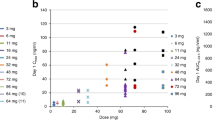
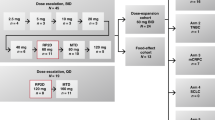
Key Points
-
Metronomic chemotherapy is the frequent, regular administration of drug doses designed to maintain low, but active, concentrations of chemotherapeutic drugs over prolonged periods of time, without causing serious toxicities
-
Despite the important information they provide, preclinical and clinical pharmacokinetic studies have had a secondary role during the conceptual development of metronomic chemotherapy
-
Different metronomic drug concentrations and schedules might affect different prevalent mechanisms of antitumour action, which suggests that therapy protocols could be selected on the basis of different prevalent effects
-
To develop better computational models for future metronomic chemotherapy studies, pharmacokinetic parameters of metronomic chemotherapy should be investigated more extensively in relation to pharmacodynamics (with a PK/PD approach) in future clinical trials
-
Therapeutic drug monitoring of metronomic chemotherapy is essential to maintain drug concentrations in the 'activity range', while maintaining a low toxicity profile
-
Randomized, prospective, clinical studies on metronomic chemotherapy should include pharmacokinetic/pharmacodynamic substudies with the aim of achieving personalized metronomic chemotherapy protocols in the future
Abstract
Metronomic chemotherapy describes the close, regular administration of chemotherapy drugs at less-toxic doses over prolonged periods of time. In 2015, the results of randomized phase III clinical trials demonstrated encouraging, albeit limited, efficacy benefits of metronomic chemotherapy regimens administered as adjuvant maintenance therapy for the treatment of breast cancer, or as maintenance therapy in combination with an antiangiogenic agent for metastatic colorectal cancer. Owing to the investigational nature of this approach, metronomic chemotherapy regimens are highly empirical in terms of the optimal dose and schedule for the drugs administered; therefore, greater knowledge of the pharmacokinetics of metronomic chemotherapy is critical to the future success of this treatment strategy. Unfortunately, such preclinical and clinical pharmacokinetic studies are rare. Herein, we present situations in which active drug concentrations have been achieved with metronomic schedules, and discuss their associated pharmacokinetic parameters. We summarize examples from the limited number of clinical studies in order to illustrate the importance of assessing such pharmacokinetic parameters, and discuss the influence this information can have on improving efficacy and reducing toxicity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kerbel, R. S. & Kamen, B. A. The anti-angiogenic basis of metronomic chemotherapy. Nat. Rev. Cancer 4, 423–436 (2004).
Andre, N., Carre, M. & Pasquier, E. Metronomics: towards personalized chemotherapy? Nat. Rev. Clin. Oncol. 11, 413–431 (2014).
Munzone, E. & Colleoni, M. Clinical overview of metronomic chemotherapy in breast cancer. Nat. Rev. Clin. Oncol. 12, 631–644 (2015).
Browder, T. et al. Antiangiogenic scheduling of chemotherapy improves efficacy against experimental drug-resistant cancer. Cancer Res. 60, 1878–1886 (2000).
Klement, G. et al. Continuous low-dose therapy with vinblastine and VEGF receptor-2 antibody induces sustained tumor regression without overt toxicity. J. Clin. Invest. 105, R15–R24 (2000).
Bocci, G., Nicolaou, K. C. & Kerbel, R. S. Protracted low-dose effects on human endothelial cell proliferation and survival in vitro reveal a selective antiangiogenic window for various chemotherapeutic drugs. Cancer Res. 62, 6938–6943 (2002).
Hao, Y. B., Yi, S. Y., Ruan, J., Zhao, L. & Nan, K. J. New insights into metronomic chemotherapy-induced immunoregulation. Cancer Lett. 354, 220–226 (2014).
Folkins, C. et al. Anticancer therapies combining antiangiogenic and tumor cell cytotoxic effects reduce the tumor stem-like cell fraction in glioma xenograft tumors. Cancer Res. 67, 3560–3564 (2007).
Derosa, L. et al. Docetaxel plus oral metronomic cyclophosphamide: a phase II study with pharmacodynamic and pharmacogenetic analyses in castration-resistant prostate cancer patients. Cancer 120, 3923–3931 (2014).
Shaked, Y. et al. Low-dose metronomic combined with intermittent bolus-dose cyclophosphamide is an effective long-term chemotherapy treatment strategy. Cancer Res. 65, 7045–7051 (2005).
Jedeszko, C. et al. Postsurgical adjuvant or metastatic renal cell carcinoma therapy models reveal potent antitumor activity of metronomic oral topotecan with pazopanib. Sci. Transl. Med. 7, 282ra50 (2015).
Cruz-Munoz, W. et al. Analysis of acquired resistance to metronomic oral topotecan chemotherapy plus pazopanib after prolonged preclinical potent responsiveness in advanced ovarian cancer. Angiogenesis 17, 661–673 (2014).
Srivastava, K. et al. Postsurgical adjuvant tumor therapy by combining anti-angiopoietin-2 and metronomic chemotherapy limits metastatic growth. Cancer Cell 26, 880–895 (2014).
Dellapasqua, S. et al. Increased mean corpuscular volume of red blood cells predicts response to metronomic capecitabine and cyclophosphamide in combination with bevacizumab. Breast 21, 309–313 (2012).
Bocci, G. et al. Increased plasma vascular endothelial growth factor (VEGF) as a surrogate marker for optimal therapeutic dosing of VEGF receptor-2 monoclonal antibodies. Cancer Res. 64, 6616–6625 (2004).
Orlando, L. et al. Trastuzumab in combination with metronomic cyclophosphamide and methotrexate in patients with HER-2 positive metastatic breast cancer. BMC Cancer 6, 225 (2006).
Denies, S., Cicchelero, L., Van Audenhove, I. & Sanders, N. N. Combination of interleukin-12 gene therapy, metronomic cyclophosphamide and DNA cancer vaccination directs all arms of the immune system towards tumor eradication. J. Control. Release 187, 175–182 (2014).
Weir, G. M. et al. Metronomic cyclophosphamide enhances HPV16E7 peptide vaccine induced antigen-specific and cytotoxic T-cell mediated antitumor immune response. Oncoimmunology 3, e953407 (2014).
Bottini, A. et al. Randomized phase II trial of letrozole and letrozole plus low-dose metronomic oral cyclophosphamide as primary systemic treatment in elderly breast cancer patients. J. Clin. Oncol. 24, 3623–3628 (2006).
Emmenegger, U. et al. Pharmacodynamic and pharmacokinetic study of chronic low-dose metronomic cyclophosphamide therapy in mice. Mol. Cancer Ther. 6, 2280–2289 (2007).
Wang, Z., Butner, J. D., Cristini, V. & Deisboeck, T. S. Integrated PK-PD and agent-based modeling in oncology. J. Pharmacokinet. Pharmacodyn. 42, 179–189 (2015).
Penel, N. et al. Megestrol acetate versus metronomic cyclophosphamide in patients having exhausted all effective therapies under standard care. Br. J. Cancer 102, 1207–1212 (2010).
Patil, V. M. et al. A prospective randomized phase II study comparing metronomic chemotherapy with chemotherapy (single agent cisplatin), in patients with metastatic, relapsed or inoperable squamous cell carcinoma of head and neck. Oral Oncol. 51, 279–286 (2015).
Chen, Y. M. et al. A phase II randomized trial of gefitinib alone or with tegafur/uracil treatment in patients with pulmonary adenocarcinoma who had failed previous chemotherapy. J. Thorac. Oncol. 6, 1110–1116 (2011).
Simkens, L. H. et al. Maintenance treatment with capecitabine and bevacizumab in metastatic colorectal cancer (CAIRO3): a phase 3 randomised controlled trial of the Dutch Colorectal Cancer Group. Lancet 385, 1843–1852 (2015).
Rowland, M. & Tozer, T. N. in Clinical Pharmacokinetics: Concepts and Applications 53–105 (Lippincot Williams & Wilkins, 1995).
Gillis, N. K., Patel, J. N. & Innocenti, F. Clinical implementation of germ line cancer pharmacogenetic variants during the next-generation sequencing era. Clin. Pharmacol. Ther. 95, 269–280 (2014).
Roy, P. & Waxman, D. J. Activation of oxazaphosphorines by cytochrome P450: application to gene-directed enzyme prodrug therapy for cancer. Toxicol. In Vitro 20, 176–186 (2006).
Bocci, G. et al. A pharmacokinetic-based test to prevent severe 5-fluorouracil toxicity. Clin. Pharmacol. Ther. 80, 384–395 (2006).
Kerbel, R. S. & Grothey, A. Gastrointestinal cancer: rationale for metronomic chemotherapy in phase III trials. Nat. Rev. Clin. Oncol. 12, 313–314 (2015).
Fan, J. & de Lannoy, I. A. Pharmacokinetics. Biochem. Pharmacol. 87, 93–120 (2014).
Barbolosi, D. et al. Metronomics chemotherapy: time for computational decision support. Cancer Chemother. Pharmacol. 74, 647–652 (2014).
McCune, J. S., Jacobson, P., Wiseman, A. & Militano, O. Optimizing drug therapy in pediatric SCT: focus on pharmacokinetics. Bone Marrow Transplant. 50, 165–172 (2015).
Widmer, N. et al. Review of therapeutic drug monitoring of anticancer drugs part two — targeted therapies. Eur. J. Cancer 50, 2020–2036 (2014).
Zhou, Q., Guo, P., Wang, X., Nuthalapati, S. & Gallo, J. M. Preclinical pharmacokinetic and pharmacodynamic evaluation of metronomic and conventional temozolomide dosing regimens. J. Pharmacol. Exp. Ther. 321, 265–275 (2007).
Banissi, C., Ghiringhelli, F., Chen, L. & Carpentier, A. F. Treg depletion with a low-dose metronomic temozolomide regimen in a rat glioma model. Cancer Immunol. Immunother. 58, 1627–1634 (2009).
Penel, N., Adenis, A. & Bocci, G. Cyclophosphamide-based metronomic chemotherapy: after 10 years of experience, where do we stand and where are we going? Crit. Rev. Oncol. Hematol. 82, 40–50 (2012).
Bocci, G., Francia, G., Man, S., Lawler, J. & Kerbel, R. S. Thrombospondin 1, a mediator of the antiangiogenic effects of low-dose metronomic chemotherapy. Proc. Natl Acad. Sci. USA 100, 12917–12922 (2003).
Man, S. et al. Antitumor effects in mice of low-dose (metronomic) cyclophosphamide administered continuously through the drinking water. Cancer Res. 62, 2731–2735 (2002).
Klink, T. et al. Metronomic trofosfamide inhibits progression of human lung cancer xenografts by exerting anti-angiogenic effects. J. Cancer Res. Clin. Oncol. 132, 643–652 (2006).
Chen, C. S., Doloff, J. C. & Waxman, D. J. Intermittent metronomic drug schedule is essential for activating antitumor innate immunity and tumor xenograft regression. Neoplasia 16, 84–96 (2014).
Bocci, G., Di Paolo, A. & Danesi, R. The pharmacological bases of the antiangiogenic activity of paclitaxel. Angiogenesis 16, 481–492 (2013).
Di Paolo, A., Bocci, G. & Danesi, R. The preclinical bases of the rational combination of paclitaxel and antiangiogenic drugs. Clin. Cancer Drugs 1, 100–115 (2014).
Pasquier, E., Andre, N. & Braguer, D. Targeting microtubules to inhibit angiogenesis and disrupt tumour vasculature: implications for cancer treatment. Curr. Cancer Drug Targets 7, 566–581 (2007).
Schwartz, E. L. Antivascular actions of microtubule-binding drugs. Clin. Cancer Res. 15, 2594–2601 (2009).
Huang, Y. et al. Antiangiogenic activity of sterically stabilized liposomes containing paclitaxel (SSL-PTX): in vitro and in vivo. AAPS PharmSciTech 11, 752–759 (2010).
Koziara, J. M., Whisman, T. R., Tseng, M. T. & Mumper, R. J. In-vivo efficacy of novel paclitaxel nanoparticles in paclitaxel-resistant human colorectal tumors. J. Control. Release 112, 312–319 (2006).
Hammady, T., Rabanel, J. M., Dhanikula, R. S., Leclair, G. & Hildgen, P. Functionalized nanospheres loaded with anti-angiogenic drugs: cellular uptake and angiosuppressive efficacy. Eur. J. Pharm. Biopharm. 72, 418–427 (2009).
Ng, S. S. et al. Influence of formulation vehicle on metronomic taxane chemotherapy: albumin-bound versus cremophor EL-based paclitaxel. Clin. Cancer Res. 12, 4331–4338 (2006).
Miele, E., Spinelli, G. P., Tomao, F. & Tomao, S. Albumin-bound formulation of paclitaxel (Abraxane® ABI-007) in the treatment of breast cancer. Int. J. Nanomedicine 4, 99–105 (2009).
Lee, S. J. et al. Metronomic activity of CD44-targeted hyaluronic acid-paclitaxel in ovarian carcinoma. Clin. Cancer Res. 18, 4114–4121 (2012).
Luo, L. M. et al. Anti-tumor and anti-angiogenic effect of metronomic cyclic NGR-modified liposomes containing paclitaxel. Biomaterials 34, 1102–1114 (2013).
Bradshaw-Pierce, E. L., Eckhardt, S. G. & Gustafson, D. L. A physiologically based pharmacokinetic model of docetaxel disposition: from mouse to man. Clin. Cancer Res. 13, 2768–2776 (2007).
Bradshaw-Pierce, E. L., Steinhauer, C. A., Raben, D. & Gustafson, D. L. Pharmacokinetic-directed dosing of vandetanib and docetaxel in a mouse model of human squamous cell carcinoma. Mol. Cancer Ther. 7, 3006–3017 (2008).
Kumar, S. et al. Metronomic oral topotecan with pazopanib is an active antiangiogenic regimen in mouse models of aggressive pediatric solid tumor. Clin. Cancer Res. 17, 5656–5667 (2011).
van Geel, R. M., Beijnen, J. H. & Schellens, J. H. Concise drug review: pazopanib and axitinib. Oncologist 17, 1081–1089 (2012).
Hartmann, J. T. & Lipp, H. P. Camptothecin and podophyllotoxin derivatives: inhibitors of topoisomerase I and II — mechanisms of action, pharmacokinetics and toxicity profile. Drug Saf. 29, 209–230 (2006).
Andre, N., Padovani, L. & Verschuur, A. Metronomic chemotherapy: back to the future! Drug News Perspect. 23, 143–151 (2010).
Allegrini, G. et al. A pharmacokinetic and pharmacodynamic study on metronomic irinotecan in metastatic colorectal cancer patients. Br. J. Cancer 98, 1312–1319 (2008).
Tillmanns, T. D. et al. Daily oral topotecan: utilization of a metronomic dosing schedule to treat recurrent or persistent solid tumors. J. Clin. Oncol. 26, 2571 (2008).
Turner, D. C., Tillmanns, T. D., Harstead, K. E., Throm, S. L. & Stewart, C. F. Combination metronomic oral topotecan and pazopanib: a pharmacokinetic study in patients with gynecological cancer. Anticancer Res. 33, 3823–3829 (2013).
Briasoulis, E. et al. Dose-ranging study of metronomic oral vinorelbine in patients with advanced refractory cancer. Clin. Cancer Res. 15, 6454–6461 (2009).
Briasoulis, E. et al. Dose selection trial of metronomic oral vinorelbine monotherapy in patients with metastatic cancer: a hellenic cooperative oncology group clinical translational study. BMC Cancer 13, 263 (2013).
Allegrini, G. et al. Clinical, pharmacokinetic and pharmacodynamic evaluations of metronomic UFT and cyclophosphamide plus celecoxib in patients with advanced refractory gastrointestinal cancers. Angiogenesis 15, 275–286 (2012).
Moes, J. et al. Development of an oral solid dispersion formulation for use in low-dose metronomic chemotherapy of paclitaxel. Eur. J. Pharm. Biopharm. 83, 87–94 (2013).
Bazzola, L. et al. Combination of letrozole, metronomic cyclophosphamide and sorafenib is well-tolerated and shows activity in patients with primary breast cancer. Br. J. Cancer 112, 52–60 (2015).
Stempak, D. et al. A pilot pharmacokinetic and antiangiogenic biomarker study of celecoxib and low-dose metronomic vinblastine or cyclophosphamide in pediatric recurrent solid tumors. J. Pediatr. Hematol. Oncol. 28, 720–728 (2006).
Baruchel, S. et al. Safety and pharmacokinetics of temozolomide using a dose-escalation, metronomic schedule in recurrent paediatric brain tumours. Eur. J. Cancer 42, 2335–2342 (2006).
Haefeli, W. E. & Carls, A. Drug interactions with phytotherapeutics in oncology. Expert Opin. Drug Metab. Toxicol. 10, 359–377 (2014).
Roby, C. A., Anderson, G. D., Kantor, E., Dryer, D. A. & Burstein, A. H. St John's Wort: effect on CYP3A4 activity. Clin. Pharmacol. Ther. 67, 451–457 (2000).
Leveque, D. et al. Mechanisms of pharmacokinetic interactions involving oral anticancer agents. Bull. Cancer 102, 65–72 (2015).
Lasalvia-Prisco, E. et al. Insulin-induced enhancement of antitumoral response to methotrexate in breast cancer patients. Cancer Chemother. Pharmacol. 53, 220–224 (2004).
Damyanov, C., Gerasimova, D., Maslev, I. & Gavrilov, V. Low-dose chemotherapy with insulin (insulin potentiation therapy) in combination with hormone therapy for treatment of castration-resistant prostate cancer. ISRN Urol. 2012, 140182 (2012).
Chen, C., Xu, T., Lu, Y., Chen, J. & Wu, S. The efficacy of temozolomide for recurrent glioblastoma multiforme. Eur. J. Neurol. 20, 223–230 (2013).
Adenis, A. et al. A dose-escalating phase I of imatinib mesylate with fixed dose of metronomic cyclophosphamide in targeted solid tumours. Br. J. Cancer 109, 2574–2578 (2013).
Villano, J. L., Seery, T. E. & Bressler, L. R. Temozolomide in malignant gliomas: current use and future targets. Cancer Chemother. Pharmacol. 64, 647–655 (2009).
US National Library of Science. ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT01285817?term=NCT01285817&rank=1 (2015).
US National Library of Science. ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT00200161?term=NCT00200161&rank=1 (2015).
Di Paolo, A., Bocci, G., Danesi, R. & Del Tacca, M. Clinical pharmacokinetics of irinotecan-based chemotherapy in colorectal cancer patients. Curr. Clin. Pharmacol. 1, 311–323 (2006).
Falcone, A. et al. Sequence effect of irinotecan and fluorouracil treatment on pharmacokinetics and toxicity in chemotherapy-naive metastatic colorectal cancer patients. J. Clin. Oncol. 19, 3456–3462 (2001).
Herben, V. M. et al. Phase I and pharmacokinetic study of irinotecan administered as a low-dose, continuous intravenous infusion over 14 days in patients with malignant solid tumors. J. Clin. Oncol. 17, 1897–1905 (1999).
Bocci, G. et al. Antiangiogenic and anticolorectal cancer effects of metronomic irinotecan chemotherapy alone and in combination with semaxinib. Br. J. Cancer 98, 1619–1629 (2008).
Hashimoto, K. et al. Potent preclinical impact of metronomic low-dose oral topotecan combined with the antiangiogenic drug pazopanib for the treatment of ovarian cancer. Mol. Cancer Ther. 9, 996–1006 (2010).
Merritt, W. M. et al. Anti-angiogenic properties of metronomic topotecan in ovarian carcinoma. Cancer Biol. Ther. 8, 1596–1603 (2009).
Minturn, J. E. et al. A phase II study of metronomic oral topotecan for recurrent childhood brain tumors. Pediatr. Blood Cancer 56, 39–44 (2011).
Herben, V. M. et al. Oral topotecan: bioavailablity and effect of food co-administration. Br. J. Cancer 80, 1380–1386 (1999).
Schellens, J. H. et al. Bioavailability and pharmacokinetics of oral topotecan: a new topoisomerase I inhibitor. Br. J. Cancer 73, 1268–1271 (1996).
Kerklaan, B. M. et al. Phase I study of safety, tolerability, and pharmacokinetics of pazopanib in combination with oral topotecan in patients with advanced solid tumors. J. Clin. Oncol. 31, 2536 (2013).
Kerklaan, B. M. et al. Phase I and pharmacological study of pazopanib in combination with oral topotecan in patients with advanced solid tumours. Br. J. Cancer 113, 706–715 (2015).
US National Library of Science. ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT01931098?term=NCT01931098&rank=1 (2016).
Cazzaniga, M. E. et al. Metronomic oral vinorelbine in advanced breast cancer and non-small-cell lung cancer: current status and future development. Future Oncol. 12, 373–387 (2016).
Kontopodis, E. et al. A phase II study of metronomic oral vinorelbine administered in the second line and beyond in non-small cell lung cancer (NSCLC): a phase II study of the Hellenic Oncology Research Group. J. Chemother. 25, 49–55 (2013).
Addeo, R. et al. Low-dose metronomic oral administration of vinorelbine in the first-line treatment of elderly patients with metastatic breast cancer. Clin. Breast Cancer 10, 301–306 (2010).
Addeo, R. et al. Protracted low dose of oral vinorelbine and temozolomide with whole-brain radiotherapy in the treatment for breast cancer patients with brain metastases. Cancer Chemother. Pharmacol. 70, 603–609 (2012).
Pappas, P., Biziota, I., Marselos, M. & Briasoulis, E. Evaluation of antiproliferative and molecular effects of vinorelbine and its active metabolite 4-Odeacetyl-vinorelbine on human endothelial cells in an in vitro simulation model of metronomic chemotherapy. Eur. J. Cancer 6, 138–139 (2008).
Mavroeidis, L. et al. Metronomic vinorelbine: anti-angiogenic activity in vitro in normoxic and severe hypoxic conditions, and severe hypoxia-induced resistance to its anti-proliferative effect with reversal by Akt inhibition. Int. J. Oncol. 47, 455–464 (2015).
Vacca, A. et al. Antiangiogenesis is produced by nontoxic doses of vinblastine. J. Clin. Oncol. 94, 4143–4155 (1999).
Rock, B. M., Hengel, S. M., Rock, D. A., Wienkers, L. C. & Kunze, K. L. Characterization of ritonavir-mediated inactivation of cytochrome P450 3A4. Mol. Pharmacol. 86, 665–674 (2014).
Wang, J., Lou, P., Lesniewski, R. & Henkin, J. Paclitaxel at ultra low concentrations inhibits angiogenesis without affecting cellular microtubule assembly. Anticancer Drugs 14, 13–19 (2003).
Gianni, L. et al. Nonlinear pharmacokinetics and metabolism of paclitaxel and its pharmacokinetic/pharmacodynamic relationships in humans. J. Clin. Oncol. 13, 180–190 (1995).
Chatelut, E., Delord, J. P. & Canal, P. Toxicity patterns of cytotoxic drugs. Invest. New Drugs 21, 141–148 (2003).
US National Library of Science. ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT02555007?term=02555007&rank=1 (2015).
Bennouna, J., Saunders, M. & Douillard, J. Y. The role of UFT in metastatic colorectal cancer. Oncology 76, 301–310 (2009).
Tanaka, F., Wada, H. & Fukushima, M. UFT and S-1 for treatment of primary lung cancer. Gen. Thorac. Cardiovasc. Surg. 58, 3–13 (2010).
Kato, H. et al. A randomized trial of adjuvant chemotherapy with uracil-tegafur for adenocarcinoma of the lung. N. Engl. J. Med. 350, 1713–1721 (2004).
Bocci, G. & Francia, G. in Metronomic Chemotherapy. Pharmacology and Clinical Applications (eds Bocci, G. & Francia, G.) 229–246 (Springer-Verlag, 2014).
Paci, A. et al. Review of therapeutic drug monitoring of anticancer drugs part 1 — cytotoxics. Eur. J. Cancer 50, 2010–2019 (2014).
Pesenti, C., Gusella, M., Sirchia, S. M. & Miozzo, M. Germline oncopharmacogenetics, a promising field in cancer therapy. Cell. Oncol. (Dordr.) 38, 65–89 (2015).
Hubbard, J. M. & Grothey, A. When less is more: maintenance therapy in colorectal cancer. Lancet 385, 1808–1810 (2015).
Colleoni, F. et al. Low-dose oral cyclophosphamide-methotrexate maintenance (CMM) for receptor-negative early breast cancer (BC) [abstract]. J. Clin. Oncol. 33 (Suppl.), 1002 (2015).
Malik, P. S., Raina, V. & Andre, N. Metronomics as maintenance treatment in oncology: time for chemo-switch. Front. Oncol. 4, 76 (2014).
Dellapasqua, S. et al. Metronomic cyclophosphamide and capecitabine combined with bevacizumab in advanced breast cancer. J. Clin. Oncol. 26, 4899–4905 (2008).
Farkouh, A. et al. Clinical pharmacokinetics of capecitabine and its metabolites in combination with the monoclonal antibody bevacizumab. Anticancer Res. 34, 3669–3673 (2014).
US National Library of Science. ClinicalTrials.gov, https://clinicaltrials.gov/ct2/show/NCT02271464?term=NCT02271464&rank=1 (2015).
Lutsiak, M. E. et al. Inhibition of CD4+25+ T regulatory cell function implicated in enhanced immune response by low-dose cyclophosphamide. Blood 105, 2862–2868 (2005).
Wu, J. & Waxman, D. J. Metronomic cyclophosphamide eradicates large implanted GL261 gliomas by activating antitumor Cd8 T-cell responses and immune memory. Oncoimmunology 4, e1005521 (2015).
Doloff, J. C. & Waxman, D. J. Transcriptional profiling provides insights into metronomic cyclophosphamide-activated, innate immune-dependent regression of brain tumor xenografts. BMC Cancer 15, 375 (2015).
Wu, J. & Waxman, D. J. Metronomic cyclophosphamide schedule-dependence of innate immune cell recruitment and tumor regression in an implanted glioma model. Cancer Lett. 353, 272–280 (2014).
Doloff, J. C., Chen, C. S. & Waxman, D. J. Anti-tumor innate immunity activated by intermittent metronomic cyclophosphamide treatment of 9L brain tumor xenografts is preserved by anti-angiogenic drugs that spare VEGF receptor 2. Mol. Cancer 13, 158 (2014).
Doloff, J. C. & Waxman, D. J. VEGF receptor inhibitors block the ability of metronomically dosed cyclophosphamide to activate innate immunity-induced tumor regression. Cancer Res. 72, 1103–1115 (2012).
Jia, L. & Waxman, D. J. Thrombospondin-1 and pigment epithelium-derived factor enhance responsiveness of KM12 colon tumor to metronomic cyclophosphamide but have disparate effects on tumor metastasis. Cancer Lett. 330, 241–249 (2013).
Shaked, Y. et al. Optimal biologic dose of metronomic chemotherapy regimens is associated with maximum antiangiogenic activity. Blood 106, 3058–3061 (2005).
Mancuso, P. et al. Circulating endothelial-cell kinetics and viability predict survival in breast cancer patients receiving metronomic chemotherapy. Blood 108, 452–459 (2006).
Calleri, A. et al. Predictive potential of angiogenic growth factors and circulating endothelial cells in breast cancer patients receiving metronomic chemotherapy plus bevacizumab. Clin. Cancer Res. 15, 7652–7657 (2009).
Ge, Y. et al. Metronomic cyclophosphamide treatment in metastasized breast cancer patients: immunological effects and clinical outcome. Cancer Immunol. Immunother. 61, 353–362 (2012).
Ghiringhelli, F. et al. Metronomic cyclophosphamide regimen selectively depletes CD4+CD25+ regulatory T cells and restores T and NK effector functions in end stage cancer patients. Cancer Immunol. Immunother. 56, 641–648 (2007).
Koumarianou, A. et al. The effect of metronomic versus standard chemotherapy on the regulatory to effector T-cell equilibrium in cancer patients. Exp. Hematol. Oncol. 3, 3 (2014).
Nars, M. S. & Kaneno, R. Immunomodulatory effects of low dose chemotherapy and perspectives of its combination with immunotherapy. Int. J. Cancer 132, 2471–2478 (2013).
Tagliamonte, M. et al. Novel metronomic chemotherapy and cancer vaccine combinatorial strategy for hepatocellular carcinoma in a mouse model. Cancer Immunol. Immunother. 64, 1305–1314 (2015).
Chen, C. A. et al. Metronomic chemotherapy enhances antitumor effects of cancer vaccine by depleting regulatory T lymphocytes and inhibiting tumor angiogenesis. Mol. Ther. 18, 1233–1243 (2010).
Hermans, I. F., Chong, T. W., Palmowski, M. J., Harris, A. L. & Cerundolo, V. Synergistic effect of metronomic dosing of cyclophosphamide combined with specific antitumor immunotherapy in a murine melanoma model. Cancer Res. 63, 8408–8413 (2003).
Bouche, G. et al. Lessons from the Fourth Metronomic and Anti-angiogenic Therapy Meeting, 24–25 June 2014, Milan. ecancer 8, 463 (2014).
Barbolosi, D., Ciccolini, J., Lacarelle, B., Barlesi, F. & Andre, N. Computational oncology — mathematical modelling of drug regimens for precision medicine. Nat. Rev. Clin. Oncol. 13, 242–254 (2015).
Benzekry, S. et al. Metronomic reloaded: theoretical models bringing chemotherapy into the era of precision medicine. Semin. Cancer Biol. 35, 53–61 (2015).
Faivre, C., Barbolosi, D., Pasquier, E. & Andre, N. A mathematical model for the administration of temozolomide: comparative analysis of conventional and metronomic chemotherapy regimens. Cancer Chemother. Pharmacol. 71, 1013–1019 (2013).
Panetta, J. C. et al. Population pharmacokinetics of temozolomide and metabolites in infants and children with primary central nervous system tumors. Cancer Chemother. Pharmacol. 52, 435–441 (2003).
Fioravanti, A. et al. Metronomic 5-fluorouracil, oxaliplatin and irinotecan in colorectal cancer. Eur. J. Pharmacol. 619, 8–14 (2009).
Chow, A. et al. Preclinical analysis of resistance and cross-resistance to low-dose metronomic chemotherapy. Invest. New Drugs 32, 47–59 (2014).
Bocci, G. et al. Cyclophosphamide-methotrexate 'metronomic' chemotherapy for the palliative treatment of metastatic breast cancer. A comparative pharmacoeconomic evaluation. Ann. Oncol. 16, 1243–1252 (2005).
Andre, N., Banavali, S., Snihur, Y. & Pasquier, E. Has the time come for metronomics in low-income and middle-income countries? Lancet Oncol. 14, e239–e248 (2013).
Craven, O., Hughes, C. A., Burton, A., Saunders, M. P. & Molassiotis, A. Is a nurse-led telephone intervention a viable alternative to nurse-led home care and standard care for patients receiving oral capecitabine? Results from a large prospective audit in patients with colorectal cancer. Eur. J. Cancer Care (Engl.) 22, 413–419 (2013).
Cowan, D. A. Drug testing. Essays Biochem. 44, 139–148 (2008).
Adaway, J. E. & Keevil, B. G. Therapeutic drug monitoring and LC-MS/MS. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 883–884, 33–49 (2012).
Millership, J. S. Microassay of drugs and modern measurement techniques. Paediatr. Anaesth. 21, 197–205 (2011).
Anderson, L. W., Ludeman, S. M., Colvin, O. M., Grochow, L. B. & Strong, J. M. Quantitation of 4-hydroxycyclophosphamide/aldophosphamide in whole blood. J. Chromatogr. B Biomed. Appl. 667, 247–257 (1995).
Gurney, H. Dose calculation of anticancer drugs: a review of the current practice and introduction of an alternative. J. Clin. Oncol. 14, 2590–2611 (1996).
Gao, B., Klumpen, H. J. & Gurney, H. Dose calculation of anticancer drugs. Expert Opin. Drug Metab. Toxicol. 4, 1307–1319 (2008).
Gurney, H. Developing a new framework for dose calculation. J. Clin. Oncol. 24, 1489–1490 (2006).
Gurney, H. I don't underdose my patients...do I? Lancet Oncol. 6, 637–638 (2005).
Gurney, H. How to calculate the dose of chemotherapy. Br. J. Cancer 86, 1297–1302 (2002).
Bergh, J. et al. Tailored fluorouracil, epirubicin, and cyclophosphamide compared with marrow-supported high-dose chemotherapy as adjuvant treatment for high-risk breast cancer: a randomised trial. Scandinavian Breast Group 9401 study. Lancet 356, 1384–1391 (2000).
Zhang, A. Y. et al. Effect of toxicity-adjusted dose (TAD) of sunitinib on intra-patient variation of trough levels: a longitudinal study in metastatic renal cell cancer (mRCC) [abstract]. J. Clin. Oncol. 32 (Suppl.), 2597 (2014).
Morabito, A. et al. A multicenter, randomised, phase II trial comparing fixed dose versus toxicity-adjusted dose of cisplatin + etoposide in advanced SCLC patients. The STAD-1 trial [abstract]. J. Clin. Oncol. 33 (Suppl.), 7505 (2015).
Minasian, L. et al. Optimizing dosing of oncology drugs. Clin. Pharmacol. Ther. 96, 572–579 (2014).
Acknowledgements
G.B. and R.S.K. would like to thank C. Cheng for excellent secretarial assistance, and the whole editorial team of Nature Reviews Clinical Oncology for the important editing process. G.B.'s research is currently supported by grants from the Italian Association of Cancer Research (AIRC, IG 17672) and the Istituto Toscano Tumori (ITT). R.S.K's research on metronomic chemotherapy is currently supported by grants from the Canadian Institute of Health Research, the Israel Cancer Research Fund, and the Canadian Breast Cancer Foundation.
Author information
Authors and Affiliations
Contributions
G.B. and R.S.K. researched data for article, contributed to discussion of the content, wrote the manuscript, and reviewed/edited the article before submission.
Corresponding author
Ethics declarations
Competing interests
R.S.K. is a Scientific Advisory Board member of Angiocrine Biosciences, Eli Lilly, and MolMed; is a consultant to Cerulean Pharma, Merrimack Pharmaceuticals, and Triphase Accelerator; and holds stock from Angiocrine Biosciences. Over the past 2 years, R.S.K. has received honoraria from Boehringer-Ingelheim, Eli Lilly, Oncology Education, NED Biosciences and Regeneron. G.B. declares no competing interests.
PowerPoint slides
Glossary
- Clearance
-
(Cl). Is the volume of blood cleared of the drug over the time unit. In other words, the loss of drug across an organ of elimination (for example, the liver or kidney). The systemic clearance is the sum of the clearances by each of the eliminating organs26.
- Volume of distribution
-
(Vd). Is the theoretical volume in which the drug is distributed to achieve a mean concentration equal to that measured in plasma26.
- Steady state concentrations
-
(CSS). Also referred to as concentrations of the drug achieved at the steady state. Is the equilibrium reached such that the amount of drug eliminated during each dosing interval is equivalent to the amount of drug administered during that same interval. The time to achieve the steady state, regardless of the drug or dose, corresponds to five drug elimination half-lives26.
- Area under the curve
-
(AUC). Refers to the area under the plasma drug-concentration time curve and represents the total drug exposure within the body over time26.
- Mean residence time
-
(MRT). Is the average time the number of molecules introduced (injected or taken orally) reside in the body26.
- Bioavailability
-
(F). Is the percentage of a drug dose that reaches the systemic circulation, whatever the route of administration. In other words, a term commonly applied to both the rate and extent of drug input into the systemic circulation26.
- Elimination half-life
-
(elimination t1/2). Is the time taken for the plasma concentration, as well as the total amount of the drug in the body, to decline by one-half26.
Rights and permissions
About this article
Cite this article
Bocci, G., Kerbel, R. Pharmacokinetics of metronomic chemotherapy: a neglected but crucial aspect. Nat Rev Clin Oncol 13, 659–673 (2016). https://doi.org/10.1038/nrclinonc.2016.64
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2016.64
This article is cited by
-
Revisiting metronomic vinorelbine with mathematical modelling: a Phase I trial in lung cancer
Cancer Chemotherapy and Pharmacology (2022)
-
A redox-responsive dihydroartemisinin dimeric nanoprodrug for enhanced antitumor activity
Journal of Nanobiotechnology (2021)
-
Evaluation of solid tumor response to sequential treatment cycles via a new computational hybrid approach
Scientific Reports (2021)
-
Pharmacodynamic biomarkers in metronomic chemotherapy: multiplex cytokine measurements in gastrointestinal cancer patients
Clinical and Experimental Medicine (2021)
-
Which treatment is better than concurrent chemoradiotherapy about survival for stage III or IV locally advanced nasopharyngeal carcinoma? An updated Bayesian network meta-analysis of randomized controlled trials
European Archives of Oto-Rhino-Laryngology (2021)