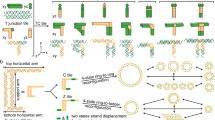
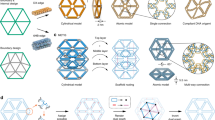
Abstract
Scaling up the complexity and diversity of synthetic molecular structures will require strategies that exploit the inherent stochasticity of molecular systems in a controlled fashion. Here we demonstrate a framework for programming random DNA tilings and show how to control the properties of global patterns through simple, local rules. We constructed three general forms of planar network—random loops, mazes and trees—on the surface of self-assembled DNA origami arrays on the micrometre scale with nanometre resolution. Using simple molecular building blocks and robust experimental conditions, we demonstrate control of a wide range of properties of the random networks, including the branching rules, the growth directions, the proximity between adjacent networks and the size distribution. Much as combinatorial approaches for generating random one-dimensional chains of polymers have been used to revolutionize chemical synthesis and the selection of functional nucleic acids, our strategy extends these principles to random two-dimensional networks of molecules and creates new opportunities for fabricating more complex molecular devices that are organized by DNA nanostructures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Rao, C. V., Wolf, D. M. & Arkin, A. P. Control, exploitation and tolerance of intracellular noise. Nature 420, 231–237 (2002).
Kærn, M., Elston, T. C., Blake, W. J. & Collins, J. J. Stochasticity in gene expression: from theories to phenotypes. Nat. Rev. Genet. 6, 451–464 (2005).
Eldar, A. & Elowitz, M. B. Functional roles for noise in genetic circuits. Nature 467, 167–173 (2010).
Losick, R. & Desplan, C. Stochasticity and cell fate. Science 320, 65–68 (2008).
Zipursky, S. L. & Sanes, J. R. Chemoaffinity revisited: dscams, protocadherins, and neural circuit assembly. Cell 143, 343–353 (2010).
Livet, J. et al. Transgenic strategies for combinatorial expression of fluorescent proteins in the nervous system. Nature 450, 56–62 (2007).
Adleman, L. M. Molecular computation of solutions to combinatorial problems. Science 266, 1021–1024 (1994).
Seeman, N. C. Nucleic acid junctions and lattices. J. Theor. Biol. 99, 237–247 (1982).
Rothemund, P. W. K. Folding DNA to create nanoscale shapes and patterns. Nature 440, 297–302 (2006).
Rinker, S., Ke, Y., Liu, Y., Chhabra, R. & Yan, H. Self-assembled DNA nanostructures for distance-dependent multivalent ligand–protein binding. Nat. Nanotech. 3, 418–422 (2008).
Schreiber, R. et al. Hierarchical assembly of metal nanoparticles, quantum dots and organic dyes using DNA origami scaffolds. Nat. Nanotech. 9, 74–78 (2014).
Pal, S., Deng, Z., Ding, B., Yan, H. & Liu, Y. DNA-origami-directed self-assembly of discrete silver-nanoparticle architectures. Angew. Chem. 122, 2760–2764 (2010).
Pal, S. et al. DNA directed self-assembly of anisotropic plasmonic nanostructures. J. Am. Chem. Soc. 133, 17606–17609 (2011).
Knudsen, J. B. et al. Routing of individual polymers in designed patterns. Nat. Nanotech. 10, 892–898 (2015).
Pinheiro, A. V., Han, D., Shih, W. M. & Yan, H. Challenges and opportunities for structural DNA nanotechnology. Nat. Nanotech. 6, 763–772 (2011).
Winfree, E., Liu, F., Wenzler, L. A. & Seeman, N. C. Design and self-assembly of two-dimensional DNA crystals. Nature 394, 539–544 (1998).
Yan, H., Park, S. H., Finkelstein, G., Reif, J. H. & LaBean, T. H. DNA-templated self-assembly of protein arrays and highly conductive nanowires. Science 301, 1882–1884 (2003).
Malo, J. et al. Engineering a 2D protein–DNA crystal. Angew. Chem. Int. Ed. 44, 3057–3061 (2005).
Zheng, J. et al. Two-dimensional nanoparticle arrays show the organizational power of robust DNA motifs. Nano Letters 6, 1502–1504 (2006).
Zheng, J. et al. From molecular to macroscopic via the rational design of a self-assembled 3D DNA crystal. Nature 461, 74–77 (2009).
Liu, W., Zhong, H., Wang, R. & Seeman, N. C. Crystalline two-dimensional DNA-origami arrays. Angew. Chem. 123, 278–281 (2011).
Woo, S. & Rothemund, P. W. K. Self-assembly of two-dimensional DNA origami lattices using cation-controlled surface diffusion. Nat. Commun. 5, 4889 (2014).
Aghebat Rafat, A., Pirzer, T. & Scheible, M. B., Kostina, A. & Simmel, F. C. Surface-assisted large-scale ordering of DNA origami tiles. Angew. Chem. Int. Ed. 53, 7665–7668 (2014).
Rothemund, P. W. K., Papadakis, N. & Winfree, E. Algorithmic self-assembly of DNA Sierpinski triangles. PLoS Biol. 2, e424 (2004).
Barish, R. D., Schulman, R., Rothemund, P. W. K. & Winfree, E. An information-bearing seed for nucleating algorithmic self-assembly. Proc. Natl Acad. Sci. USA 106, 6054–6059 (2009).
Schulman, R., Yurke, B. & Winfree, E. Robust self-replication of combinatorial information via crystal growth and scission. Proc. Natl Acad. Sci. USA 109, 6405–6410 (2012).
Woo, S. & Rothemund, P. W. K. Programmable molecular recognition based on the geometry of DNA nanostructures. Nat. Chem. 3, 620–627 (2011).
Gerling, T., Wagenbauer, K. F., Neuner, A. M. & Dietz, H. Dynamic DNA devices and assemblies formed by shape-complementary, non–base pairing 3D components. Science 347, 1446–1452 (2015).
Truchet, S. Mémoire sur les combinaisons. Mém. Acad. R. Sci. 1704, 363–372 (1704).
Smith, C. S. & Boucher, P. The tiling patterns of Sebastien Truchet and the topology of structural hierarchy. Leonardo 20, 373–385 (1987).
Murphy, C. J. et al. Structure and energetics of hydrogen-bonded networks of methanol on close packed transition metal surfaces. J. Chem. Phys. 141, 014701 (2014).
Less, J. R., Skalak, T. C., Sevick, E. M. & Jain, R. K. Microvascular architecture in a mammary carcinoma: branching patterns and vessel dimensions. Cancer Res. 51, 265–273 (1991).
Portmann, O., Vaterlaus, A. & Pescia, D. An inverse transition of magnetic domain patterns in ultrathin films. Nature 422, 701–704 (2003).
Blagodatski, A., Sergeev, A., Kryuchkov, M., Lopatina, Y. & Katanaev, V. L. Diverse set of Turing nanopatterns coat corneae across insect lineages. Proc. Natl Acad. Sci. USA 112, 10750–10755 (2015).
Metzger, R. J., Klein, O. D., Martin, G. R. & Krasnow, M. A. The branching programme of mouse lung development. Nature 453, 745–750 (2008).
Lefebvre, J. L., Kostadinov, D., Chen, W. V., Maniatis, T. & Sanes, J. R. Protocadherins mediate dendritic self-avoidance in the mammalian nervous system. Nature 488, 517–521 (2012).
Svitkina, T. M. & Borisy, G. G. Arp2/3 complex and actin depolymerizing factor/cofilin in dendritic organization and treadmilling of actin filament array in lamellipodia. J. Cell Biol. 145, 1009–1026 (1999).
Temperley, H. N. V. & Lieb, E. H. Relations between the ‘percolation’ and ‘colouring’ problem and other graph-theoretical problems associated with regular planar lattices: some exact results for the ‘percolation’ problem. Proc. R. Soc. Lond. A 322, 251–280 (1971).
Blöte, H. W. J. & Nienhuis, B. Fully packed loop model on the honeycomb lattice. Phys. Rev. Lett. 72, 1372–1375 (1994).
Nahum, A., Chalker, J. T., Serna, P., Ortuño, M. & Somoza, A. M. 3D loop models and the CP(n−1) sigma model. Phys. Rev. Lett. 107, 110601 (2011).
Rothemund, P. W. K. & Winfree, E. The program-size complexity of self-assembled squares. In Proc. 32nd Annual ACM Symp. Theory Computing 459–468 (Association for Computing Machinery, 2000).
Rajendran, A., Endo, M., Katsuda, Y., Hidaka, K. & Sugiyama, H. Programmed two-dimensional self-assembly of multiple DNA origami jigsaw pieces. ACS Nano 5, 665–671 (2011).
Zhao, Z., Liu, Y. & Yan, H. Organizing DNA origami tiles into larger structures using preformed scaffold frames. Nano Letters 11, 2997–3002 (2011).
Browne, C. Truchet curves and surfaces. Comput. Graph. 32, 268–281 (2008).
Marchi, A. N., Saaem, I., Vogen, B. N., Brown, S. & LaBean, T. H. Toward larger DNA origami. Nano Lett. 14, 5740–5747 (2014).
Hirokawa, N. Kinesin and dynein superfamily proteins and the mechanism of organelle transport. Science 279, 519–526 (1998).
Vale, R. D. The molecular motor toolbox for intracellular transport. Cell 112, 467–480 (2003).
Lund, K. et al. Molecular robots guided by prescriptive landscapes. Nature 465, 206–210 (2010).
Gu, H., Chao, J., Xiao, S. -J. & Seeman, N. C. A proximity-based programmable DNA nanoscale assembly line. Nature 465, 202–205 (2010).
Wickham, S. F. J. et al. A DNA-based molecular motor that can navigate a network of tracks. Nat. Nanotech. 7, 169–173 (2012).
Maune, H. T. et al. Self-assembly of carbon nanotubes into two-dimensional geometries using DNA origami templates. Nat. Nanotech. 5, 61–66 (2010).
Zhou, C., Duan, X. & Liu, N. A plasmonic nanorod that walks on DNA origami. Nat. Commun. 6, 8102 (2015).
Gordon, E. M., Barrett, R. W., Dower, W. J., Fodor, S. P. A. & Gallop, M. A. Applications of combinatorial technologies to drug discovery. 2. Combinatorial organic synthesis, library screening strategies, and future directions. J. Med. Chem. 37, 1385–1401 (1994).
Ellington, A. D. & Szostak, J. W. In vitro selection of RNA molecules that bind specific ligands. Nature 346, 818–822 (1990).
Acknowledgements
We thank S. Wang for designing the fish and gear tile (Supplementary Fig. 38) and J. Parkin, A. Karan and S. Wang for designing and creating a heart shape that is self-assembled from square DNA origami tiles (Supplementary Fig. 68). We thank E. Winfree, P. Rothemund, D. Soloveichik and A. Condon for critique on the manuscript. G.T. was supported by an NSF grant (1317694). P.P. was supported by a NIH/NRSA training grant (5 T32 GM07616). L.Q. was supported by a Career Award at the Scientific Interface from the Burroughs Wellcome Fund (1010684) and a Faculty Early Career Development Award from the NSF (1351081).
Author information
Authors and Affiliations
Contributions
G.T. and P.P. performed the experiments and analysed the data. P.P. performed the simulations and developed the software tools. G.T., P.P. and L.Q. designed the systems and wrote the manuscript. L.Q. initiated and guided the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 11513 kb)
Rights and permissions
About this article
Cite this article
Tikhomirov, G., Petersen, P. & Qian, L. Programmable disorder in random DNA tilings. Nature Nanotech 12, 251–259 (2017). https://doi.org/10.1038/nnano.2016.256
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2016.256
This article is cited by
-
Synthetic molecular switches driven by DNA-modifying enzymes
Nature Communications (2024)
-
Barcoded DNA origami structures for multiplexed optimization and enrichment of DNA-based protein-binding cavities
Nature Chemistry (2020)
-
DNA Nanotechnology-based Biocomputing
Chemical Research in Chinese Universities (2020)
-
Programming DNA origami patterning with non-canonical DNA-based metallization reactions
Nature Communications (2019)
-
Information-based autonomous reconfiguration in systems of interacting DNA nanostructures
Nature Communications (2018)