Key Points
-
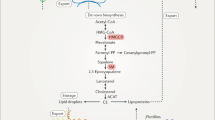
The physiological functions of cholesterol occur in cells, rather than in plasma where it can easily be measured. Therefore, plasma cholesterol levels reflect the exchange of cholesterol between different cell types and tissues. In cells, cholesterol functions as an essential structural membrane component, and the metabolites of cholesterol have important biological roles as signal transducers and solubilizers of other lipids.
-
There is a limited molecular understanding of the pathways and metabolic processes that are associated with cholesterol. Experimental approaches need to be improved to detect the short-range spatial and temporal order that lipids generate within membranes. This is important to validate the postulated models of cholesterol-dependent lateral membrane organization in cellular membranes.
-
Cholesterol and sterol metabolizing enzymes (which produce biologically active products) are distributed heterogeneously between intracellular membranes and different cell types. Sterol delivery between these sites is an important means of regulating these enzyme reactions, and thus cholesterol activity.
-
Lipid transfer proteins can facilitate the inter-membrane transport of cholesterol, possibly through membrane contact sites. START and ORP family proteins function as sterol transporters or sensors that indirectly take part in sterol trafficking.
-
Lipoprotein cholesterol is delivered to acidic hydrolytic organelles by membrane transport. This process is also important for the release of cholesterol to physiological extracellular acceptors and for lipoprotein cholesterol secretion. In many cell types, several ABC transporters cooperate to release cholesterol to physiological extracellular acceptors, thereby facilitating cholesterol excretion from the body.
-
Finally, cellular cholesterol compartmentalization largely dictates the exchange of cholesterol between tissues and determines cholesterol physiology and pathology at the whole body level.
Abstract
Cholesterol is an essential structural component in the cell membranes of most vertebrates. The biophysical properties of cholesterol and the enzymology of cholesterol metabolism provide the basis for how cells handle cholesterol and exchange it with one another. A tightly controlled — but only partially characterized — network of cellular signalling and lipid transfer systems orchestrates the functional compartmentalization of this lipid within and between organellar membranes. This largely dictates the exchange of cholesterol between tissues at the whole body level. Increased understanding of these processes and their integration at the organ systems level provides fundamental insights into the physiology of cholesterol trafficking.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Cherezov, V. et al. High-resolution crystal structure of an engineered human β2-adrenergic G protein-coupled receptor. Science 318, 1258–1265 (2007).
Ikonen, E. Mechanisms for cellular cholesterol transport: defects and human disease. Physiol. Rev. 86, 1237–1261 (2006).
Maxfield, F. R. & Tabas, I. Role of cholesterol and lipid organization in disease. Nature 438, 612–621 (2005).
Getz, G. S. & Reardon, C. A. Diet and murine atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 26, 242–249 (2006).
Hanai, J. I. et al. The muscle-specific ubiquitin ligase atrogin-1/MAFbx mediates statin-induced muscle toxicity. J. Clin. Invest. 117, 3940–3951 (2007).
Yang, F. et al. An ARC/Mediator subunit required for SREBP control of cholesterol and lipid homeostasis. Nature 442, 700–704 (2006). Identifies a nuclear interaction partner of SREBP, ARC105, and shows that it is a key effector of SREBP-dependent gene regulation in metazoans.
Takahashi, M. et al. Cholesterol controls lipid endocytosis through Rab11. Mol. Biol. Cell 18, 2667–2677 (2007).
Coxey, R. A., Pentchev, P. G., Campbell, G. & Blanchette-Mackie, E. J. Differential accumulation of cholesterol in Golgi compartments of normal and Niemann–Pick type C fibroblasts incubated with LDL: a cytochemical freeze-fracture study. J. Lipid Res. 34, 1165–1176 (1993).
Mukherjee, S., Zha, X., Tabas, I. & Maxfield, F. R. Cholesterol distribution in living cells: fluorescence imaging using dehydroergosterol as a fluorescent cholesterol analog. Biophys. J. 75, 1915–1925 (1998).
Lange, Y. Disposition of intracellular cholesterol in human fibroblasts. J. Lipid Res. 32, 329–339 (1991).
Goldstein, J. L., DeBose-Boyd, R. A. & Brown, M. S. Protein sensors for membrane sterols. Cell 124, 35–46 (2006).
Feng, B. et al. The endoplasmic reticulum is the site of cholesterol-induced cytotoxicity in macrophages. Nature Cell Biol. 5, 781–792 (2003).
Ridsdale, A. et al. Cholesterol is required for efficient endoplasmic reticulum-to-Golgi transport of secretory membrane proteins. Mol. Biol. Cell 17, 1593–1605 (2006).
Runz, H., Miura, K., Weiss, M. & Pepperkok, R. Sterols regulate ER-export dynamics of secretory cargo protein ts-O45-G. EMBO J. 25, 2953–2965 (2006).
Nohturfft, A., Brown, M. S. & Goldstein, J. L. Sterols regulate processing of carbohydrate chains of wild-type SREBP cleavage-activating protein (SCAP), but not sterol-resistant mutants Y298C or D443N. Proc. Natl Acad. Sci. USA 95, 12848–12853 (1998).
Sun, L. P., Li, L., Goldstein, J. L. & Brown, M. S. Insig required for sterol-mediated inhibition of Scap/SREBP binding to COPII proteins in vitro. J. Biol. Chem. 280, 26483–26490 (2005).
Miller, E., Antonny, B., Hamamoto, S. & Schekman, R. Cargo selection into COPII vesicles is driven by the Sec24p subunit. EMBO J. 21, 6105–6113 (2002).
Simons, K. & Vaz, W. L. Model systems, lipid rafts, and cell membranes. Annu. Rev. Biophys. Biomol. Struct. 33, 269–295 (2004).
Murata, M. et al. VIP21/caveolin is a cholesterol-binding protein. Proc. Natl Acad. Sci. USA 92, 10339–10343 (1995).
Radhakrishnan, A., Sun, L. P., Kwon, H. J., Brown, M. S. & Goldstein, J. L. Direct binding of cholesterol to the purified membrane region of SCAP: mechanism for a sterol-sensing domain. Mol. Cell 15, 259–268 (2004).
Olkkonen, V. M. et al. The OSBP-related proteins (ORPs): global sterol sensors for co-ordination of cellular lipid metabolism, membrane trafficking and signalling processes? Biochem. Soc. Trans. 34, 389–391 (2006).
Alpy, F. & Tomasetto, C. Give lipids a START: the StAR-related lipid transfer (START) domain in mammals. J. Cell Sci. 118, 2791–2801 (2005).
Li, H. & Papadopoulos, V. Peripheral-type benzodiazepine receptor function in cholesterol transport. Identification of a putative cholesterol recognition/interaction amino acid sequence and consensus pattern. Endocrinology 139, 4991–4997 (1998).
Parton, R. G., Hanzal-Bayer, M. & Hancock, J. F. Biogenesis of caveolae: a structural model for caveolin-induced domain formation. J. Cell Sci. 119, 787–796 (2006).
Lee, A. G. How lipids affect the activities of integral membrane proteins. Biochim. Biophys. Acta 1666, 62–87 (2004).
Huang, J. & Feigenson, G. W. A microscopic interaction model of maximum solubility of cholesterol in lipid bilayers. Biophys. J. 76, 2142–2157 (1999).
McConnell, H. M. & Radhakrishnan, A. Condensed complexes of cholesterol and phospholipids. Biochim. Biophys. Acta 1610, 159–173 (2003).
Jacobson, K., Mouritsen, O. G. & Anderson, R. G. Lipid rafts: at a crossroad between cell biology and physics. Nature Cell Biol. 9, 7–14 (2007).
Simons, K. & Ikonen, E. Functional rafts in cell membranes. Nature 387, 569–572 (1997).
Balasubramanian, N., Scott, D. W., Castle, J. D., Casanova, J. E. & Schwartz, M. A. Arf6 and microtubules in adhesion-dependent trafficking of lipid rafts. Nature Cell Biol. 9, 1381–1391 (2007).
Viola, A. & Gupta, N. Tether and trap: regulation of membrane-raft dynamics by actin-binding proteins. Nature Rev. Immunol. 7, 889–896 (2007).
Anderson, R. G. & Jacobson, K. A role for lipid shells in targeting proteins to caveolae, rafts, and other lipid domains. Science 296, 1821–1825 (2002).
Brown, D. A. Lipid rafts, detergent-resistant membranes, and raft targeting signals. Physiology 21, 430–439 (2006).
Kenworthy, A. K. et al. Dynamics of putative raft-associated proteins at the cell surface. J. Cell Biol. 165, 735–746 (2004).
Nakada, C. et al. Accumulation of anchored proteins forms membrane diffusion barriers during neuronal polarization. Nature Cell Biol. 5, 626–632 (2003).
Sharma, P. et al. Nanoscale organization of multiple GPI-anchored proteins in living cell membranes. Cell 116, 577–589 (2004).
Ortegren, U. et al. Lipids and glycosphingolipids in caveolae and surrounding plasma membrane of primary rat adipocytes. Eur. J. Biochem. 271, 2028–2036 (2004).
Wustner, D. Plasma membrane sterol distribution resembles the surface topography of living cells. Mol. Biol. Cell 18, 211–228 (2007).
Pelkmans, L. & Zerial, M. Kinase-regulated quantal assemblies and kiss-and-run recycling of caveolae. Nature 436, 128–133 (2005).
Steck, T. L., Ye, J. & Lange, Y. Probing red cell membrane cholesterol movement with cyclodextrin. Biophys. J. 83, 2118–2125 (2002).
Maxfield, F. R. & Menon, A. K. Intracellular sterol transport and distribution. Curr. Opin. Cell Biol. 18, 379–385 (2006).
Grundy, S. M. Absorption and metabolism of dietary cholesterol. Annu. Rev. Nutr. 3, 71–96 (1983).
Lange, Y., Echevarria, F. & Steck, T. L. Movement of zymosterol, a precursor of cholesterol, among three membranes in human fibroblasts. J. Biol. Chem. 266, 21439–21443 (1991).
Lusa, S., Heino, S. & Ikonen, E. Differential mobilization of newly synthesized cholesterol and biosynthetic sterol precursors from cells. J. Biol. Chem. 278, 19844–19851 (2003).
Yamauchi, Y. et al. Plasma membrane rafts complete cholesterol synthesis by participating in retrograde movement of precursor sterols. J. Biol. Chem. 282, 34994–35004 (2007).
Relas, H., Gylling, H. & Miettinen, T. A. Dietary squalene increases cholesterol synthesis measured with serum non-cholesterol sterols after a single oral dose in humans. Atherosclerosis 152, 377–383 (2000).
Debeljak, N., Fink, M. & Rozman, D. Many facets of mammalian lanosterol 14α-demethylase from the evolutionarily conserved cytochrome P450 family CYP51. Arch. Biochem. Biophys. 409, 159–171 (2003).
Tontonoz, P. & Mangelsdorf, D. J. Liver X receptor signaling pathways in cardiovascular disease. Mol. Endocrinol. 17, 985–993 (2003).
Yan, D. et al. OSBP-related protein 8 (ORP8) suppresses ABCA1 expression and cholesterol efflux from macrophages. J. Biol. Chem. 283, 332–340 (2008).
Radhakrishnan, A., Ikeda, Y., Kwon, H. J., Brown, M. S. & Goldstein, J. L. Sterol-regulated transport of SREBPs from endoplasmic reticulum to Golgi: oxysterols block transport by binding to Insig. Proc. Natl Acad. Sci. USA 104, 6511–6518 (2007). This work defines INSIGs as oxysterol binding proteins, thereby providing an explanation for the long-known ability of oxysterols to inhibit cholesterol synthesis in animal cells.
Sun, L. P., Seemann, J., Goldstein, J. L. & Brown, M. S. Sterol-regulated transport of SREBPs from endoplasmic reticulum to Golgi: Insig renders sorting signal in Scap inaccessible to COPII proteins. Proc. Natl Acad. Sci. USA 104, 6519–6526 (2007).
Song, B. L., Javitt, N. B. & DeBose-Boyd, R. A. Insig-mediated degradation of HMG CoA reductase stimulated by lanosterol, an intermediate in the synthesis of cholesterol. Cell Metab. 1, 179–189 (2005).
Soccio, R. E. & Breslow, J. L. Intracellular cholesterol transport. Arterioscler. Thromb. Vasc. Biol. 24, 1150–1160 (2004).
Dupree, P., Parton, R. G., Raposo, G., Kurzchalia, T. V. & Simons, K. Caveolae and sorting in the trans-Golgi network of epithelial cells. EMBO J. 12, 1597–1605 (1993).
Mobius, W. et al. Recycling compartments and the internal vesicles of multivesicular bodies harbor most of the cholesterol found in the endocytic pathway. Traffic 4, 222–231 (2003).
Hanada, K. et al. Molecular machinery for non-vesicular trafficking of ceramide. Nature 426, 803–809 (2003).
Tsujishita, Y. & Hurley, J. H. Structure and lipid transport mechanism of a StAR-related domain. Nature Struct. Biol. 7, 408–414 (2000).
Dawson, P. A., Van der Westhuyzen, D. R., Goldstein, J. L. & Brown, M. S. Purification of oxysterol binding protein from hamster liver cytosol. J. Biol. Chem. 264, 9046–9052 (1989).
Taylor, F. R., Saucier, S. E., Shown, E. P., Parish, E. J. & Kandutsch, A. A. Correlation between oxysterol binding to a cytosolic binding protein and potency in the repression of hydroxymethylglutaryl coenzyme A reductase. J. Biol. Chem. 259, 12382–12387 (1984).
Perry, R. J. & Ridgway, N. D. Oxysterol-binding protein and vesicle-associated membrane protein-associated protein are required for sterol-dependent activation of the ceramide transport protein. Mol. Biol. Cell 17, 2604–2616 (2006).
Raychaudhuri, S., Im, Y. J., Hurley, J. H. & Prinz, W. A. Nonvesicular sterol movement from plasma membrane to ER requires oxysterol-binding protein-related proteins and phosphoinositides. J. Cell Biol. 173, 107–119 (2006). Provides evidence that proteins related to oxysterol-binding proteins move sterols among cellular compartments and that sterol transport and distribution are regulated by phosphoinositides.
Fairn, G. D. & McMaster, C. R. Emerging roles of the oxysterol-binding protein family in metabolism, transport, and signaling. Cell. Mol. Life. Sci. 16 Oct 2007 (doi:10.1007/s00018-007-7325-2).
Schulz, T. A. & Prinz, W. A. Sterol transport in yeast and the oxysterol binding protein homologue (OSH) family. Biochim. Biophys. Acta 1771, 769–780 (2007).
Baumann, N. A. et al. Transport of newly synthesized sterol to the sterol-enriched plasma membrane occurs via nonvesicular equilibration. Biochemistry 44, 5816–5826 (2005).
Heino, S. et al. Dissecting the role of the Golgi complex and lipid rafts in biosynthetic transport of cholesterol to the cell surface. Proc. Natl Acad. Sci. USA 97, 8375–8380 (2000).
Cruz, J. C. & Chang, T. Y. Fate of endogenously synthesized cholesterol in Niemann–Pick type C1 cells. J. Biol. Chem. 275, 41309–41316 (2000).
Sullivan, D. P., Ohvo-Rekila, H., Baumann, N. A., Beh, C. T. & Menon, A. K. Sterol trafficking between the endoplasmic reticulum and plasma membrane in yeast. Biochem. Soc. Trans. 34, 356–358 (2006).
Hynynen, R. et al. Overexpression of OSBP-related protein 2 (ORP2) induces changes in cellular cholesterol metabolism and enhances endocytosis. Biochem. J. 390, 273–283 (2005).
Martin, S. & Parton, R. G. Lipid droplets: a unified view of a dynamic organelle. Nature Rev. Mol. Cell. Biol. 7, 373–378 (2006).
Ploegh, H. L. A lipid-based model for the creation of an escape hatch from the endoplasmic reticulum. Nature 448, 435–438 (2007).
Robenek, H. et al. Adipophilin-enriched domains in the ER membrane are sites of lipid droplet biogenesis. J. Cell Sci. 119, 4215–4224 (2006).
Prattes, S. et al. Intracellular distribution and mobilization of unesterified cholesterol in adipocytes: triglyceride droplets are surrounded by cholesterol-rich ER-like surface layer structures. J. Cell Sci. 113, 2977–2989 (2000).
Parton, R. G. & Simons, K. The multiple faces of caveolae. Nature Rev. Mol. Cell. Biol. 8, 185–194 (2007).
Le Lay, S. et al. Cholesterol-induced caveolin targeting to lipid droplets in adipocytes: a role for caveolar endocytosis. Traffic 7, 549–561 (2006).
Ost, A., Ortegren, U., Gustavsson, J., Nystrom, F. H. & Stralfors, P. Triacylglycerol is synthesized in a specific subclass of caveolae in primary adipocytes. J. Biol. Chem. 280, 5–8 (2005).
Buchmann, J. et al. Ablation of the cholesterol transporter adenosine triphosphate-binding cassette transporter G1 reduces adipose cell size and protects against diet-induced obesity. Endocrinology 148, 1561–1573 (2007). In this study, an unexpected role for the cholesterol transport protein ABCG1 in the regulation of energy balance and triglyceride storage was unravelled.
Miller, W. L. Steroidogenic acute regulatory protein (StAR), a novel mitochondrial cholesterol transporter. Biochim. Biophys. Acta 1771, 663–676 (2007).
Bose, H., Lingappa, V. R. & Miller, W. L. Rapid regulation of steroidogenesis by mitochondrial protein import. Nature 417, 87–91 (2002).
Tuckey, R. C., Bose, H. S., Czerwionka, I. & Miller, W. L. Molten globule structure and steroidogenic activity of N-218 MLN64 in human placental mitochondria. Endocrinology 145, 1700–1707 (2004).
Murcia, M., Faraldo-Gomez, J. D., Maxfield, F. R. & Roux, B. Modeling the structure of the StART domains of MLN64 and StAR proteins in complex with cholesterol. J. Lipid Res. 47, 2614–2630 (2006).
Liu, J., Rone, M. B. & Papadopoulos, V. Protein–protein interactions mediate mitochondrial cholesterol transport and steroid biosynthesis. J. Biol. Chem. 281, 38879–38893 (2006).
Hauet, T. et al. Peripheral-type benzodiazepine receptor-mediated action of steroidogenic acute regulatory protein on cholesterol entry into Leydig cell mitochondria. Mol. Endocrinol. 19, 540–554 (2005).
Papadopoulos, V., Liu, J. & Culty, M. Is there a mitochondrial signaling complex facilitating cholesterol import? Mol. Cell. Endocrinol. 265–266, 59–64 (2007).
Kolter, T. & Sandhoff, K. Principles of lysosomal membrane digestion: stimulation of sphingolipid degradation by sphingolipid activator proteins and anionic lysosomal lipids. Annu. Rev. Cell. Dev. Biol. 21, 81–103 (2005).
Kobayashi, T. et al. Late endosomal membranes rich in lysobisphosphatidic acid regulate cholesterol transport. Nature Cell Biol. 1, 113–118 (1999).
Matsuo, H. et al. Role of LBPA and Alix in multivesicular liposome formation and endosome organization. Science 303, 531–534 (2004).
Le Blanc, I. et al. Endosome-to-cytosol transport of viral nucleocapsids. Nature Cell Biol. 7, 653–664 (2005).
Sturley, S. L., Patterson, M. C., Balch, W. & Liscum, L. The pathophysiology and mechanisms of NP-C disease. Biochim. Biophys. Acta 1685, 83–87 (2004).
Sleat, D. E. et al. Genetic evidence for nonredundant functional cooperativity between NPC1 and NPC2 in lipid transport. Proc. Natl Acad. Sci. USA 101, 5886–5891 (2004).
Ohgami, N. et al. Binding between the Niemann–Pick C1 protein and a photoactivatable cholesterol analog requires a functional sterol-sensing domain. Proc. Natl Acad. Sci. USA 101, 12473–12478 (2004).
Xu, S., Benoff, B., Liou, H. L., Lobel, P. & Stock, A. M. Structural basis of sterol binding by NPC2, a lysosomal protein deficient in Niemann–Pick type C2 disease. J. Biol. Chem. 282, 23525–23531 (2007).
Koivusalo, M., Jansen, M., Somerharju, P. & Ikonen, E. Endocytic trafficking of sphingomyelin depends on its acyl chain length. Mol. Biol. Cell. 18, 5113–5123 (2007). This study shows that NPC1 selectively affects the endocytic recycling of long-chain sphingomyelins but not short-chain sphingomyelins.
Malathi, K. et al. Mutagenesis of the putative sterol-sensing domain of yeast Niemann Pick C-related protein reveals a primordial role in subcellular sphingolipid distribution. J. Cell Biol. 164, 547–556 (2004).
Schrantz, N. et al. The Niemann–Pick type C2 protein loads isoglobotrihexosylceramide onto CD1d molecules and contributes to the thymic selection of NKT cells. J. Exp. Med. 204, 841–852 (2007). Shows that NPC2 also functions as a glycolipid transporter that loads the T-cell surface antigen CD1d with lipid and contributes to the selection of the T-cell repertoire.
Cheruku, S. R., Xu, Z., Dutia, R., Lobel, P. & Storch, J. Mechanism of cholesterol transfer from the Niemann–Pick type C2 protein to model membranes supports a role in lysosomal cholesterol transport. J. Biol. Chem. 281, 31594–31604 (2006).
Davies, J. P. & Ioannou, Y. A. Topological analysis of Niemann–Pick C1 protein reveals that the membrane orientation of the putative sterol-sensing domain is identical to those of 3-hydroxy-3-methylglutaryl-CoA reductase and sterol regulatory element binding protein cleavage-activating protein. J. Biol. Chem. 275, 24367–24374 (2000).
Infante, R. E. et al. Purified NPC1 protein: I. Binding of cholesterol and oxysterols to a 1278-amino acid membrane protein. J. Biol. Chem. 283, 1052–1063 (2008).
Infante, R. E. et al. Purified NPC1 protein: II. Localization of sterol binding to a 240-amino acid soluble luminal loop. J. Biol. Chem. 283, 1064–1075 (2008).
Suchanek, M. et al. The mammalian oxysterol-binding protein-related proteins (ORPs) bind 25-hydroxycholesterol in an evolutionarily conserved pocket. Biochem. J. 405, 473–480 (2007).
Holtta-Vuori, M. et al. MLN64 is involved in actin-mediated dynamics of late endocytic organelles. Mol. Biol. Cell 16, 3873–3886 (2005).
Johansson, M. et al. Activation of endosomal dynein motors by stepwise assembly of Rab7-RILP-p150Glued, ORP1L, and the receptor βlll spectrin. J. Cell Biol. 176, 459–471 (2007).
Gagescu, R. et al. The recycling endosome of Madin–Darby canine kidney cells is a mildly acidic compartment rich in raft components. Mol. Biol. Cell 11, 2775–2791 (2000).
Wustner, D., Mondal, M., Tabas, I. & Maxfield, F. R. Direct observation of rapid internalization and intracellular transport of sterol by macrophage foam cells. Traffic 6, 396–412 (2005).
Holtta-Vuori, M., Tanhuanpaa, K., Mobius, W., Somerharju, P. & Ikonen, E. Modulation of cellular cholesterol transport and homeostasis by Rab11. Mol. Biol. Cell 13, 3107–3122 (2002).
Choudhury, A., Sharma, D. K., Marks, D. L. & Pagano, R. E. Elevated endosomal cholesterol levels in Niemann–Pick cells inhibit Rab4 and perturb membrane recycling. Mol. Biol. Cell 15, 4500–4511 (2004).
Ganley, I. G. & Pfeffer, S. R. Cholesterol accumulation sequesters Rab9 and disrupts late endosome function in NPC1-deficient cells. J. Biol. Chem. 281, 17890–17899 (2006).
Lebrand, C. et al. Late endosome motility depends on lipids via the small GTPase Rab7. EMBO J. 21, 1289–1300 (2002).
Choudhury, A. et al. Rab proteins mediate Golgi transport of caveola-internalized glycosphingolipids and correct lipid trafficking in Niemann–Pick C cells. J. Clin. Invest. 109, 1541–1550 (2002).
Zhao, B. et al. Macrophage-specific transgenic expression of cholesteryl ester hydrolase significantly reduces atherosclerosis and lesion necrosis in Ldlr mice. J. Clin. Invest. 117, 2983–2992 (2007). This work shows an important role for cholesteryl ester hydrolase activity in regulating macrophage cholesterol efflux and reverse cholesterol transport in vivo.
Oram, J. F. & Vaughan, A. M. ATP-binding cassette cholesterol transporters and cardiovascular disease. Circ. Res. 99, 1031–1043 (2006).
Brunham, L. R. et al. Intestinal ABCA1 directly contributes to HDL biogenesis in vivo. J. Clin. Invest. 116, 1052–1062 (2006).
Hajj Hassan, H. et al. Identification of an ABCA1-dependent phospholipid-rich plasma membrane apolipoprotein A-I binding site for nascent HDL formation: implications for current models of HDL biogenesis. J. Lipid Res. 48, 2428–2442 (2007).
Vedhachalam, C. et al. Mechanism of ATP-binding cassette transporter AI (ABCA1)-mediated cellular lipid efflux to apolipoprotein A-I and formation of high density lipoprotein particles. J. Biol. Chem. 282, 25123–25130 (2007).
Bared, S. M. et al. Association of ABCA1 with syntaxin 13 and flotillin-1 and enhanced phagocytosis in Tangier cells. Mol. Biol. Cell 15, 5399–5407 (2004).
Mendez, A. J., Lin, G., Wade, D. P., Lawn, R. M. & Oram, J. F. Membrane lipid domains distinct from cholesterol/sphingomyelin-rich rafts are involved in the ABCA1-mediated lipid secretory pathway. J. Biol. Chem. 276, 3158–3166 (2001).
Landry, Y. D. et al. ATP-binding cassette transporter A1 expression disrupts raft membrane microdomains through its ATPase-related functions. J. Biol. Chem. 281, 36091–36101 (2006).
Frank, P. G. et al. Caveolin-1 and regulation of cellular cholesterol homeostasis. Am. J. Physiol. Heart Circ. Physiol. 291, H677–H686 (2006).
Choi, H. Y. et al. Impaired ABCA1-dependent lipid efflux and hypoalphalipoproteinemia in human Niemann–Pick type C disease. J. Biol. Chem. 278, 32569–32577 (2003).
Haidar, B. et al. Cathepsin D, a lysosomal protease, regulates ABCA1-mediated lipid efflux. J. Biol. Chem. 281, 39971–39981 (2006).
Linder, M. D. et al. Rab8-dependent recycling promotes endosomal cholesterol removal in normal and sphingolipidosis cells. Mol. Biol. Cell 18, 47–56 (2007).
Baldan, A. et al. Impaired development of atherosclerosis in hyperlipidemic Ldlr−/− and ApoE−/− mice transplanted with Abcg1−/− bone marrow. Arterioscler. Thromb. Vasc. Biol. 26, 2301–2307 (2006).
Out, R. et al. Macrophage ABCG1 deletion disrupts lipid homeostasis in alveolar macrophages and moderately influences atherosclerotic lesion development in LDL receptor-deficient mice. Arterioscler. Thromb. Vasc. Biol. 26, 2295–2300 (2006).
Ranalletta, M. et al. Decreased atherosclerosis in low-density lipoprotein receptor knockout mice transplanted with Abcg1−/− bone marrow. Arterioscler. Thromb. Vasc. Biol. 26, 2308–2315 (2006).
Gelissen, I. C. et al. ABCA1 and ABCG1 synergize to mediate cholesterol export to apoA-I. Arterioscler. Thromb. Vasc. Biol. 26, 534–540 (2006).
Vaughan, A. M. & Oram, J. F. ABCA1 and ABCG1 or ABCG4 act sequentially to remove cellular cholesterol and generate cholesterol-rich HDL. J. Lipid Res. 47, 2433–2443 (2006).
Out, R. et al. Coexistence of foam cells and hypocholesterolemia in mice lacking the ABC transporters A1 and G1. Circ. Res. 102, 113–120 (2008). This paper, together with reference 128, provides evidence that the combined effects of ABCA1 and ABCG1 are crucial for macrophage cholesterol efflux and reverse cholesterol transport in vivo.
Yvan-Charvet, L. et al. Combined deficiency of ABCA1 and ABCG1 promotes foam cell accumulation and accelerates atherosclerosis in mice. J. Clin. Invest. 117, 3900–3908 (2007).
Wang, X. et al. Macrophage ABCA1 and ABCG1, but not SR-BI, promote macrophage reverse cholesterol transport in vivo. J. Clin. Invest. 117, 2216–2224 (2007). This paper, together with reference 126, provides evidence that the combined effects of ABCA1 and ABCG1 are crucial for macrophage cholesterol efflux and reverse cholesterol transport in vivo.
Yvan-Charvet, L. et al. SR-BI inhibits ABCG1-stimulated net cholesterol efflux from cells to plasma HDL. J. Lipid Res. 49, 107–114 (2008).
Wang, D. Q. Regulation of intestinal cholesterol absorption. Annu. Rev. Physiol. 69, 221–248 (2007).
Altmann, S. W. et al. Niemann–Pick C1 like 1 protein is critical for intestinal cholesterol absorption. Science 303, 1201–1204 (2004).
Voght, S. P., Fluegel, M. L., Andrews, L. A. & Pallanck, L. J. Drosophila NPC1b promotes an early step in sterol absorption from the midgut epithelium. Cell Metab. 5, 195–205 (2007).
Dixit, S. S., Sleat, D. E., Stock, A. M. & Lobel, P. Do mammalian NPC1 and NPC2 play a role in intestinal cholesterol absorption? Biochem. J. 408, 1–5 (2007).
Shoulders, C. C., Stephens, D. J. & Jones, B. The intracellular transport of chylomicrons requires the small GTPase, Sar1b. Curr. Opin. Lipidol. 15, 191–197 (2004).
Rava, P., Ojakian, G. K., Shelness, G. S. & Hussain, M. M. Phospholipid transfer activity of microsomal triacylglycerol transfer protein is sufficient for the assembly and secretion of apolipoprotein B lipoproteins. J. Biol. Chem. 281, 11019–11027 (2006).
Fisher, E. A. & Ginsberg, H. N. Complexity in the secretory pathway: the assembly and secretion of apolipoprotein B-containing lipoproteins. J. Biol. Chem. 277, 17377–17380 (2002).
Adiels, M., Olofsson, S. O., Taskinen, M. R. & Boren, J. Diabetic dyslipidaemia. Curr. Opin. Lipidol. 17, 238–246 (2006).
Asp, L., Claesson, C., Boren, J. & Olofsson, S. O. ADP-ribosylation factor 1 and its activation of phospholipase D are important for the assembly of very low density lipoproteins. J. Biol. Chem. 275, 26285–26292 (2000).
Kulinski, A. & Vance, J. E. Lipid homeostasis and lipoprotein secretion in Niemann–Pick C1-deficient hepatocytes. J. Biol. Chem. 282, 1627–1637 (2007).
Twisk, J. et al. The role of the LDL receptor in apolipoprotein B secretion. J. Clin. Invest. 105, 521–532 (2000).
Maric, J., Kiss, R. S., Franklin, V. & Marcel, Y. L. Intracellular lipidation of newly synthesized apolipoprotein A-I in primary murine hepatocytes. J. Biol. Chem. 280, 39942–39949 (2005).
Wang, M. D. et al. Differential regulation of ATP binding cassette protein A1 expression and ApoA-I lipidation by Niemann–Pick type C1 in murine hepatocytes and macrophages. J. Biol. Chem. 282, 22525–22533 (2007).
Xie, C., Turley, S. D. & Dietschy, J. M. Centripetal cholesterol flow from the extrahepatic organs through the liver is normal in mice with mutated Niemann–Pick type C protein (NPC1). J. Lipid Res. 41, 1278–1289 (2000).
Harder, C. J., Meng, A., Rippstein, P., McBride, H. M. & McPherson, R. SR-BI undergoes cholesterol-stimulated transcytosis to the bile canaliculus in polarized WIF-B cells. J. Biol. Chem. 282, 1445–1455 (2007).
Wustner, D., Mondal, M., Huang, A. & Maxfield, F. R. Different transport routes for high density lipoprotein and its associated free sterol in polarized hepatic cells. J. Lipid Res. 45, 427–437 (2004).
Martinez, L. O. et al. Ectopic β-chain of ATP synthase is an apolipoprotein A-I receptor in hepatic HDL endocytosis. Nature 421, 75–79 (2003).
Yu, L. et al. Disruption of Abcg5 and Abcg8 in mice reveals their crucial role in biliary cholesterol secretion. Proc. Natl Acad. Sci. USA 99, 16237–16242 (2002).
Temel, R. E. et al. Hepatic Niemann–Pick C1-like 1 regulates biliary cholesterol concentration and is a target of ezetimibe. J. Clin. Invest. 117, 1968–1978 (2007).
Karten, B., Campenot, R. B., Vance, D. E. & Vance, J. E. Expression of ABCG1, but not ABCA1, correlates with cholesterol release by cerebellar astroglia. J. Biol. Chem. 281, 4049–4057 (2006).
Kim, W. S. et al. Role of ABCG1 and ABCA1 in regulation of neuronal cholesterol efflux to apolipoprotein E discs and suppression of amyloid-β peptide generation. J. Biol. Chem. 282, 2851–2861 (2007).
Wang, N. et al. ATP-binding cassette transporters G1 and G4 mediate cholesterol and desmosterol efflux to HDL and regulate sterol accumulation in the brain. FASEB J. 26 Nov 2007 (doi:10.1096/fj.07-9944com).
Lund, E. G., Guileyardo, J. M. & Russell, D. W. cDNA cloning of cholesterol 24-hydroxylase, a mediator of cholesterol homeostasis in the brain. Proc. Natl Acad. Sci. USA 96, 7238–7243 (1999).
Dietschy, J. M. & Turley, S. D. Thematic review series: brain lipids. Cholesterol metabolism in the central nervous system during early development and in the mature animal. J. Lipid Res. 45, 1375–1397 (2004).
Lund, E. G. et al. Knockout of the cholesterol 24-hydroxylase gene in mice reveals a brain-specific mechanism of cholesterol turnover. J. Biol. Chem. 278, 22980–22988 (2003).
Goritz, C., Mauch, D. H. & Pfrieger, F. W. Multiple mechanisms mediate cholesterol-induced synaptogenesis in a CNS neuron. Mol. Cell. Neurosci. 29, 190–201 (2005).
Funfschilling, U., Saher, G., Xiao, L., Mobius, W. & Nave, K. A. Survival of adult neurons lacking cholesterol synthesis in vivo. BMC Neurosci. 8, 1 (2007).
Bloch, K. Sterol molecule: structure, biosynthesis, and function. Steroids 57, 378–383 (1992).
Kandutsch, A. A. & Russell, A. E. Preputial gland tumor sterols. 3. A metabolic pathway from lanosterol to cholesterol. J. Biol. Chem. 235, 2256–2261 (1960).
Ladinsky, M. S., Mastronarde, D. N., McIntosh, J. R., Howell, K. E. & Staehelin, L. A. Golgi structure in three dimensions: functional insights from the normal rat kidney cell. J. Cell Biol. 144, 1135–1149 (1999).
Staehelin, L. A. The plant ER: a dynamic organelle composed of a large number of discrete functional domains. Plant J. 11, 1151–1165 (1997).
Author information
Authors and Affiliations
Related links
Glossary
- Familial hypercholesterolaemia
-
The most common monogenic defect of cellular cholesterol processing, with the majority caused by low density lipoprotein (LDL) receptor mutations. It results in high circulating LDL–cholesterol levels and premature atherosclerosis.
- Lysosomal cholesterol–sphingolipid storage diseases
-
Monogenic diseases that are characterized by the accumulation of cholesterol and distinct sphingolipid species in late endocytic compartments, typically caused by defective degradation or endolysosomal export of the accumulating lipid(s), which manifest as neurodegenerative and visceral (liver, spleen) symptoms.
- Enterocyte
-
The absorptive cell type in the intestine.
- HDL
-
An ∼8–11-nm high density (1.063–1.210 g ml−1) lipoprotein with 55% protein (with APOA-I as the major apoprotein), 25% phospholipids, 15% cholesterol and 5% triglycerides.
- Lipoprotein
-
A spherical particle that is used for the transfer of lipids and lipophilic substances in the circulation. It is composed of various amounts of phospholipids, cholesterol and triglycerides as well as apoproteins.
- Statin
-
An inhibitor of hydroxymethylglutaryl CoA reductase (HMG-CoAR), the rate-limiting enzyme of cholesterol synthesis.
- Sterol regulatory element binding protein
-
A membrane-tethered transcription factor that controls certain genes that are required for lipid synthesis and uptake.
- COPII vesicle
-
A coated vesicle that is involved in transport from the endoplasmic reticulum to the Golgi.
- Caveolae
-
Flask-shaped invaginations of the plasma membrane with a diameter of 60–80 nm. Caveolae are formed by the polymerization of caveolins and contain cholesterol and sphingolipids.
- Flip-flop
-
Transmembrane translocation of lipids.
- Oxysterol
-
A family of oxidized derivatives of cholesterol, which may arise from either enzymatic or non-enzymatic oxidation reactions.
- Chylomicron
-
A ∼50–200-nm diameter, lowest density (<1.006 g ml−1) lipoprotein that is composed of ∼85% triglycerides, 9% phospholipids, 4% cholesterol, and 2% protein (with APOB48 as the major apoprotein).
- VLDL
-
A ∼30–70-nm very low density (0.95–1.006 g ml−1) lipoprotein, with ∼50% triglycerides, 20% cholesterol, 20% phospholipids and 10% protein (with APOB100 as the major apoprotein).
- LDL
-
A ∼20–25-nm low density (1.006–1.063 g ml−1) lipoprotein with ∼45% cholesterol, 20% phospholipids, 10% triglycerides and 25% protein (with APOB as the major apoprotein).
- Reverse cholesterol transport
-
The process by which cholesterol is transported by HDL from extrahepatic tissues to the liver for excretion into bile.
- ABC transporter
-
One of a family of membrane transport proteins that use the energy of ATP hydrolysis to transport various molecules, including cholesterol and other lipids, across the membrane.
- Clathrin-coated vesicle
-
A transport vesicle that buds with the aid of a coat protein known as clathrin.
- Multivesicular endosome
-
An intermediate organelle in endosome–lysosome transport that is characterized by small membrane vesicles enclosed within a limiting membrane. The internal vesicles are thought to form by invagination and budding from the limiting membrane.
- Rab
-
A family of Ras-like small G proteins that control membrane trafficking. Rab proteins are modified by geranylgeranyl groups that anchor them to membranes and can be specifically localized to different compartments.
- Atherosclerotic lesion
-
Deposition of cholesterol in the arterial wall, resulting in the invasion of monocyte–macrophages that scavenge the lipids and transform into foam cells. In advanced lesions, many cell types are affected and the lesion becomes prone to rupture, which may clog the artery.
- Discoidal HDL
-
A precursor for a mature, spherical high density lipoprotein (HDL) particle that migrates with pre-β mobility in crossed immunoelectrophoresis (therefore, it is also called pre-β-HDL). It contains APOA-I, phospholipid and small amounts of cholesterol.
- Stanol
-
A family of hydrated plant sterols.
- Canalicular membrane
-
The apical plasma membrane of the hepatocyte that faces the bile canaliculus.
Rights and permissions
About this article
Cite this article
Ikonen, E. Cellular cholesterol trafficking and compartmentalization. Nat Rev Mol Cell Biol 9, 125–138 (2008). https://doi.org/10.1038/nrm2336
Issue Date:
DOI: https://doi.org/10.1038/nrm2336
This article is cited by
-
Nonlinear relationship between untraditional lipid parameters and the risk of prediabetes: a large retrospective study based on Chinese adults
Cardiovascular Diabetology (2024)
-
HIF-2α/LINC02609/APOL1-mediated lipid storage promotes endoplasmic reticulum homeostasis and regulates tumor progression in clear-cell renal cell carcinoma
Journal of Experimental & Clinical Cancer Research (2024)
-
Highly sensitive electrochemical detection of cholesterol based on Au–Pt NPs/PAMAM–ZIF-67 nanomaterials
Analytical Sciences (2024)
-
Mutation in the TRKB Cholesterol Recognition Site that blocks Antidepressant Binding does not Influence the Basal or BDNF-Stimulated Activation of TRKB
Cellular and Molecular Neurobiology (2024)
-
Cholesterol-dependent amyloid β production: space for multifarious interactions between amyloid precursor protein, secretases, and cholesterol
Cell & Bioscience (2023)