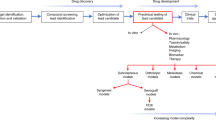
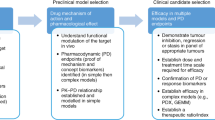
Abstract
For decades, xenografts using well-established human tumor cell lines have been the most commonly used models to study human cancers in mice. More recently, human tumors implanted directly into immunodeficient mice have become increasingly popular as evidence accrues that they more accurately recapitulate features of patient tumors. Here we describe our protocols for the orthotopic and heterotopic implantation of pancreatic cancer cell lines and freshly isolated patient tumors into immunodeficient mice. We also describe procedures for the digestion of tumors into single-cell suspensions for the isolation of subpopulations of tumor cells. Orthotopic or heterotopic implantation of established cell lines requires 1–2 h, with 1-cm tumors arising after 2–5 weeks. Engraftment of patient tumor samples takes ∼2 h and growth of palpable tumor requires ∼14 weeks. Once established, direct xenograft tumors require 2 and 5 h for heterotopic and orthotopic implantation, respectively, and 5–6 weeks for palpable tumor growth.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Jones, S. et al. Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science 321, 1801–1806 (2008).
Sharpless, N.E. & Depinho, R.A. The mighty mouse: genetically engineered mouse models in cancer drug development. Nat. Rev. Drug Discov. 5, 741–754 (2006).
Frese, K.K. & Tuveson, D.A. Maximizing mouse cancer models. Nat. Rev. Cancer 7, 645–658 (2007).
Olive, K.P. & Tuveson, D.A. The use of targeted mouse models for preclinical testing of novel cancer therapeutics. Clin. Cancer Res. 12, 5277–5287 (2006).
Rangarajan, A. & Weinberg, R.A. Opinion: comparative biology of mouse versus human cells: modelling human cancer in mice. Nat. Rev. Cancer 3, 952–959 (2003).
Fu, X., Guadagni, F. & Hoffman, R.M. A metastatic nude-mouse model of human pancreatic cancer constructed orthotopically with histologically intact patient specimens. Proc. Natl. Acad. Sci. USA 89, 5645–5649 (1992).
Loukopoulos, P. et al. Orthotopic transplantation models of pancreatic adenocarcinoma derived from cell lines and primary tumors and displaying varying metastatic activity. Pancreas 29, 193–203 (2004).
Rubio-Viqueira, B. et al. An in vivo platform for translational drug development in pancreatic cancer. Clin. Cancer Res. 12, 4652–4661 (2006).
Rubio-Viqueira, B. & Hidalgo, M. Direct in vivo xenograft tumor model for predicting chemotherapeutic drug response in cancer patients. Clin. Pharmacol. Ther. 85, 217–221 (2009).
Flanagan, S.P. 'Nude', a new hairless gene with pleiotropic effects in the mouse. Genet. Res. 8, 295–309 (1966).
Trevino, J.G. et al. Inhibition of SRC expression and activity inhibits tumor progression and metastasis of human pancreatic adenocarcinoma cells in an orthotopic nude mouse model. Am. J. Pathol. 168, 962–972 (2006).
Gray, M.J. et al. Neuropilin-1 suppresses tumorigenic properties in a human pancreatic adenocarcinoma cell line lacking neuropilin-1 coreceptors. Cancer Res. 65, 3664–3670 (2005).
Bosma, G.C., Custer, R.P. & Bosma, M.J. A severe combined immunodeficiency mutation in the mouse. Nature 301, 527–530 (1983).
Ito, M. et al. NOD/SCID/gamma(c)(null) mouse: an excellent recipient mouse model for engraftment of human cells. Blood 100, 3175–3182 (2002).
Quintana, E. et al. Efficient tumour formation by single human melanoma cells. Nature 456, 593–598 (2008).
Voskoglou-Nomikos, T., Pater, J.L. & Seymour, L. Clinical predictive value of the in vitro cell line, human xenograft, and mouse allograft preclinical cancer models. Clin. Cancer Res. 9, 4227–4239 (2003).
Johnson, J.I. et al. Relationships between drug activity in NCI preclinical in vitro and in vivo models and early clinical trials. Br. J. Cancer 84, 1424–1431 (2001).
End, D.W. et al. Characterization of the antitumor effects of the selective farnesyl protein transferase inhibitor R115777 in vivo and in vitro . Cancer Res. 61, 131–137 (2001).
Van Cutsem, E. et al. Phase III trial of gemcitabine plus tipifarnib compared with gemcitabine plus placebo in advanced pancreatic cancer. J. Clin. Oncol. 22, 1430–1438 (2004).
Garber, K. From human to mouse and back: 'tumorgraft' models surge in popularity. J. Natl. Cancer Inst. 101, 6–8 (2009).
Fichtner, I. et al. Establishment of patient-derived non-small cell lung cancer xenografts as models for the identification of predictive biomarkers. Clin. Cancer Res. 14, 6456–6468 (2008).
Houghton, P.J. et al. The pediatric preclinical testing program: description of models and early testing results. Pediatr. Blood Cancer 49, 928–940 (2007).
Peterson, J.K. & Houghton, P.J. Integrating pharmacology and in vivo cancer models in preclinical and clinical drug development. Eur. J. Cancer 40, 837–844 (2004).
Hoffman, R.M. Orthotopic metastatic mouse models for anticancer drug discovery and evaluation: a bridge to the clinic. Invest. New Drugs 17, 343–359 (1999).
Li, C. et al. Identification of pancreatic cancer stem cells. Cancer Res. 67, 1030–1037 (2007).
Al-Hajj, M., Wicha, M.S., Benito-Hernandez, A., Morrison, S.J. & Clarke, M.F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl. Acad. Sci. USA 100, 3983–3988 (2003).
O'Brien, C.A., Pollett, A., Gallinger, S. & Dick, J.E. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature 445, 106–110 (2007).
Kim, M.P., Gallick, G.E., Evans, D.B., Wang, H.M. & Fleming, J.B. Effect of preoperative therapy on engraftment of pancreatic adenocarcinoma tumor in mice after pancreatectomy. Proceedings of the 2009 American Society of Clinical Oncology—Gastrointestinal Cancers Symposium, 15–17 January 2009, San Francisco, CA. Abstract no. 165, p 142.
Kim, M.P., Fleming, J.B., Evans, D.B., Wang, H.M. & Gallick, G.E. Pancreatic cancer xenografts contain distinct populations of cells expressing the cancer stem cell marker CD133. Proceedings of the 2009 American Society of Clinical Oncology—Gastrointestinal Cancers Symposium, 15–17 January 2009, San Francisco, CA. Abstract no. 150, p 135.
Bruns, C.J., Harbison, M.T., Kuniyasu, H., Eue, I. & Fidler, I.J. In vivo selection and characterization of metastatic variants from human pancreatic adenocarcinoma by using orthotopic implantation in nude mice. Neoplasia 1, 50–62 (1999).
Khanbolooki, S. et al. Nuclear factor-kappaB maintains TRAIL resistance inhuman pancreatic cancer cells. Mol. Cancer Ther. 5, 2251–2260 (2006).
Nawrocki, S.T., Sweeney-Gotsch, B., Takamori, R. & McConkey, D.J. The proteasome inhibitor bortezomib enhances the activity of docetaxel in orthotopic human pancreatic tumor xenografts. Mol. Cancer Ther. 3, 59–70 (2004).
Acknowledgements
This work was supported by the Various Donor Fund for Pancreatic Cancer Research, M.D. Anderson Cancer Center (J.B.F. and D.B.E.), NIH T-32 09599 (M.P.K.), and NIH 5P20A101936 (G.E.G. and J.L.A.).
Author information
Authors and Affiliations
Contributions
M.P.K., J.B.F. and G.E.G. designed experiments and analyzed data; M.P.K. carried out the animal study and conducted experiments; M.P.K., J.L.A. and G.E.G. wrote the paper; D.B.E., J.B.F. and H.W. acquired and processed surgical specimens; and H.W. examined pathologic specimens.
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, M., Evans, D., Wang, H. et al. Generation of orthotopic and heterotopic human pancreatic cancer xenografts in immunodeficient mice. Nat Protoc 4, 1670–1680 (2009). https://doi.org/10.1038/nprot.2009.171
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2009.171
This article is cited by
-
Inhibition of ADAM9 promotes the selective degradation of KRAS and sensitizes pancreatic cancers to chemotherapy
Nature Cancer (2024)
-
Advances towards the use of gastrointestinal tumor patient-derived organoids as a therapeutic decision-making tool
Biological Research (2023)
-
High-throughput functional screen identifies YWHAZ as a key regulator of pancreatic cancer metastasis
Cell Death & Disease (2023)
-
Fully implantable and battery-free wireless optoelectronic system for modulable cancer therapy and real-time monitoring
npj Flexible Electronics (2023)
-
RBFOX2 deregulation promotes pancreatic cancer progression and metastasis through alternative splicing
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.