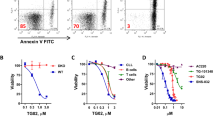
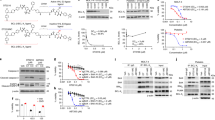
Abstract
Proteins in the B cell CLL/lymphoma 2 (BCL-2) family are key regulators of the apoptotic process. This family comprises proapoptotic and prosurvival proteins, and shifting the balance toward the latter is an established mechanism whereby cancer cells evade apoptosis. The therapeutic potential of directly inhibiting prosurvival proteins was unveiled with the development of navitoclax, a selective inhibitor of both BCL-2 and BCL-2–like 1 (BCL-XL), which has shown clinical efficacy in some BCL-2–dependent hematological cancers. However, concomitant on-target thrombocytopenia caused by BCL-XL inhibition limits the efficacy achievable with this agent. Here we report the re-engineering of navitoclax to create a highly potent, orally bioavailable and BCL-2–selective inhibitor, ABT-199. This compound inhibits the growth of BCL-2–dependent tumors in vivo and spares human platelets. A single dose of ABT-199 in three patients with refractory chronic lymphocytic leukemia resulted in tumor lysis within 24 h. These data indicate that selective pharmacological inhibition of BCL-2 shows promise for the treatment of BCL-2–dependent hematological cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hanahan, D. & Weinberg, R.A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Adams, J.M. & Cory, S. The Bcl-2 apoptotic switch in cancer development and therapy. Oncogene 26, 1324–1337 (2007).
Youle, R.J. & Strasser, A. The BCL-2 protein family: opposing activities that mediate cell death. Nat. Rev. Mol. Cell Biol. 9, 47–59 (2008).
Tsujimoto, Y., Cossman, J., Jaffe, E. & Croce, C.M. Involvement of the bcl-2 gene in human follicular lymphoma. Science 228, 1440–1443 (1985).
Cleary, M.L., Smith, S.D. & Sklar, J. Cloning and structural analysis of cDNAs for bcl-2 and a hybrid bcl-2/immunoglobulin transcript resulting from the t(14;18) translocation. Cell 47, 19–28 (1986).
Boise, L.H. et al. Bcl-x, a bcl-2–related gene that functions as a dominant regulator of apoptotic cell death. Cell 74, 597–608 (1993).
Vaux, D.L., Cory, S. & Adams, J.M. Bcl-2 gene promotes haemopoietic cell survival and cooperates with c-myc to immortalize pre-B cells. Nature 335, 440–442 (1988).
Huang, J.Z. et al. The t(14;18) defines a unique subset of diffuse large B-cell lymphoma with a germinal center B-cell gene expression profile. Blood 99, 2285–2290 (2002).
Minn, A.J., Rudin, C.M., Boise, L.H. & Thompson, C.B. Expression of bcl-xl can confer a multidrug resistance phenotype. Blood 86, 1903–1910 (1995).
Amundson, S.A. et al. An informatics approach identifying markers of chemosensitivity in human cancer cell lines. Cancer Res. 60, 6101–6110 (2000).
Zhang, H. et al. Bcl-2 family proteins are essential for platelet survival. Cell Death Differ. 14, 943–951 (2007).
Mason, K.D. et al. Programmed anuclear cell death delimits platelet life span. Cell 128, 1173–1186 (2007).
Oltersdorf, T. et al. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Nature 435, 677–681 (2005).
Park, C.M. et al. Discovery of an orally bioavailable small molecule inhibitor of prosurvival B-cell lymphoma 2 proteins. J. Med. Chem. 51, 6902–6915 (2008).
Tse, C. et al. ABT-263: a potent and orally bioavailable Bcl-2 family inhibitor. Cancer Res. 68, 3421–3428 (2008).
Wilson, W.H. et al. Navitoclax, a targeted high-affinity inhibitor of BCL-2, in lymphoid malignancies: a phase 1 dose-escalation study of safety, pharmacokinetics, pharmacodynamics, and antitumour activity. Lancet Oncol. 11, 1149–1159 (2010).
Roberts, A.W. et al. Substantial susceptibility of chronic lymphocytic leukemia to BCL2 inhibition: results of a phase I study of navitoclax in patients with relapsed or refractory disease. J. Clin. Oncol. 30, 488–496 (2012).
Gandhi, L. et al. Phase I study of Navitoclax (ABT-263), a novel Bcl-2 family inhibitor, in patients with small-cell lung cancer and other solid tumors. J. Clin. Oncol. 29, 909–916 (2011).
Petros, A.M. et al. Solution structure of the antiapoptotic protein bcl-2. Proc. Natl. Acad. Sci. USA 98, 3012–3017 (2001).
Muchmore, S.W. et al. X-ray and NMR structure of human Bcl-xL, an inhibitor of programmed cell death. Nature 381, 335–341 (1996).
Wendt, M.D. Discovery of ABT-263, a Bcl-family protein inhibitor: observations on targeting a large protein-protein interaction. Expert Opin. Drug. Discov. 3, 1123–1143 (2008).
Sattler, M. et al. Structure of Bcl-xL-Bak peptide complex: recognition between regulators of apoptosis. Science 275, 983–986 (1997).
Lee, E.F. et al. Crystal structure of ABT-737 complexed with Bcl-xL: implications for selectivity of antagonists of the Bcl-2 family. Cell Death Differ. 14, 1711–1713 (2007).
Atwell, S., Ultsch, M., De Vos, A.M. & Wells, J.A. Structural plasticity in a remodeled protein-protein interface. Science 278, 1125–1128 (1997).
Wendt, M.D. et al. Discovery and structure-activity relationship of antagonists of B-cell lymphoma 2 family proteins with chemopotentiation activity in vitro and in vivo. J. Med. Chem. 49, 1165–1181 (2006).
Bruncko, M. et al. Studies leading to potent, dual inhibitors of Bcl-2 and Bcl-xL. J. Med. Chem. 50, 641–662 (2007).
Harada, H., Quearry, B., Ruiz-Vela, A. & Korsmeyer, S.J. Survival factor–induced extracellular signal-regulated kinase phosphorylates BIM, inhibiting its association with BAX and proapoptotic activity. Proc. Natl. Acad. Sci. USA 101, 15313–15317 (2004).
Del Gaizo Moore, V., Schlis, K.D., Sallan, S.E., Armstrong, S.A. & Letai, A. BCL-2 dependence and ABT-737 sensitivity in acute lymphoblastic leukemia. Blood 111, 2300–2309 (2008).
Shoemaker, A.R. et al. Activity of the Bcl-2 family inhibitor ABT-263 in a panel of small cell lung cancer xenograft models. Clin. Cancer Res. 14, 3268–3277 (2008).
Singh, R.R. et al. Hedgehog signaling pathway is activated in diffuse large B-cell lymphoma and contributes to tumor cell survival and proliferation. Leukemia 24, 1025–1036 (2010).
Ackler, S. et al. Navitoclax (ABT-263) and bendamustine ± rituximab induce enhanced killing of non-Hodgkin's lymphoma tumors in vivo. Br. J. Pharmacol. 167, 881–891 (2012).
Marschitz, I. Analysis of Bcl-2 protein expression in chronic lymphocytic leukemia. Am. J. Clin. Pathol. 113, 219–229 (2000).
Samuel, S. et al. VSV oncolysis in combination with the BCL-2 inhibitor obatoclax overcomes apoptosis resistance in chronic lymphocytic leukemia. Mol. Ther. 18, 2094–2103 (2010).
Rampello, E., Fricia, T. & Malaguarnera, M. The management of tumor lysis syndrome. Nat. Clin. Pract. Oncol. 3, 438–447 (2006).
American Cancer Society. Cancer Facts & Figures 2011. <http://www.cancer.org/acs/groups/content/@epidemiologysurveilance/documents/document/acspc-029771.pdf> (2011).
Sawas, A., Diefenbach, C. & O'Connor, O.A. New therapeutic targets and drugs in non-Hodgkin's lymphoma. Curr. Opin. Hematol. 18, 280–287 (2011).
Bea, S. et al. Diffuse large B-cell lymphoma subgroups have distinct genetic profiles that influence tumor biology and improve gene-expression based survival prediction. Blood 106, 3183–3190 (2005).
Iqbal, J. et al. BCL2 expression is a prognostic marker for the activated B-cell–like type of diffuse large B-cell lymphoma. J. Clin. Oncol. 24, 961–968 (2006).
Irish, J.M. et al. Flt3 Y591 duplication and Bcl-2 overexpression are detected in acute myeloid leukemia cells with high levels of phosphorylated wild-type p53. Blood 109, 2589–2596 (2007).
Coustan-Smith, E. et al. Clinical relevance of BCL-2 overexpression in childhood acute lymphoblastic leukemia. Blood 87, 1140–1146 (1996).
Campàs, C. et al. Bcl-2 inhibitors induce apoptosis in chronic lymphocytic leukemia cells. Exp. Hematol. 34, 1663–1669 (2006).
Cimmino, A. et al. MiR-15 and miR-16 induce apoptosis by targeting BCL2. Proc. Natl. Acad. Sci. USA 102, 13944–13949 (2005).
Calin, G.A. et al. MiR-15a and miR-16–1 cluster functions in human leukemia. Proc. Natl. Acad. Sci. USA 105, 5166–5171 (2008).
Matsumura, N. et al. mRNA display selection of a high-affinity, Bcl-X(L)-specific binding peptide. FASEB J. 24, 2201–2210 (2010).
Roberts, A.W. et al. Selective inhibition of BCL-2 is active against chronic lymphocytic leukemia (CLL): first clinical experience with the BH3-mimetic ABT-199. Abstract 546 (European Hematology Association 2012, Amsterdam, The Netherlands, June 14–17, 2012).
La Thangue, N.B. & Kerr, D.J. Predictive biomarkers: a paradigm shift towards personalized cancer medicine. Nat. Rev. Clin. Oncol. 8, 587–596 (2011).
Zhang, H., Nimmer, P., Rosenberg, S.H., Ng, S.C. & Joseph, M. Development of a high-throughput fluorescence polarization assay for Bcl-x(L). Anal. Biochem. 307, 70–75 (2002).
Chen, J. et al. The Bcl-2/Bcl-X(L)/Bcl-w inhibitor, navitoclax, enhances the activity of chemotherapeutic agents in vitro and in vivo. Mol. Cancer Ther. 10, 2340–2349 (2011).
Phillips, D.C., Garrison, S.P., Jeffers, J.R. & Zambetti, G.P. Assays to measure p53-dependent and -independent apoptosis. Methods Mol. Biol. 559, 143–159 (2009).
Mason, K.D. et al. The BH3 mimetic compound, ABT-737, synergizes with a range of cytotoxic chemotherapy agents in chronic lymphocytic leukemia. Leukemia 23, 2034–2041 (2009).
Acknowledgements
We thank M. Bruncko, E. Fry, L. Hasvold, L. Hexamer, A. Kunzer, A. Petros, X. Song, Z. Tao, L. Wang and X. Wang for contributions to the generation of ABT-199 and related analogs and J. Bouska, and D. Osterling for analytical support. The authors acknowledge G. Chiang and A. Vasudevan for critical reading of this manuscript, L. Belmont, I. Wertz, J. Adams, S. Cory, P. Colman, P. Czabotar and G. Lessene for useful discussions and K. Lowes, E. Litvinovich and L. Roberts for technical analysis. Research performed at the Walter and Eliza Hall Institute (WEHI) was supported by grants and fellowships from the Australian National Health and Medical Research Council (NHMRC, including an Independent Research Institutes Infrastructure Support Scheme (IRIISS) grant), the Australian Cancer Research Foundation, the Leukaemia Foundation of Australia, the Cancer Council of Victoria, the Victorian Cancer Agency, the Victorian State Government Operational Infrastructure Support and the Leukemia Lymphoma Society. The authors acknowledge L.M. Staudt (US National Institutes of Health) for DLBCL cell lines.
Author information
Authors and Affiliations
Contributions
A.J.S., S.W.E., S.H.R., S.G.H., W.J.F., D.C.S.H., J.D.L. and C.T. directed aspects of the preclinical research. A.J.S. and J.D.L. interpreted data and wrote the manuscript. A.W.R., S.H.E., J.F.S. and R.A.H. directed the clinical trial design, generated patient data and interpreted results, and A.W.R. contributed in writing the manuscript. S.L.K. generated in vitro CLL experiments. K.D.M. generated in vitro normal volunteer platelet data. M.J.M., E.R.B., S.L.A., D.S., H.L.M., J.L. and A.O. generated in vivo pharmacology data, and E.R.B. contributed in writing of the manuscript. M.D.W., C.-M.P., G.M.S., H.D. and A.J.S. conceived of or generated the compounds described in the manuscript. C.H.P. generated the X-ray structures. J.D.L., D.C.P., S.J., S.K.T., J.C., J.C.X., P.M.N., Y.X., H.Z., P.J.K., C.T., L.T.L. and M.L.S. performed the biological characterization of ABT-199 and other compounds described in the manuscript. N.D.C., B.D.D. and K.C.M. generated the pharmacokinetic and platelet data in conscious dogs.
Corresponding author
Ethics declarations
Competing interests
A.J.S., J.D.L., E.R.B., S.L.A., N.D.C., J.C., H.D., S.H.E., S.J., P.J.K., L.T.L., K.C.M., M.J.M., P.M.N., A.O., C.H.P., D.C.P., M.L.S., G.M.S., S.K.T., C.T., M.D.W., Y.X., J.C.X., H.Z., R.A.H., S.H.R. and S.W.E. are employees and stockholders of Abbott Laboratories. W.J.F., S.G.H., H.L.M. and D.S. are employees of Genentech, Inc., a member of the Roche group, and are stockholders of Roche Holding, AG. D.C.S.H., S.L.K., K.D.M. and A.W.R. are employees of Walter and Eliza Hall Institute, which receives commercial income and research funding from Genentech, Inc. and Abbott Laboratories.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Tables 1–6 (PDF 318 kb)
Rights and permissions
About this article
Cite this article
Souers, A., Leverson, J., Boghaert, E. et al. ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat Med 19, 202–208 (2013). https://doi.org/10.1038/nm.3048
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3048
This article is cited by
-
Venetoclax efficacy on acute myeloid leukemia is enhanced by the combination with butyrate
Scientific Reports (2024)
-
Protein-based bandpass filters for controlling cellular signaling with chemical inputs
Nature Chemical Biology (2024)
-
MicroRNA miR-20a-5p targets CYCS to inhibit apoptosis in hepatocellular carcinoma
Cell Death & Disease (2024)
-
Structure-based drug design and molecular dynamics studies of an allosteric modulator targeting the protein–protein interaction site of PDK1
Journal of Molecular Modeling (2024)
-
Identification of potential oral cancer drugs as Bcl-2 inhibitors from known anti-neoplastic agents through docking studies
Journal of Mathematical Chemistry (2024)