Abstract
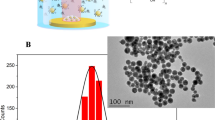
Quantum dots have been used in biomedical research for imaging1,2, diagnostics3,4 and sensing purposes5,6. However, concerns over the cytotoxicity of their heavy metal constituents7,8 and conflicting results from in vitro7,9 and small animal10,11,12,13,14 toxicity studies have limited their translation towards clinical applications. Here, we show in a pilot study that rhesus macaques injected with phospholipid micelle-encapsulated CdSe/CdS/ZnS quantum dots do not exhibit evidence of toxicity. Blood and biochemical markers remained within normal ranges following treatment, and histology of major organs after 90 days showed no abnormalities. Our results show that acute toxicity of these quantum dots in vivo can be minimal. However, chemical analysis revealed that most of the initial dose of cadmium remained in the liver, spleen and kidneys after 90 days. This means that the breakdown and clearance of quantum dots is quite slow, suggesting that longer-term studies will be required to determine the ultimate fate of these heavy metals and the impact of their persistence in primates.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Medintz, I. L., Uyeda, H. T., Goldman, E. R. & Mattoussi, H. Quantum dot bioconjugates for imaging, labelling and sensing. Nature Mater. 4, 435–446 (2005).
Yong, K-T. Mn-doped near-infrared quantum dots as multimodal targeted probes for pancreatic cancer imaging. Nanotechnology 20, 015102 (2009).
Michalet, X. et al. Quantum dots for live cells, in vivo imaging, and diagnostics. Science 307, 538–544 (2005).
Bruchez, M., Moronne, M., Gin, P., Weiss, S. & Alivisatos, A. P. Semiconductor nanocrystals as fluorescent biological labels. Science 281, 2013–2016 (1998).
Chan, W. C. W. & Nie, S. Quantum dot bioconjugates for ultrasensitive nonisotopic detection. Science 281, 2016–2018 (1998).
Mattoussi, H., Palui, G. & Na, H. B. Luminescent quantum dots as platforms for probing in vitro and in vivo biological processes. Adv. Drug Deliv. Rev. 64, 138–166 (2012).
Derfus, A. M., Chan, W. C. W. & Bhatia, S. N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett. 4, 11–18 (2004).
Choi, H. S. et al. Tissue- and organ-selective biodistribution of NIR fluorescent quantum dots. Nano Lett. 9, 2354–2359 (2009).
Cho, S. J. et al. Long-term exposure to CdTe quantum dots causes functional impairments in live cells. Langmuir 23, 1974–1980 (2007).
Yong, K-T., Roy, I., Ding, H., Bergey, E. J. & Prasad, P. N. Biocompatible near-infrared quantum dots as ultrasensitive probes for long-term in vivo imaging applications. Small 5, 1997–2004 (2009).
Hauck, T. S., Anderson, R. E., Fischer, H. C., Newbigging, S. & Chan, W. C. W. In vivo quantum-dot toxicity assessment. Small 6, 138–144 (2010).
Ballou, B., Lagerholm, B. C., Ernst, L. A., Bruchez, M. P. & Waggoner, A. S. Noninvasive imaging of quantum dots in mice. Bioconj. Chem. 15, 79–86 (2004).
Fischer, H. C., Liu, L., Pang, K. S. & Chan, W. C. W. Pharmacokinetics of nanoscale quantum dots: in vivo distribution, sequestration, and clearance in the rat. Adv. Funct. Mater. 16, 1299–1305 (2006).
Dubertret, B. et al. In vivo imaging of quantum dots encapsulated in phospholipid micelles. Science 298, 1759–1762 (2002).
Colvin, V. L. The potential environmental impact of engineered nanomaterials. Nature Biotechnol. 21, 1166–1170 (2003).
Werlin, R. et al. Biomagnification of cadmium selenide quantum dots in a simple experimental microbial food chain. Nature Nanotech. 6, 65–71 (2011).
Hardman, R. A toxicologic review of quantum dots: toxicity depends on physicochemical and environmental factors. Environ. Health Perspect. 114, 165–172 (2006).
Choi, H. S. et al. Renal clearance of quantum dots. Nature Biotechnol. 25, 1165–1170 (2007).
Hoshino, A. et al. Physicochemical properties and cellular toxicity of nanocrystal quantum dots depend on their surface modification. Nano Lett. 4, 2163–2169 (2004).
Kirchner, C. et al. Cytotoxicity of colloidal CdSe and CdSe/ZnS nanoparticles. Nano Lett. 5, 331–338 (2005).
Xia, H-J., Zhang, G-H., Wang, R-R. & Zheng, Y-T. The influence of age and sex on the cell counts of peripheral blood leukocyte subpopulations in Chinese rhesus macaques. Cell Mol. Immunol. 6, 433–440 (2009).
Xia, H., Liu, H., Zhang, G. & Zheng, Y. Phenotype and function of monocyte-derived dendritic cells from Chinese rhesus macaques. Cell Mol. Immunol. 6, 159–165 (2009).
Ho, C-C. et al. Quantum dot 705, a cadmium-based nanoparticle, induces persistent inflammation and granuloma formation in the mouse lung. Nanotoxicology http://dx.doi.org/10.3109/17435390.2011.635814 (2011).
Fitzpatrick, J. A. J. et al. Long-term persistence and spectral blue shifting of quantum dots in vivo. Nano Lett. 9, 2736–2741 (2009).
Schipper, M. L. et al. Particle size, surface coating, and PEGylation influence the biodistribution of quantum dots in living mice. Small 5, 126–134 (2009).
Al-Jamal, W. T., Al-Jamal, K. T., Cakebread, A., Halket, J. M. & Kostarelos, K. Blood circulation and tissue biodistribution of lipid–quantum dot (L-QD) hybrid vesicles intravenously administered in mice. Bioconj. Chem. 20, 1696–1702 (2009).
Yang, R. S. H. et al. Persistent tissue kinetics and redistribution of nanoparticles, quantum dot 705, in mice: ICP-MS quantitative assessment. Environ. Health Perspect. 115, 1339–1343 (2007).
Larson, D. R. et al. Water-soluble quantum dots for multiphoton fluorescence imaging in vivo. Science 300, 1434–1436 (2003).
Gao, X., Cui, Y., Levenson, R. M., Chung, L. W. K. & Nie, S. In vivo cancer targeting and imaging with semiconductor quantum dots. Nature Biotechnol. 22, 969–976 (2004).
Manna, L., Scher, E. C., Li, L-S. & Alivisatos, A. P. Epitaxial growth and photochemical annealing of graded CdS/ZnS shells on colloidal CdSe nanorods. J. Am. Chem. Soc. 124, 7136–7145 (2002).
Acknowledgements
This work was supported by The John R. Oishei Foundation, Air Force Office of Scientific Research (grant no. FA95500610398), the Singapore Ministry of Education (Grants Tier 2 MOE2010-T2-2-010 (M4020020.040 ARC2/11) and Tier 1 M4010360.040 RG29/10), Nanyang Technological University (start-up grant no. M4080141.040), the Beijing Natural Science Foundation (no. 7092097) and the National Natural Science Foundation of China (no. 21071150). The authors thank A. Maitra of Johns Hopkins University for helpful discussions.
Author information
Authors and Affiliations
Contributions
K.T.Y. and L.Y. designed the research. K.T.Y, L.Y., R.H., L.L., J.Z., I.R. W.C.L., J.L., K.W., J.L., Y.L. and Y.H. performed the research. L.Y., K.T.Y., L.L., I.R., R.H., J.Z., H.C., W.C.L., J.L., K.W., J.L., Y.L., Y.H., X.Z., M.T.S. and P.N.P. analysed the data. K.T.Y., L.Y., I.R., M.T.S. and P.N.P. co-wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 21264 kb)
Rights and permissions
About this article
Cite this article
Ye, L., Yong, KT., Liu, L. et al. A pilot study in non-human primates shows no adverse response to intravenous injection of quantum dots. Nature Nanotech 7, 453–458 (2012). https://doi.org/10.1038/nnano.2012.74
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2012.74
This article is cited by
-
Systematic evaluation of membrane-camouflaged nanoparticles in neutralizing Clostridium perfringens ε-toxin
Journal of Nanobiotechnology (2023)
-
A hepatocyte-targeting nanoparticle for enhanced hepatobiliary magnetic resonance imaging
Nature Biomedical Engineering (2022)
-
Broadening the biocompatibility of gold nanorods from rat to Macaca fascicularis: advancing clinical potential
Journal of Nanobiotechnology (2021)
-
Molecular imaging and disease theranostics with renal-clearable optical agents
Nature Reviews Materials (2021)
-
Aggregation-induced emission luminogens for image-guided surgery in non-human primates
Nature Communications (2021)