Abstract
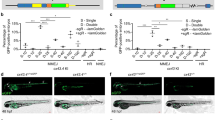
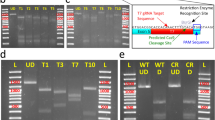
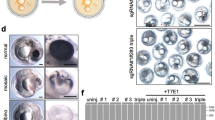
Small fish are a popular laboratory model for studying gene expression and function by transgenesis. If, however, the transgenes are not readily detectable by visual inspection, a large number of embryos must be injected, raised and screened to identify positive founder fish. Here, we describe a strategy to efficiently generate and preselect transgenic lines harbouring any transgene of interest. Co-injection of a selectable reporter construct (e.g., GFP), together with the transgene of interest on a separate plasmid using the I-SceI meganuclease approach, results in co-distribution of the two plasmids. The quality of GFP expression within the F0 generation therefore reflects the quality of injection and allows efficient and reliable selection of founder fish that are also positive for the second transgene of interest. In our experience, a large fraction (up to 50%) of GFP-positive fish will also be transgenic for the second transgene, thus providing a rapid (within 3–4 months) and efficient way to establish transgenic lines for any gene of interest in medaka and zebrafish.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Gaiano, N., Allende, M., Amsterdam, A., Kawakami, K. & Hopkins, N. Highly efficient germ-line transmission of proviral insertions in zebrafish. Proc. Natl. Acad. Sci. USA 93, 7777–7782 (1996).
Lin, S. et al. Integration and germ-line transmission of a pseudotyped retroviral vector in zebrafish. Science 265, 666–669 (1994).
Linney, E., Hardison, N.L., Lonze, B.E., Lyons, S. & DiNapoli, L. Transgene expression in zebrafish: a comparison of retroviral-vector and DNA-injection approaches. Dev. Biol. 213, 201–216 (1999).
Ellingsen, S. et al. Large-scale enhancer detection in the zebrafish genome. Development 132, 3799–3811 (2005).
Grabher, C. et al. Transposon-mediated enhancer trapping in medaka. Gene 322, 57–66 (2003).
Kawakami, K. et al. A transposon-mediated gene trap approach identifies developmentally regulated genes in zebrafish. Dev. Cell 7, 133–144 (2004).
Inoue, K. et al. Electroporation as a new technique for producing transgenic fish. Cell Differ. Dev. 29, 123–128 (1990).
Ono, H., Hirose, E., Miyazaki, K., Yamamoto, H. & Matsumoto, J. Transgenic medaka fish bearing the mouse tyrosinase gene: expression and transmission of the transgene following electroporation of the orange-colored variant. Pigment Cell Res. 10, 168–175 (1997).
Tawk, M., Tuil, D., Torrente, Y., Vriz, S. & Paulin, D. High-efficiency gene transfer into adult fish: a new tool to study fin regeneration. Genesis 32, 27–31 (2002).
Sussman, R. Direct DNA delivery into zebrafish embryos employing tissue culture techniques. Genesis 31, 1–5 (2001).
Muller, F. et al. Introducing foreign genes into fish eggs with electroporated sperm as a carrier. Mol. Mar. Biol. Biotech. 1, 276–281 (1992).
Sin, F.Y. et al. Electroporation of salmon sperm for gene transfer: efficiency, reliability, and fate of transgene. Mol. Reprod. Dev. 56, 285–288 (2000).
Zelenin, A.V. et al. The delivery of foreign genes into fertilized fish eggs using high-velocity microprojectiles. FEBS Lett. 287, 118–120 (1991).
Yamauchi, M. et al. Introduction of a foreign gene into medakafish using the particle gun method. J. Exp. Zool. 287, 285–293 (2000).
Hong, Y., Winkler, C. & Schartl, M. Production of medakafish chimeras from a stable embryonic stem cell line. Proc. Natl. Acad. Sci. USA 95, 3679–3684 (1998).
Ma, C.G., Fan, L.C., Ganassin, R., Bols, N. & Collodi, P. Production of zebrafish germ-line chimeras from embryo cell cultures. Proc. Natl. Acad. Sci. USA 98, 2461–2466 (2001).
Zhu, Z.Y. & Sun, Y.H. Embryonic and genetic manipulation in fish. Cell Res. 10, 17–27 (2000).
Wakamatsu, Y. et al. Fertile and diploid nuclear transplants derived from embryonic cells of a small laboratory fish, medaka (Oryzias latipes). Proc. Natl. Acad. Sci. USA 98, 1071–1076 (2001).
Lee, K.-Y., Huang, H., Ju, B., Yang, Z. & Lin, S. Cloned zebrafish by nuclear transfer from long-term-cultured cells. Natl. Biotechnol. 20, 795–799 (2002).
Ozato, K. et al. Production of transgenic fish: introduction and expression of chicken delta-crystallin gene in medaka embryos. Cell Differ. 19, 237–244 (1986).
Stuart, G.W., McMurray, J.V. & Westerfield, M. Replication, integration and stable germ-line transmission of foreign sequences injected into early zebrafish embryos. Development 103, 403–412 (1988).
Lin, S. Transgenic zebrafish. Methods Mol. Biol. 136, 375–383 (2000).
Chou, C.Y., Horng, L.S. & Tsai, H.J. Uniform GFP-expression in transgenic medaka (Oryzias latipes) at the F0 generation. Transgenic Res. 10, 303–315 (2001).
Thermes, V. et al. I-SceI meganuclease mediates highly efficient transgenesis in fish. Mech. Dev. 118, 91–98 (2002).
Grabher, C., Joly, J.S. & Wittbrodt, J. Highly efficient zebrafish transgenesis mediated by the meganuclease I-SceI. Methods Cell Biol. 77, 381–401 (2004).
Terasaki, H. et al. Transgenic analysis of the medaka mesp-b enhancer in somitogenesis. Dev. Growth Differ. 48, 153–168 (2006).
Deschet, K., Nakatani, Y. & Smith, W.C. Generation of Ci-Brachyury-GFP stable transgenic lines in the ascidian Ciona savignyi. Genesis 35, 248–259 (2003).
Hosemann, K.E., Colosimo, P.F., Summers, B.R. & Kingsley, D.M. A simple and efficient microinjection protocol for making transgenic sticklebacks. Behaviour 141, 1345–1355 (2004).
Ogino, H., McConnell, W.B. & Grainger, R.M. Highly efficient transgenesis in Xenopus tropicalis using I-SceI meganuclease. Mech. Dev. 123, 103–113 (2006).
Pan, F.C., Chen, Y., Loeber, J., Henningfeld, K. & Pieler, T. I-SceI meganuclease-mediated transgenesis in Xenopus. Dev. Dyn. 235, 247–252 (2006).
Jacquier, A. & Dujon, B. An intron-encoded protein is active in a gene conversion process that spreads an intron into a mitochondrial gene. Cell 41, 383–394 (1985).
Vallone, D., Gondi, S.B., Whitmore, D. & Foulkes, N.S. E-box function in a period gene repressed by light. Proc. Natl. Acad. Sci. USA 101, 4106–4111 (2004).
Kaneko, M. & Cahill, G.M. Light-dependent development of circadian gene expression in transgenic zebrafish. PLoS Biol. 3, e34 (2005).
Muller, F. et al. Activator effect of coinjected enhancers on the muscle-specific expression of promoters in zebrafish embryos. Mol. Reprod. Dev. 47, 404–412 (1997).
Grabher, C. & Wittbrodt, J. Efficient activation of gene expression using a heat-shock inducible Gal4/Vp16-UAS system in medaka. BMC Biotechnol. 4, 26 (2004).
Wittbrodt, J., Shima, A. & Schartl, M. Medaka – A model organism from the Far East. Nature Rev. Genet. 3, 53–64 (2002).
Yamamoto, T. Medaka (Killifish), Biology and Strains (Keigaku Publishing Company, Tokyo, 1975).
Westerfield, M. The Zebrafish Book (The University of Oregon Press, Eugene, 1995).
Acknowledgements
We thank Aldona Nowicka and Diana Hofmann for expert fish husbandry and Felix Loosli for critical input to the manuscript. This work was supported by a grant from the European Union (FP6 Strep: Plurigenes) to J.W.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Rembold, M., Lahiri, K., Foulkes, N. et al. Transgenesis in fish: efficient selection of transgenic fish by co-injection with a fluorescent reporter construct. Nat Protoc 1, 1133–1139 (2006). https://doi.org/10.1038/nprot.2006.165
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.165
This article is cited by
-
Deep learning-enhanced light-field imaging with continuous validation
Nature Methods (2021)
-
A detailed procedure for CRISPR/Cas9-mediated gene editing in tilapia
Hydrobiologia (2021)
-
Establishment and application of distant hybridization technology in fish
Science China Life Sciences (2019)
-
Asymmetric pitx2 expression in medaka epithalamus is regulated by nodal signaling through an intronic enhancer
Development Genes and Evolution (2018)
-
Expression of the novel maternal centrosome assembly factor Wdr8 is required for vertebrate embryonic mitoses
Nature Communications (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.