Abstract
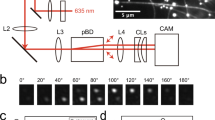
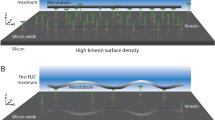
Owing to their wide spectrum of in vivo functions, motor proteins, such as kinesin-1, show great potential for application as nanomachines in engineered environments. When attached to a substrate surface, these motors are envisioned to shuttle cargo that is bound to reconstituted microtubules—one component of the cell cytoskeleton—from one location to another1,2. One potentially serious problem for such applications is, however, the rotation of the microtubules around their longitudinal axis3,4. Here we explore this issue by labelling the gliding microtubules with quantum dots to simultaneously follow their sinusoidal side-to-side and up-and-down motion in three dimensions with nanometre accuracy. Microtubule rotation, which originates from the kinesin moving along the individual protofilaments of the microtubule, was not impeded by the quantum dots. However, pick-up of large cargo inhibited the rotation but did not affect the velocity of microtubule gliding. Our data show that kinesin-driven microtubules make flexible, responsive and effective molecular shuttles for nanotransport applications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hess, H., Bachand, G. D. & Vogel, V. Powering nanodevices with biomolecular motors. Chem. Eur. J. 10, 2110–2116 (2004).
van den Heuvel, M. G. & Dekker, C. Motor proteins at work for nanotechnology. Science 317, 333–336 (2007).
Ray, S., Meyhofer, E., Milligan, R. A. & Howard, J. Kinesin follows the microtubule's protofilament axis. J. Cell Biol. 121, 1083–1093 (1993).
Yajima, J. & Cross, R. A. A torque component in the kinesin-1 power stroke. Nature Chem. Biol. 1, 338–341 (2005).
Gelles, J., Schnapp, B. J. & Sheetz, M. P. Tracking kinesin-driven movements with nanometre-scale precision. Nature 331, 450–453 (1988).
Svoboda, K., Schmidt, C. F., Schnapp, B. J. & Block, S. M. Direct observation of kinesin stepping by optical trapping interferometry. Nature 365, 721–727 (1993).
Seitz, A. & Surrey, T. Processive movement of single kinesins on crowded microtubules visualized using quantum dots. EMBO J. 25, 267–277 (2006).
Reck-Peterson, S. L. et al. Single-molecule analysis of dynein processivity and stepping behaviour. Cell 126, 335–348 (2006).
Vale, R. D. et al. Direct observation of single kinesin molecules moving along microtubules. Nature 380, 451–453 (1996).
Yildiz, A., Tomishige, M., Vale, R. D. & Selvin, P. R. Kinesin walks hand-over-hand. Science 303, 676–678 (2004).
Brunner, C., Wahnes, C. & Vogel, V. Cargo pick-up from engineered loading stations by kinesin driven molecular shuttles. Lab. Chip 7, 1263–1271 (2007).
Ramachandran, S., Ernst, K. H., Bachand, G. D., Vogel, V. & Hess, H. Selective loading of kinesin-powered molecular shuttles with protein cargo and its application to biosensing. Small 2, 330–334 (2006).
Block, S. M., Asbury, C. L., Shaevitz, J. W. & Lang, M. J. Probing the kinesin reaction cycle with a 2D optical force clamp. Proc. Natl Acad. Sci. USA 100, 2351–2356 (2003).
Chan, W. C. et al. Luminescent quantum dots for multiplexed biological detection and imaging. Curr. Opin. Biotechnol. 13, 40–46 (2002).
Mansson, A. et al. In vitro sliding of actin filaments labelled with single quantum dots. Biochem. Biophys. Res. Commun. 314, 529–534 (2004).
Bachand, G. D. et al. Assembly and transport of nanocrystal CdSe quantum dot nanocomposites using microtubules and kinesin motor proteins. Nano Lett. 4, 817–821 (2004).
Thompson, R. E., Larson, D. R. & Webb, W. W. Precise nanometre localization analysis for individual fluorescent probes. Biophys. J. 82, 2775–2783 (2002).
Lambacher, A. & Fromherz, P. Fluorescence interference-contrast microscopy on oxidized silicon using a monomolecular dye layer. Appl. Phys. A 63, 207–216 (1996).
Kerssemakers, J., Howard, J., Hess, H. & Diez, S. The distance that kinesin-1 holds its cargo from the microtubule surface measured by fluorescence interference contrast microscopy. Proc. Natl Acad. Sci. USA 103, 15812–15817 (2006).
Nishizaka, T., Yagi, T., Tanaka, Y. & Ishiwata, S. Right-handed rotation of an actin filament in an in vitro motile system. Nature 361, 269–271 (1993).
Mimori, Y. & Mikinoumura, T. Extrusion of rotating microtubules on the dynein-track from a microtubule–dynein gamma-complex. Cell Motil. Cytoskeleton 30, 17–25 (1995).
Vale, R. D. & Toyoshima, Y. Y. Rotation and translocation of microtubules in vitro induced by dyneins from Tetrahymena cilia. Cell 52, 459–469 (1988).
Walker, R. A., Salmon, E. D. & Endow, S. A. The Drosophila claret segregation protein is a minus-end directed motor molecule. Nature 347, 780–782 (1990).
Hyman, A. A., Chretien, D., Arnal, I. & Wade, R. H. Structural changes accompanying GTP hydrolysis in microtubules: information from a slowly hydrolyzable analogue guanylyl-(alpha, beta)-methylene-diphosphonate. J. Cell Biol. 128, 117–125 (1995).
Wang, Z. & Sheetz, M. P. The C-terminus of tubulin increases cytoplasmic dynein and kinesin processivity. Biophys. J. 78, 1955–1964 (2000).
Hancock, W. O. & Howard, J. Processivity of the motor protein kinesin requires two heads. J. Cell Biol. 140, 1395–1405 (1998).
Hunter, A. W. et al. The kinesin-related protein MCAK is a microtubule depolymerase that forms an ATP-hydrolyzing complex at microtubule ends. Mol. Cell 11, 445–457 (2003).
Andreu, J. M. et al. Low resolution structure of microtubules in solution. Synchrotron X-ray scattering and electron microscopy of taxol-induced microtubules assembled from purified tubulin in comparison with glycerol and MAP-induced microtubules. J. Mol. Biol. 226, 169–184 (1992).
Wade, R. H. & Chretien, D. Cryoelectron microscopy of microtubules. J. Struct. Biol. 110, 1–27 (1993).
Acknowledgements
The authors thank C. Bräuer, D. Naumburger and F. Friedrich for technical support, D. Zwicker and Y. Kalaidzidis for help with the tracking algorithms, G. Brouhard for advice on the subtilisin digestion of microtubules, C. Leduc, J. Kerssemakers, J. Helenius, S. Mashaghi and J. Howard for fruitful discussions, and C. Gell and T. Korten for comments on the manuscript. This work was supported by the German Federal Ministry of Education and Research (grant 03 N 8712) and the Max-Planck Society.
Author information
Authors and Affiliations
Contributions
B.N. and S.D. conceived and designed the experiments. B.N. performed the experiments. F.R. developed the nanometre tracking software. B.N. and F.R. analysed the data. All authors discussed the results. B.N. and S.D. co-wrote the paper.
Corresponding author
Rights and permissions
About this article
Cite this article
Nitzsche, B., Ruhnow, F. & Diez, S. Quantum-dot-assisted characterization of microtubule rotations during cargo transport. Nature Nanotech 3, 552–556 (2008). https://doi.org/10.1038/nnano.2008.216
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2008.216
This article is cited by
-
Motor generated torque drives coupled yawing and orbital rotations of kinesin coated gold nanorods
Communications Biology (2022)
-
Kinesin-14 motors drive a right-handed helical motion of antiparallel microtubules around each other
Nature Communications (2020)
-
Molecular dynamics simulation of the nanosecond pulsed electric field effect on kinesin nanomotor
Scientific Reports (2019)
-
Parallel mapping of optical near-field interactions by molecular motor-driven quantum dots
Nature Nanotechnology (2018)
-
Single Molecule Investigation of Kinesin-1 Motility Using Engineered Microtubule Defects
Scientific Reports (2017)