Abstract
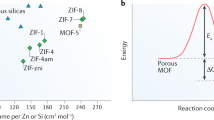
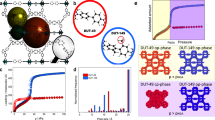
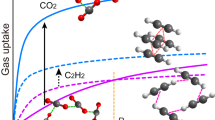
The selective capture of carbon dioxide in porous materials has potential for the storage and purification of fuel and flue gases. However, adsorption capacities under dynamic conditions are often insufficient for practical applications, and strategies to enhance CO2–host selectivity are required. The unique partially interpenetrated metal–organic framework NOTT-202 represents a new class of dynamic material that undergoes pronounced framework phase transition on desolvation. We report temperature-dependent adsorption/desorption hysteresis in desolvated NOTT-202a that responds selectively to CO2. The CO2 isotherm shows three steps in the adsorption profile at 195 K, and stepwise filling of pores generated within the observed partially interpenetrated structure has been modelled by grand canonical Monte Carlo simulations. Adsorption of N2, CH4, O2, Ar and H2 exhibits reversible isotherms without hysteresis under the same conditions, and this allows capture of gases at high pressure, but selectively leaves CO2 trapped in the nanopores at low pressure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Magudeswaran, P. N., George, S. & John, J. Reduction of global warming gas emissions from the manufacture of portland cement using high volume fly ash concrete. Nature Environ. Pollut. Technol. 6, 495–497 (2007).
Davis, M. E. Ordered porous materials for emerging applications. Nature 417, 813–821 (2002).
Long, J. R. & Yaghi, O. M. (eds) Special issue on metal–organic frameworks. Chem. Soc. Rev. 38, 1201–1507 (2009).
Banerjee, R. et al. High-throughput synthesis of zeolitic imidazolate frameworks and application to CO2 capture. Science 319, 939–943 (2008).
Sumida, K. et al. Hydrogen and carbon dioxide capture in an iron-based sodalite-type metal–organic framework (FE-BTT) discovered via high-throughput methods. Chem. Sci. 1, 184–191 (2010).
Millward, A. R. & Yaghi, O. M. Metal-organic frameworks with exceptionally high capacity for storage of carbon dioxide at room temperature. J. Am. Chem. Soc. 127, 17998–17999 (2005).
Llewellyn, P. L. et al. High uptakes of CO2 and CH4 in mesoporous metal–organic frameworks MIL-100 and MIL-101. Langmuir 24, 7245–7250 (2008).
Furukawa, H. et al. Ultrahigh porosity in metal–organic frameworks. Science 329, 424–428 (2010).
Britt, R. D., Furukawa, H., Wang, B., Glover, T. G. & Yaghi, O. M. Highly efficient separation of carbon dioxide by a metal–organic framework replete with open metal sites. Proc. Natl Acad. Sci. USA 106, 20637–20640 (2009).
Vaidhyanathan, R. et al. Direct observation and quantification of CO2 binding within an amine-functionalised nanoporous solid. Science 330, 650–653 (2010).
Demessence, A., D’Alessandro, D. M., Foo, M. L. & Long, J. R. Strong CO2 binding in a water-stable, triazolate-bridged metal–organic framework functionalized with ethylenediamine. J. Am. Chem. Soc. 131, 8784–8786 (2009).
Dietzel, P. D. C., Besikiotis, V. & Blom, R. Application of metal–organic frameworks with coordinatively unsaturated metal sites in storage and separation of methane and carbon dioxide. J. Mater. Chem. 19, 7362–7370 (2009).
Dybtsev, D. N., Chun, H., Yoon, S. H., Kim, D. & Kim, K. Microporous manganese formate: A simple metal–organic porous material with high framework stability and highly selective gas sorption properties. J. Am. Chem. Soc. 126, 32–33 (2004).
Shimomura, S. et al. Selective sorption of oxygen and nitric oxide by an electron-donating flexible porous coordination polymer. Nature Chem. 2, 633–637 (2010).
Llewellyn, P. L., Bourrelly, S., Serre, C., Filinchuk, Y. & Férey, G. How hydration drastically improves adsorption selectivity for CO2 over CH4 in the flexible chromium terephthalate MIL-53. Angew. Chem. Int. Ed. 45, 7751–7754 (2006).
Walton, K. S. et al. Understanding inflections and steps in carbon dioxide adsorption isotherms in metal–organic frameworks. J. Am. Chem. Soc. 130, 406–407 (2008).
Thallapally, P. K. et al. Gas-induced transformation and expansion of a non-porous organic solid. Nature Mater. 7, 146–150 (2008).
Férey, G. & Serre, C. Large breathing effects in three-dimensional porous hybrid matter: Facts, analyses, rules and consequences. Chem. Soc. Rev. 38, 1380–1399 (2009).
Thallapally, P. K. et al. Flexible (breathing) interpenetrated metal–organic frameworks for CO2 separation applications. J. Am. Chem. Soc. 130, 16842–16843 (2008).
Wang, Z. & Cohen, S. M. Modulating metal–organic frameworks to breathe: A postsynthetic covalent modification approach. J. Am. Chem. Soc. 131, 16675–16677 (2009).
Seo, J., Matsuda, R., Sakamoto, H., Bonneau, C. & Kitagawa, S. A pillared-layer coordination polymer with a rotatable pillar acting as a molecular gate for guest molecules. J. Am. Chem. Soc. 131, 12792–12800 (2009).
Choi, H-S. & Suh, M. P. Highly selective CO2 capture in flexible 3D coordination polymer networks. Angew. Chem. Int. Ed. 48, 6865–6869 (2009).
Rabone, J. et al. An adaptable peptide-based porous material. Science 329, 1053–1057 (2010).
Tanaka, D. et al. Preparation of flexible porous coordination polymer nanocrystals with accelerated guest adsorption kinetics. Nature Chem. 2, 410–416 (2010).
Spek, A. L. Single-crystal structure validation with the program PLATON. J. Appl. Crystallogr. 36, 7–13 (2003).
Yaghi, O. M. Metal-organic frameworks: A tale of two entanglements. Nature Mater. 6, 92–93 (2007).
Yang, S. et al. Cation-induced kinetic trapping and enhanced hydrogen adsorption in a modulated anionic metal–organic framework. Nature Chem. 1, 487–493 (2009).
Chen, B., Eddaoudi, M., Hyde, S. T., O’Keefe, M. & Yaghi, O. M. Interwoven metal–organic framework on a periodic minimal surface with extra-large pores. Science 291, 1021–1023 (2001).
Chen, B. et al. Rationally designed micropores within a metal–organic framework for selective sorption of gas molecules. Inorg. Chem. 46, 1233–1236 (2007).
Chen, B., Ma, S., Hurtado, E. J., Lobkovsky, E. B. & Zhou, H. C. A triply interpenetrated microporous metal–organic framework for selective sorption of gas molecules. Inorg. Chem. 14, 8490–8492 (2007).
Zhang, J., Wojtas, L., Larsen, R. W., Eddaoudi, M. & Zaworotko, M. J. Temperature and concentration control over interpenetration in a metal–organic material. J. Am. Chem. Soc. 131, 17040–17041 (2009).
Shekhah, O. et al. Controlling interpenetration in metal–organic frameworks by liquid-phase epitaxy. Nature Mater. 8, 481–484 (2009).
Farha, O. K., Malliakas, C. D., Kanatzidis, M. G. & Hupp, J. T. Control over catenation in metal–organic frameworks via rational design of the organic building block. J. Am. Chem. Soc. 132, 950–952 (2010).
Lin, X. et al. High capacity hydrogen adsorption in Cu(II) tetracarboxylate framework materials: The role of pore size, ligand functionalization, and exposed metal sites. J. Am. Chem. Soc. 131, 2159–2171 (2009).
Zheng, S. et al. Porous indium-organic frameworks and systematization of structural building blocks. Angew. Chem. Int. Ed. 123, 9020–9024 (2011).
Takamizawa, S. & Nakata, E. I. Direct observation of H2 adsorbed state within a porous crystal by single crystal X-ray diffraction analysis. Cryst. Eng. Commun. 7, 476–479 (2005).
Takamizawa, S., Nakata, E. I., Yokoyama, H., Mochizuki, K. & Mori, W. Carbon dioxide inclusion phase of a transformable 1D coordination polymer host [Rh2(O2CPh)4(pyz)]n . Angew. Chem. Int. Ed. 42, 4331–4334 (2003).
Mulfort, K. L., Farha, O. K., Malliakas, C. D., Kanatzidis, M. G. & Hupp, J. T. An interpenetrated framework material with hysteretic CO2 uptake. Chem. Eur. J. 16, 276–281 (2010).
Horike, S., Shimomura, S. & Kitagawa, S. Soft porous crystals. Nature Chem. 1, 695–704 (2009).
Yuan, D., Getman, R. B., Wei, Z., Snurr, R. Q. & Zhou, H-C. Stepwise adsorption in a mesoporous metal–organic framework: Experimental and computational analysis. Chem. Commun. 22, 10228–10234 (2012).
An, J., Geib, S. J. & Rosi, N. L. High and selective CO2 uptake in a cobalt adeninate metal–organic framework exhibiting pyrimidine- and amino-decorated pores. J. Am. Chem. Soc. 132, 38–39 (2010).
Banerjee, R. et al. Control of pore size and functionality in isoreticular zeolitic imidazolate frameworks and their carbon dioxide selective capture properties. J. Am. Chem. Soc. 131, 3875–3877 (2009).
Reid, C. R. & Thomas, K. M. Adsorption of gases on a carbon molecular sieve used for air separation: Linear adsorptives as probes for kinetic selectivity. Langmuir 15, 3206–3218 (1999).
Acknowledgements
S.Y. thanks EPSRC for a PhD Plus Fellowship and the Leverhulme Trust for an Early Career Fellowship. We thank EPSRC (UKSHEC) and the University of Nottingham for support and financial support of X-ray equipment, STFC for awarding access to Station 9.8 of the Daresbury Synchrotron Radiation Source, Diamond Light Source for beam time on Beamlines I11 and I19, and J. Potter for technical help at Diamond Beamline I11. We thank J. Sun (University of Stockholm) for helpful discussions on powder diffraction and A. Linden (University of Zürich) for discussions on the structural analysis of NOTT-202. N.R.C. gratefully acknowledges receipt of a Royal Society Leverhulme Trust Senior Research Fellowship. E.B. gratefully acknowledges financial support from an EPSRC Career Acceleration Fellowship (EP/G005060). M. Schröder gratefully acknowledges receipt of a Royal Society Wolfson Merit Award and an ERC Advanced Grant.
Author information
Authors and Affiliations
Contributions
S.Y., X.L., K.M.T., M. Schröder: syntheses, characterization, measurements and analysis of adsorption isotherms. E.B. and M. Suyetin: grand canonical Monte Carlo modelling. S.Y., A.J.B., C.C.T. and J.E.P.: synchrotron X-ray powder data analysis. S.Y., A.J.B., W.L., D.R.A. and P.J.R.: single-crystal X-ray structural data analyses. S.Y., A.J.B., P.H., N.R.C., K.M.T. and M. Schröder: overall design, direction and supervision of project. All authors contributed to the writing of the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 4358 kb)
Supplementary information
Crystallographic information for NOTT-202 at 120K (CIF 17 kb)
Supplementary information
Crystallographic information for NOTT-202a at 120K (CIF 24 kb)
Supplementary information
Crystallographic information for NOTT-202a at 150K (CIF 24 kb)
Supplementary information
Crystallographic information for NOTT-202a at 180K (CIF 23 kb)
Supplementary information
Crystallographic information for NOTT-202a at 200K (CIF 24 kb)
Supplementary information
Crystallographic information for NOTT-202a at 220K (CIF 23 kb)
Supplementary information
Crystallographic information for NOTT-202a at 260K (CIF 24 kb)
Rights and permissions
About this article
Cite this article
Yang, S., Lin, X., Lewis, W. et al. A partially interpenetrated metal–organic framework for selective hysteretic sorption of carbon dioxide. Nature Mater 11, 710–716 (2012). https://doi.org/10.1038/nmat3343
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat3343
This article is cited by
-
Hetero-interpenetrated metal–organic frameworks
Nature Chemistry (2023)
-
Adaptive response of a metal–organic framework through reversible disorder–disorder transitions
Nature Chemistry (2021)
-
Anisotropic reticular chemistry
Nature Reviews Materials (2020)
-
Diverse crystal size effects in covalent organic frameworks
Nature Communications (2020)
-
Regulation of the Degree of Interpenetration in Metal–Organic Frameworks
Topics in Current Chemistry (2020)