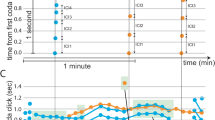
Abstract
Social recognition (SR) enables rodents to distinguish between familiar and novel conspecifics, largely through individual odor cues. SR tasks utilize the tendency for a male to sniff and interact with a novel individual more than a familiar individual. Many paradigms have been used to study the roles of the neuropeptides oxytocin and vasopressin in SR. However, inconsistencies in results have arisen within similar mouse strains, and across different paradigms and laboratories, making reliable testing of SR difficult. The current protocol details a novel approach that is replicable across investigators and in different strains of mice. We created a protocol that uses gonadally intact, singly housed females presented within corrals to group-housed males. Housing females singly before testing is particularly important for reliable discrimination. This methodology will be useful for studying short-term social memory in rodents, and may also be applicable for longer term studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ferguson, J.N., Young, L.J. & Insel, T.R. The neuroendocrine basis of social recognition. Front. Neuroendocrinol. 23, 200–224 (2002).
Spehr, M. et al. Parallel processing of social signals by the mammalian main and accessory olfactory systems. Cell. Mol. Life Sci. 63, 1476–1484 (2006).
Brennan, P.A. & Kendrick, K.M. Mammalian social odours: attraction and individual recognition. Philos. Trans. R. Soc. Lond. B Biol. Sci. 361, 2061–2078 (2006).
Mateo, J.M. Development of individually distinct recognition cues. Dev. Psychobiol. 48, 508–519 (2006).
Petrulis, A., Alvarez, P. & Eichenbaum, H. Neural correlates of social odor recognition and the representation of individual distinctive social odors within entorhinal cortex and ventral subiculum. Neuroscience 130, 259–274 (2005).
Kogan, J.H., Frankland, P.W. & Silva, A.J. Long-term memory underlying hippocampus-dependent social recognition in mice. Hippocampus 10, 47–56 (2000).
Dantzer, R., Bluthe, R.M., Koob, G.F. & Le Moal, M. Modulation of social memory in male rats by neurohypophyseal peptides. Psychopharmacology (Berl) 91, 363–368 (1987).
Winslow, J.T. & Camacho, F. Cholinergic modulation of a decrement in social investigation following repeated contacts between mice. Psychopharmacology (Berl) 121, 164–172 (1995).
Engelmann, M., Wotjak, C.T. & Landgraf, R. Social discrimination procedure: an alternative method to investigate juvenile recognition abilities in rats. Physiol. Behav. 58, 315–321 (1995).
Ferguson, J.N. et al. Social amnesia in mice lacking the oxytocin gene. Nat. Genet. 25, 284–288 (2000).
Ferguson, J.N., Aldag, J.M., Insel, T.R. & Young, L.J. Oxytocin in the medial amygdala is essential for social recognition in the mouse. J. Neurosci. 21, 8278–8285 (2001).
Wersinger, S.R., Ginns, E.I., O'Carroll, A.M., Lolait, S.J. & Young, W.S. III Vasopressin V1b receptor knockout reduces aggressive behavior in male mice. Mol. Psychiatry 7, 975–984 (2002).
Choleris, E. et al. An estrogen-dependent four-gene micronet regulating social recognition: a study with oxytocin and estrogen receptor-alpha and -beta knockout mice. Proc. Natl. Acad. Sci. USA 100, 6192–6197 (2003).
Wersinger, S.R. et al. Vasopressin 1a receptor knockout mice have a subtle olfactory deficit but normal aggression. Genes Brain Behav. 6, 540–551 (2007).
Lee, H.-J., Caldwell, H.K., Macbeth, A.H., Tolu, S.G. & Young, W.S. III A conditional knockout mouse line of the oxytocin receptor. Endocrinology 149, 3256–3263 (2008).
Bielsky, I.F., Hu, S.B., Szegda, K.L., Westphal, H. & Young, L.J. Profound impairment in social recognition and reduction in anxiety-like behavior in vasopressin V1a receptor knockout mice. Neuropsychopharmacology 29, 483–493 (2004).
Takayanagi, Y. et al. Pervasive social deficits, but normal parturition, in oxytocin receptor-deficient mice. Proc. Natl. Acad. Sci. USA 102, 16096–16101 (2005).
Jin, D. et al. CD38 is critical for social behaviour by regulating oxytocin secretion. Nature 446, 41–45 (2007).
Bielsky, I.F., Hu, S.B., Ren, X., Terwilliger, E.F. & Young, L.J. The V1a vasopressin receptor is necessary and sufficient for normal social recognition: a gene replacement study. Neuron 47, 503–513 (2005).
Benelli, A. et al. Polymodal dose–response curve for oxytocin in the social recognition test. Neuropeptides 28, 251–255 (1995).
Popik, P. & van Ree, J.M. Oxytocin but not vasopressin facilitates social recognition following injection into the medial preoptic area of the rat brain. Eur. Neuropsychopharmacol. 1, 555–560 (1991).
Popik, P., Vetulani, J. & Van Ree, J.M. Facilitation and attenuation of social recognition in rats by different oxytocin-related peptides. Eur. J. Pharmacol. 308, 113–116 (1996).
Dluzen, D.E., Muraoka, S., Engelmann, M. & Landgraf, R. The effects of infusion of arginine vasopressin, oxytocin, or their antagonists into the olfactory bulb upon social recognition responses in male rats. Peptides 19, 999–1005 (1998).
Bluthe, R.M. & Dantzer, R. Social recognition does not involve vasopressinergic neurotransmission in female rats. Brain Res. 535, 301–304 (1990).
Taylor, G.T. Urinary odors and size protect juvenile laboratory mice from adult male attack. Dev. Psychobiol. 15, 171–186 (1982).
Connor, J.L. & Lynds, P.G. Mouse aggression and the intruder-familiarity effect: evidence for multiple-factor determination c57bl. J. Comp. Physiol. Psychol. 91, 270–280 (1977).
O'Donnell, V., Blanchard, R.J. & Blanchard, D.C. Mouse aggression increases after 24 h of isolation or housing with females. Behav. Neural. Biol. 32, 89–103 (1981).
Nakamura, K., Kikusui, T., Takeuchi, Y. & Mori, Y. The critical role of familiar urine odor in diminishing territorial aggression toward a castrated intruder in mice. Physiol. Behav. 90, 512–517 (2007).
de Catanzaro, D. & Gorzalka, B.B. Isolation-induced facilitation of male sexual behavior in mice. J. Comp. Physiol. Psychol. 93, 211–222 (1979).
Sharp, J.L., Zammit, T.G., Azar, T.A. & Lawson, D.M. Stress-like responses to common procedures in male rats housed alone or with other rats. Contemp. Top. Lab. Anim. Sci. 41, 8–14 (2002).
Todrank, J., Heth, G. & Johnston, R.E. Kin recognition in golden hamsters: evidence for kinship odours. Anim. Behav. 55, 377–386 (1998).
Hopp, S.L., Owren, M.J. & Marion, J.R. Olfactory discrimination of individual littermates in rats (Rattus norvegicus). J. Comp. Psychol. 99, 248–251 (1985).
Kavaliers, M. et al. Oxytocin and estrogen receptor alpha and beta knockout mice provide discriminably different odor cues in behavioral assays. Genes Brain Behav. 3, 189–195 (2004).
Beynon, R.J. & Hurst, J.L. Urinary proteins and the modulation of chemical scents in mice and rats. Peptides 25, 1553–1563 (2004).
Hurst, J.L., Thom, M.D., Nevison, C.M., Humphries, R.E. & Beynon, R.J. MHC odours are not required or sufficient for recognition of individual scent owners. Proc. Biol. Sci. 272, 715–724 (2005).
Cheetham, S.A. et al. The genetic basis of individual-recognition signals in the mouse. Curr. Biol. 17, 1771–1777 (2007).
Sherborne, A.L. et al. The genetic basis of inbreeding avoidance in house mice. Curr. Biol. 17, 2061–2066 (2007).
Insel, T.R. & Fernald, R.D. How the brain processes social information: searching for the social brain. Annu. Rev. Neurosci. 27, 697–722 (2004).
Kudryavtseva, N.N., Bondar, N.P. & Avgustinovich, D.F. Association between experience of aggression and anxiety in male mice. Behav. Brain Res. 133, 83–93 (2002).
Muroi, Y., Ishii, T., Komori, S. & Nishimura, M. A competitive effect of androgen signaling on male mouse attraction to volatile female mouse odors. Physiol. Behav. 87, 199–205 (2006).
Carr, W.J., Loeb, L.S. & Dissinger, M.L. Responses of rats to sex odors. J. Comp. Physiol. Psychol. 59, 370–377 (1965).
Ingersoll, D.W. & Weinhold, L.L. Modulation of male mouse sniff, attack, and mount behaviors by estrous cycle-dependent urinary cues. Behav. Neural. Biol. 48, 24–42 (1987).
Macbeth, A.H., Lee, H.J., Edds, J. & Young, W.S. III Oxytocin and the oxytocin receptor underlie intrastrain, but not interstrain, social recognition. Genes. Brain. Behav. 8, 558–567 (2009).
Lim, M.M. & Young, L.J. Neuropeptidergic regulation of affiliative behavior and social bonding in animals. Horm. Behav. 50, 506–517 (2006).
Caldwell, H.K., Lee, H.J., Macbeth, A.H. & Young, W.S. III Vasopressin: behavioral roles of an 'original' neuropeptide. Prog. Neurobiol. 84, 1–24 (2008).
Caldwell, H.K. & Young, W.S. Oxytocin and Vasopressin: Genetics and Behavioral Implications in Handbook of Neurochemistry and Molecular Neurobiology: Neuroactive Proteins and Peptides, 3rd edition (ed R. Lim) 573–607 (Springer, 2006). pp. 573–607.
Popik, P. & Vetulani, J. Opposite action of oxytocin and its peptide antagonists on social memory in rats. Neuropeptides 18, 23–27 (1991).
Wersinger, S.R., Temple, J.L., Caldwell, H.K. & Young, W.S. III Inactivation of the oxytocin and the vasopressin (Avp) 1b receptor genes, but not the Avp 1a receptor gene, differentially impairs the Bruce effect in laboratory mice (Mus musculus). Endocrinology 149, 116–121 (2008).
Winslow, J.T. Mouse social recognition and preference. Curr. Protoc. Neurosci. 8 Unit 8.16 (2003).
Anagnostopoulos, A.V., Mobraaten, L.E., Sharp, J.J. & Davisson, M.T. Transgenic and knockout databases: behavioral profiles of mouse mutants. Physiol. Behav. 73, 675–689 (2001).
Ennaceur, A., Michalikova, S. & Chazot, P.L. Models of anxiety: responses of rats to novelty in an open space and an enclosed space. Behav. Brain Res. 171, 26–49 (2006).
Brooks, S.P., Pask, T., Jones, L. & Dunnett, S.B. Behavioural profiles of inbred mouse strains used as transgenic backgrounds. II: cognitive tests. Genes Brain Behav. 4, 307–317 (2005).
Moy, S.S. et al. Sociability and preference for social novelty in five inbred strains: an approach to assess autistic-like behavior in mice. Genes Brain Behav. 3, 287–302 (2004).
Sankoorikal, G.M., Kaercher, K.A., Boon, C.J., Lee, J.K. & Brodkin, E.S. A mouse model system for genetic analysis of sociability: C57BL/6J versus BALB/cJ inbred mouse strains. Biol. Psychiatry 59, 415–423 (2006).
Crawley, J. & Goodwin, F.K. Preliminary report of a simple animal behavior model for the anxiolytic effects of benzodiazepines. Pharmacol. Biochem. Behav. 13, 167–170 (1980).
Noldus, L.P., Trienes, R.J., Hendriksen, A.H., Jansen, H. & Jansen, R.G. The Observer Video-Pro: new software for the collection, management, and presentation of time-structured data from videotapes and digital media files. Behav. Res. Methods Instrum. Comput. 32, 197–206 (2000).
Guan, X. & Dluzen, D.E. Age related changes of social memory/recognition in male Fischer 344 rats. Behav. Brain Res. 61, 87–90 (1994).
Nelson, J.F., Felicio, L.S., Randall, P.K., Sims, C. & Finch, C.E. A longitudinal study of estrous cyclicity in aging C57BL/6J mice: I. Cycle frequency, length and vaginal cytology. Biol. Reprod. 27, 327–339 (1982).
Crawley, J.N. et al. Social approach behaviors in oxytocin knockout mice: comparison of two independent lines tested in different laboratory environments. Neuropeptides 41, 145–163 (2007).
Acknowledgements
We thank Dr. Scott Wersinger for his helpful review of an early version of this paper. The authors appreciate the excellent technical support provided by Emily Shepard, James Heath, Anna Brownstein and the Building 49 animal facility. This research was supported by the NIMH Intramural Research Program (Z01-MH-002498-20).
Author information
Authors and Affiliations
Contributions
A.H.M. contributed to study design, measurements, analysis and writing. J.S.E. contributed to measurements and analysis. W.S.Y. contributed to design, analysis and writing.
Corresponding author
Supplementary information
Supplementary Movie
Investigation of females during the social discrimination task. Note the insertion of the subject male’s nose between the bars of both corrals in order to investigate both stimulus females. This can be observed when the male is on the near and far side of the corrals. The beeping heard in the background is the timer used by the experimenter during live scoring; the noise does not seem to adversely affect the subject male’s behavior. (MOV 4379 kb)
Rights and permissions
About this article
Cite this article
Macbeth, A., Edds, J. & Young, W. Housing conditions and stimulus females: a robust social discrimination task for studying male rodent social recognition. Nat Protoc 4, 1574–1581 (2009). https://doi.org/10.1038/nprot.2009.141
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2009.141
This article is cited by
-
IGF-1 release in the medial prefrontal cortex mediates the rapid and sustained antidepressant-like actions of ketamine
Translational Psychiatry (2022)
-
Medial PFC AMPA receptor and BDNF signaling are required for the rapid and sustained antidepressant-like effects of 5-HT1A receptor stimulation
Neuropsychopharmacology (2020)
-
Partial loss of psychiatric risk gene Mir137 in mice causes repetitive behavior and impairs sociability and learning via increased Pde10a
Nature Neuroscience (2018)
-
High-Fat Diet Induced Anxiety and Anhedonia: Impact on Brain Homeostasis and Inflammation
Neuropsychopharmacology (2016)
-
Effects of sex and gonadectomy on social investigation and social recognition in mice
BMC Neuroscience (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.