50 (SERC Daresbury Laboratory, Warrington, UK, 1987).
Collaborative Computacional Project 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D Biol. Crystallogr. 50, 760–763 (1994).
Molina, R., Stelter, M., Kahn, R. & Hermoso, J.A. Characterization of gadolinium complexes for SAD phasing in macromolecular crystallography: application to CbpF. Acta Crystallogr. D Biol. Crystallogr. 65, 823–831 (2009).
Jones, T.A., Zou, J.Y., Cowan, S.W. & Kjelgaard, M. Improved methods for model building in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991).
Brunger, A.T. et al. Crystallographic and NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D Biol. Crystallogr. 54, 905–921 (1998).
Laskowski, R.A., MacArthur, M.W., Moss, D.S. & Thorton, J.M. PROCHECK, a program to check the stereochemical quality of protein structures. J. Appl. Crystallogr. 26, 283–291 (1993).
DeLano, W.L. The PyMOL Molecular Graphics System. (DeLano Scientific, San Carlos, California, USA, 2002) <http://www.pymol.org>.
Acknowledgements
We are indebted to E. García for his critical reading of the manuscript, to B. de las Rivas (Instituto de Fermentaciones Industriales, Consejo Superior de Investigaciones Científicas) for the generous gift of the GFP-LytC clone and to M.T. Seisdedos for skillful guidance with the fluorescence study. This work was supported by grants from the Spanish Ministry of Science and Technology (BFU2008-01711, SAF2006-00390), EU-CP223111 (CAREPNEUMO, European Union), the COMBACT program (S-BIO-0260/2006) and CIBER de Enfermedades Respiratorias (CIBERES). CIBERES is an initiative of Instituto de Salud Carlos III. The work in the US was supported by the US National Institutes of Health. W.V. was supported by the European Commission (EUR-INTAFAR project). I.P.-D. was a fellow of the Consejo Superior de Investigaciones Científicas.
Author information
Authors and Affiliations
Contributions
I.P.-D. performed crystallization and structural determination; A.G. and M.M. performed biochemical experiments; R.S. performed crystallization; W.S. and W.V. purified the PG; S.M. synthetized the PG ligands and wrote the manuscript; M.M.-R. wrote the manuscript; J.L.G., P.G. and J.A.H. conceived the study and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6, Supplementary Tables 1 and 2 and Supplementary Methods (PDF 8157 kb)
Rights and permissions
About this article
Cite this article
Pérez-Dorado, I., González, A., Morales, M. et al. Insights into pneumococcal fratricide from the crystal structures of the modular killing factor LytC. Nat Struct Mol Biol 17, 576–581 (2010). https://doi.org/10.1038/nsmb.1817
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1817
This article is cited by
-
Antibacterial Activity of a Modified Choline Binding Peptide Against Streptococcus pneumoniae with Corresponding Antibody
International Journal of Peptide Research and Therapeutics (2021)
-
A choline binding polypeptide of LytA inhibits the growth of Streptococcus pneumoniae by binding to choline in the cell wall
The Journal of Antibiotics (2018)
-
Modular Architecture and Unique Teichoic Acid Recognition Features of Choline-Binding Protein L (CbpL) Contributing to Pneumococcal Pathogenesis
Scientific Reports (2016)
-
Evidence of involvement of the mannose receptor in the internalization of Streptococcus pneumoniae by Schwann cells
BMC Microbiology (2014)
-
Identification and characterization of an autolysin gene, atlA, from Streptococcus criceti
Journal of Microbiology (2012)