Abstract
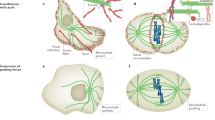
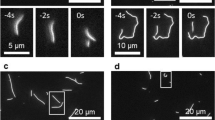
Recent experiments suggest that microtubules do not grow steadily but instead elongate at a rate that varies in time. We argue that this variation might arise from fluctuations in the length of a dynamic GTP–tubulin cap at the microtubule end. We propose that these fluctuations can lead to a switch in the dynamics of a microtubule end between growth and shrinkage, and provide insight into how the sensitivity of this switch can be changed by microtubule polymerases, such as XMAP215, and tensile forces, through the stabilization of initial contacts in the cap.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Desai, A. & Mitchison, T. J. Microtubule polymerization dynamics. Annu. Rev. Cell Dev. Biol. 13, 83–117 (1997).
Howard, J. & Hyman, A. A. Dynamics and mechanics of the microtubule plus end. Nature 422, 753–758 (2003).
Mitchison, T. & Kirschner, M. Dynamic instability of microtubule growth. Nature 312, 237–242 (1984).
Kirschner, M. & Mitchison, T. Beyond self-assembly: from microtubules to morphogenesis. Cell 45, 329–342 (1986).
Hayden, J. H., Bowser, S. S. & Rieder, C. L. Kinetochores capture astral microtubules during chromosome attachment to the mitotic spindle: direct visualization in live newt lung cells. J. Cell Biol. 111, 1039–1045 (1990).
Holy, T. E., Dogterom, M., Yurke, B. & Leibler, S. Assembly and positioning of microtubule asters in microfabricated chambers. Proc. Natl Acad. Sci. USA 94, 6228–6231 (1997).
Rodionov, V. I. & Borisy, G. G. Self-centring activity of cytoplasm. Nature 386, 170–173 (1997).
Howard, J. Elastic and damping forces generated by confined arrays of dynamic microtubules. Phys. Biol. 3, 54–66 (2006).
Tolić-Nørrelykke, I. M. Push-me-pull-you: how microtubules organize the cell interior. Eur. Biophys. J. 37, 1271–1278 (2008).
Nogales, E., Wolf, S. G. & Downing, K. H. Structure of the αβ tubulin dimer by electron crystallography. Nature 391, 199–203 (1998); erratum 393, 191 (1998).
Hyman, A. A., Salser, S., Drechsel, D. N., Unwin, N. & Mitchison, T. J. Role of GTP hydrolysis in microtubule dynamics: information from a slowly hydrolyzable analogue, GMPCPP. Mol. Biol. Cell 3, 1155–1167 (1992).
Drechsel, D. N. & Kirschner, M. W. The minimum GTP cap required to stabilize microtubules. Curr. Biol. 4, 1053–1061 (1994); erratum 5, 215 (1995).
Sloboda, R. D., Rudolph, S. A., Rosenbaum, J. L. & Greengard, P. Cyclic AMP-dependent endogenous phosphorylation of a microtubule-associated protein. Proc. Natl Acad. Sci. USA 72, 177–181 (1975).
Kinoshita, K., Habermann, B. & Hyman, A. A. XMAP215: a key component of the dynamic microtubule cytoskeleton. Trends Cell Biol. 12, 267–273 (2002).
Howard, J. & Hyman, A. A. Microtubule polymerases and depolymerases. Curr. Opin. Cell Biol. 19, 31–35 (2007).
Akhmanova, A. & Steinmetz, M. O. Tracking the ends: a dynamic protein network controls the fate of microtubule tips. Nature Rev. Mol. Cell Biol. 9, 309–322 (2008).
Bieling, P. et al. Reconstitution of a microtubule plus-end tracking system in vitro. Nature 450, 1100–1105 (2007).
Bieling, P. et al. CLIP-170 tracks growing microtubule ends by dynamically recognizing composite EB1/tubulin-binding sites. J. Cell Biol. 183, 1223–1233 (2008).
des Georges, A. et al. Mal3, the Schizosaccharomyces pombe homolog of EB1, changes the microtubule lattice. Nature Struct. Mol. Biol. 15, 1102–1108 (2008).
Vitre, B. et al. EB1 regulates microtubule dynamics and tubulin sheet closure in vitro. Nature Cell Biol. 10, 415–421 (2008).
Komarova, Y. et al. Mammalian end binding proteins control persistent microtubule growth. J. Cell Biol. 184, 691–706 (2009).
Grishchuk, E. L., Molodtsov, M. I., Ataullakhanov, F. I. & McIntosh, J. R. Force production by disassembling microtubules. Nature 438, 384–388 (2005).
Kerssemakers, J. W. et al. Assembly dynamics of microtubules at molecular resolution. Nature 442, 709–712 (2006).
Schek, H. T., Gardner, M. K., Cheng, J., Odde, D. J. & Hunt, A. J. Microtubule assembly dynamics at the nanoscale. Curr. Biol. 17, 1445–1455 (2007).
Asbury, C. L., Gestaut, D. R., Powers, A. F., Franck, A. D. & Davis, T. N. The Dam1 kinetochore complex harnesses microtubule dynamics to produce force and movement. Proc. Natl Acad. Sci. USA 103, 9873–9878 (2006).
Hill, T. L. & Kirschner, M. W. Bioenergetics and kinetics of microtubule and actin filament assembly–disassembly. Int. Rev. Cytol. 78, 1–125 (1982).
VanBuren, V., Cassimeris, L. & Odde, D. J. Mechanochemical model of microtubule structure and self-assembly kinetics. Biophys. J. 89, 2911–2926 (2005).
Mozziconacci, J., Sandblad, L., Wachsmuth, M., Brunner, D. & Karsenti, E. Tubulin dimers oligomerize before their incorporation into microtubules. PLoS ONE 3, e3821 (2008).
Chrétien, D., Fuller, S. D. & Karsenti, E. Structure of growing microtubule ends: two-dimensional sheets close into tubes at variable rates. J. Cell Biol. 129, 1311–1328 (1995).
Howard, J. Mechanics of Motor Proteins and the Cytoskeleton (Sinauer Associates, Sunderland, Massachusetts, 2001).
Müller-Reichert, T., Chrétien, D., Severin, F. & Hyman, A. A. Structural changes at microtubule ends accompanying GTP hydrolysis: information from a slowly hydrolyzable analogue of GTP, guanylyl (α,β) methylenediphosphonate. Proc. Natl Acad. Sci. USA 95, 3661–3666 (1998).
Wang, H. W. & Nogales, E. Nucleotide-dependent bending flexibility of tubulin regulates microtubule assembly. Nature 435, 911–915 (2005).
Elie-Caille, C. et al. Straight GDP–tubulin protofilaments form in the presence of taxol. Curr. Biol. 17, 1765–1770 (2007).
Mandelkow, E. M., Mandelkow, E. & Milligan, R. A. Microtubule dynamics and microtubule caps: a time-resolved cryo-electron microscopy study. J. Cell Biol. 114, 977–991 (1991).
Ravelli, R. B. et al. Insight into tubulin regulation from a complex with colchicine and a stathmin-like domain. Nature 428, 198–202 (2004).
Rice, L. M., Montabana, E. A. & Agard, D. A. The lattice as allosteric effector: structural studies of αβ- and γ-tubulin clarify the role of GTP in microtubule assembly. Proc. Natl Acad. Sci. USA 105, 5378–5383 (2008).
Wang, H. W. & Nogales, E. Nucleotide-dependent bending flexibility of tubulin regulates microtubule assembly. Nature 435, 911–915 (2005).
Nogales, E., Whittaker, M., Milligan, R. A. & Downing, K. H. High-resolution model of the microtubule. Cell 96, 79–88 (1999).
Mickey, B. & Howard, J. Rigidity of microtubules is increased by stabilizing agents. J. Cell Biol. 130, 909–917 (1995).
Gard, D. L. & Kirschner, M. W. A microtubule-associated protein from Xenopus eggs that specifically promotes assembly at the plus-end. J. Cell Biol. 105, 2203–2215 (1987).
Tournebize, R. et al. Control of microtubule dynamics by the antagonistic activities of XMAP215 and XKCM1 in Xenopus egg extracts. Nature Cell Biol. 2, 13–19 (2000).
Cassimeris, L., Gard, D., Tran, P. T. & Erickson, H. P. XMAP215 is a long thin molecule that does not increase microtubule stiffness. J. Cell Sci. 114, 3025–3033 (2001).
Al-Bassam, J., Larsen, N. A., Hyman, A. A. & Harrison, S. C. Crystal structure of a TOG domain: conserved features of XMAP215/Dis1-family TOG domains and implications for tubulin binding. Structure 15, 355–362 (2007).
Slep, K. C. & Vale, R. D. Structural basis of microtubule plus end tracking by XMAP215, CLIP-170, and EB1. Mol. Cell 27, 976–991 (2007).
Brouhard, G. J. et al. XMAP215 is a processive microtubule polymerase. Cell 132, 79–88 (2008).
Gard, D. L. & Kirschner, M. W. Microtubule assembly in cytoplasmic extracts of Xenopus oocytes and eggs. J. Cell Biol. 105, 2191–2201 (1987).
Arnal, I. & Wade, R. H. How does taxol stabilize microtubules? Curr. Biol. 5, 900–908 (1995).
Tyson, J. J., Chen, K. C. & Novak, B. Sniffers, buzzers, toggles and blinkers: dynamics of regulatory and signaling pathways in the cell. Curr. Opin. Cell Biol. 15, 221–231 (2003).
Alon, U. An Introduction to Systems Biology (Chapman & Hall/CRC, Boca Raton, Florida, 2007).
Ferrell, J. E. & Xiong, W. Bistability in cell signaling: how to make continuous processes discontinuous, and reversible processes irreversible. Chaos 11, 227–236 (2001).
Franck, A. D. et al. Tension applied through the Dam1 complex promotes microtubule elongation providing a direct mechanism for length control in mitosis. Nature Cell Biol. 9, 832–837 (2007).
McIntosh, J. R. et al. Fibrils connect microtubule tips with kinetochores: a mechanism to couple tubulin dynamics to chromosome motion. Cell 135, 322–333 (2008).
Chretien, D., Fuller, S. D. & Karsenti, E. Structure of growing microtubule ends: two-dimensional sheets close into tubes at variable rates. J. Cell Biol. 129, 1311–1328 (1995).
Sandblad, L. et al. The Schizosaccharomyces pombe EB1 homolog Mal3p binds and stabilizes the microtubule lattice seam. Cell 127, 1415–1424 (2006).
Dimitrov, A. et al. Detection of GTP-tubulin conformation in vivo reveals a role for GTP remnants in microtubule rescues. Science 322, 1353–1356 (2008).
Acknowledgements
We thank G. Brouhard, J. Stear, H. Erickson, V. Varga and M. Zanic for their comments on the manuscript, as well as M. Gardner, A. Hunt and D. Odde for figure 2.
Author information
Authors and Affiliations
Related links
Rights and permissions
About this article
Cite this article
Howard, J., Hyman, A. Growth, fluctuation and switching at microtubule plus ends. Nat Rev Mol Cell Biol 10, 569–574 (2009). https://doi.org/10.1038/nrm2713
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm2713
This article is cited by
-
Molecular basis of vasohibins-mediated detyrosination and its impact on spindle function and mitosis
Cell Research (2019)
-
Microtubule dynamics: an interplay of biochemistry and mechanics
Nature Reviews Molecular Cell Biology (2018)
-
Regulation of long-distance transport of mitochondria along microtubules
Cellular and Molecular Life Sciences (2018)
-
The nano-architecture of the axonal cytoskeleton
Nature Reviews Neuroscience (2017)
-
Alpha-synuclein facilitates to form short unconventional microtubules that have a unique function in the axonal transport
Scientific Reports (2017)