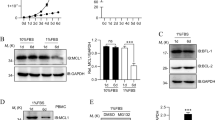
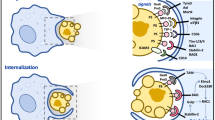
Abstract
T lymphocyte death is essential for proper function of the immune system. During the decline of an immune response, most of the activated T cells die. Cell death is also responsible for eliminating autoreactive lymphocytes. Although recent studies have focused on caspase-dependent apoptotic signals, much evidence now shows that caspase- independent, necrotic cell death pathways are as important. An understanding of the molecular control of these alternative pathways is beginning to emerge. Damage of organelles including mitochondria, endoplasmic reticulum or lysozymes, leading to an increase in calcium and reactive oxygen species and the release of effector proteins, is frequently involved in caspase-independent cell death.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

C. C.

K. R.

C. C.

K. R.
Similar content being viewed by others
References
Kerr, J.F.R., Wyllie, A.H. & Currie, A.R. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br. J. Cancer 26, 239–257 (1972).
Strasser, A., O'Connor, L. & Dixit, V.M. Apoptosis signaling. Annu. Rev. Biochem. 69, 217–245 (2000).
Kaufmann, S.H. & Hengartner, M.O. Programmed cell death: alive and well in the new millennium. Trends Cell Biol. 11, 526–534 (2001).
Leist, M. & Jäättelä, M. Four deaths and a funeral: from caspases to alternative mechanisms. Nat. Rev. Mol. Cell Biol. 2, 589–598 (2001).
Lockshin, R.A. & Zakeri, Z. Caspase-independent cell deaths. Curr. Opin. Cell Biol. 14, 727–733 (2002).
Zeuner, A., Eramo, A., Peschle, C. & De Maria, R. Caspase activation without death. Cell Death Differ. 6, 1075–1080 (1999).
Samali, A., Zhivotovsky, B., Jones, D., Nagata, S. & Orrenius, S. Apoptosis: cell death defined by caspase activation. Cell Death Differ. 6, 495–496 (1999).
Lockshin, R.A. & Zakeri, Z. Programmed cell death and apoptosis: origins of the theory. Nat. Rev. Mol. Cell Biol. 2, 545–550 (2001).
Susin, S.A. et al. Molecular characterization of mitochondrial apoptosis-inducing factor. Nature 397, 441–446 (1999).
Holler, N. et al. Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nat. Immunol. 1, 489–495 (2000).
Vercammen, D. et al. Dual signaling of the Fas receptor: initiation of both apoptotic and necrotic cell death pathways. J. Exp. Med. 188, 919–930 (1998).
Chi, S. et al. Oncogenic Ras triggers cell suicide through the activation of a caspase-independent cell death program in human cancer cells. Oncogene 18, 2281–2290 (1999).
Elliott, K., Ge, K., Du, W. & Prendergast, G.C. The c-Myc–interacting adaptor protein Bin1 activates a caspase-independent cell death program. Oncogene 19, 4669–4684 (2000).
Tolkovsky, A.M., Xue, L., Fletcher, G.C. & Borutaite, V. Mitochondrial disappearance from cells: a clue to the role of autophagy in programmed cell death and disease? Biochimie 84, 233–240 (2002).
Schulze-Osthoff, K. et al. Cytotoxic activity of tumor necrosis factor is mediated by early damage of mitochondrial functions. Evidence for the involvement of mitochondrial radical generation. J. Biol. Chem. 267, 5317–5323 (1992).
Vercammen, D. et al. Inhibition of caspases increases the sensitivity of L929 cells to necrosis mediated by tumor necrosis factor. J. Exp. Med. 187, 1477–1485 (1998).
Ha, H.C. & Snyder, S.H. Poly(ADP-ribose) polymerase is a mediator of necrotic cell death by ATP depletion. Proc. Natl. Acad. Sci. USA 96, 13978–13982 (1999).
Saeki, K. et al. Bcl-2 down-regulation causes autophagy in a caspase-independent manner in human leukemic HL60 cells. Cell Death Differ. 7, 1263–1269 (2000).
Denecker, G. et al. Death receptor-induced apoptotic and necrotic cell death: differential role of caspases and mitochondria. Cell Death Differ. 8, 829–840 (2001).
Chung, S., Gumienny, T.L., Hengartner, M.O. & Driscoll, M. A common set of engulfment genes mediates removal of both apoptotic and necrotic cell corpses in C. elegans. Nat. Cell Biol. 2, 931–937 (2000).
Hirt, U.A., Gantner, F. & Leist, M. Phagocytosis of nonapoptotic cells dying by caspase-independent mechanisms. J. Immunol. 164, 6520–6529 (2000).
Nylandsted, J. et al. Eradication of glioblastoma, and breast and colon carcinoma xenografts by hsp70 depletion. Cancer Res. 62, 7139–7142 (2002).
Ferri, K.R. & Kroemer, G. Organelle-specific initiation of cell death pathways. Nat. Cell Biol. 3, E255–E263 (2001).
Mathiasen, I.S. & Jäättelä, M. Triggering caspase-independent cell death to combat cancer. Trends Mol. Med. 8, 212–220 (2002).
Von Ahsen, O., Waterhouse, N.J., Kuwana, T., Newmeyer, D.D. & Green, D.R. The 'harmless' release of cytochrome c. Cell Death Differ. 7, 1192–1199 (2000).
Lipton, S.A. & Bossy-Wetzel, E. Dueling activities of AIF in cell death versus survival: DNA binding and redox activity. Cell 111, 147–150 (2002).
Joza, N. et al. Essential role of the mitochondrial apoptosis-inducing factor in programmed cell death. Nature 410, 549–554 (2001).
Cregan, S.P. et al. Apoptosis-inducing factor is involved in the regulation of caspase-independent neuronal cell death. J. Cell Biol. 158, 507–517 (2002).
Yu, S.W. et al. Mediation of poly(ADP-ribose) polymerase-1–dependent cell death by apoptosis-inducing factor. Science 297, 259–263 (2002).
Joseph, B. et al. Mitochondrial dysfunction is an essential step for killing of non-small cell lung carcinomas resistant to conventional treatment. Oncogene 21, 65–77 (2002).
Li, L.Y., Luo, X. & Wang, X. Endonuclease G is an apoptotic DNase when released from mitochondria. Nature 412, 95–99 (2001).
Suzuki, Y. et al. A serine protease, htra2, is released from the mitochondria and interacts with xiap, inducing cell death. Mol. Cell 8, 613–621 (2001).
Wang, X., Yang, C., Chai, J., Shi, Y. & Xue, D. Mechanisms of AIF-mediated apoptotic DNA degradation in Caenorhabditis elegans. Science 298, 1587–1592 (2002).
Elmore, S.P., Qian, T., Grissom, S.F. & Lemasters, J.J. The mitochondrial permeability transition initiates autophagy in rat hepatocytes. FASEB J. 15, 2286–2287 (2001).
Xue, L., Fletcher, G.C. & Tolkovsky, A.M. Mitochondria are selectively eliminated from eukaryotic cells after blockade of caspases during apoptosis. Curr. Biol. 11, 361–365 (2001).
Nylandsted, J. et al. Selective depletion of heat shock protein 70 (Hsp70) activates a tumor-specific death program that is independent of caspases and bypasses Bcl-2. Proc. Natl. Acad. Sci. USA 97, 7871–7876 (2000).
Sperandio, S., de Belle, I. & Bredesen, D.E. An alternative, nonapoptotic form of programmed cell death. Proc. Natl. Acad. Sci. USA 97, 14376–14381 (2000).
Mattson, M.P. Apoptosis in neurodegenerative disorders. Nat. Rev. Mol. Cell Biol. 1, 120–129 (2000).
Roberts, L.R. et al. Cathepsin B contributes to bile salt-induced apoptosis of rat hepatocytes. Gastroenterology 113, 1714–1726 (1997).
Roberg, K., Johansson, U. & Ollinger, K. Lysosomal release of cathepsin D precedes relocation of cytochrome c and loss of mitochondrial transmembrane potential during apoptosis induced by oxidative stress. Free Radic. Biol. Med. 27, 1228–1237 (1999).
Roberg, K., Kagedal, K. & Ollinger, K. Microinjection of cathepsin d induces caspase-dependent apoptosis in fibroblasts. Am. J. Pathol. 161, 89–96 (2002).
Guicciardi, M.E. et al. Cathepsin B contributes to TNF-α–mediated hepatocyte apoptosis by promoting mitochondrial release of cytochrome c. J. Clin. Invest 106, 1127–1137 (2000).
Foghsgaard, L. et al. Cathepsin B acts as a dominant execution protease in tumor cell apoptosis induced by tumor necrosis factor. J. Cell Biol. 153, 999–1009 (2001).
Roberts, L.R., Adjei, P.N. & Gores, G.J. Cathepsins as effector proteases in hepatocyte apoptosis. Cell Biochem. Biophys. 30, 71–88 (1999).
van Eijk, M. & de Groot, C. Germinal center B cell apoptosis requires both caspase and cathepsin activity. J. Immunol. 163, 2478–2482 (1999).
Yuan, X.M. et al. Lysosomal destabilization in p53-induced apoptosis. Proc. Natl. Acad. Sci. USA 99, 6286–6291 (2002).
Katz, E. et al. B cell receptor-stimulated mitochondrial phospholipase A2 activation and resultant disruption of mitochondrial membrane potential correlate with the induction of apoptosis in WEHI-231 B cells. J. Immunol. 166, 137–147 (2001).
Roberg, K. & Ollinger, K. Oxidative stress causes relocation of the lysosomal enzyme cathepsin D with ensuing apoptosis in neonatal rat cardiomyocytes. Am. J. Pathol. 152, 1151–1156 (1998).
Roberg, K. Relocalization of cathepsin D and cytochrome c early in apoptosis revealed by immunoelectron microscopy. Lab. Invest. 81, 149–158 (2001).
Kagedal, K., Zhao, M., Svensson, I. & Brunk, U.T. Sphingosine-induced apoptosis is dependent on lysosomal proteases. Biochem. J. 359, 335–343 (2001).
Foghsgaard, L., Lademann, U., Wissing, D., Poulsen, B. & Jaattela, M. Cathepsin B mediates tumor necrosis factor-induced arachidonic acid release in tumor cells. J. Biol. Chem. 277, 39499–39506 (2002).
Gobeil, S., Boucher, C.C., Nadeau, D. & Poirier, G.G. Characterization of the necrotic cleavage of poly(ADP-ribose) polymerase (PARP-1): implication of lysosomal proteases. Cell Death Differ. 8, 588–594 (2001).
Vancompernolle, K. et al. Atractyloside-induced release of cathepsin B, a protease with caspase-processing activity. FEBS Lett. 438, 150–158 (1998).
Stoka, V. et al. Lysosomal protease pathways to apoptosis. Cleavage of Bid, not pro-caspases, is the most likely route. J. Biol. Chem. 276, 3149–3157 (2001).
Schotte, P., Declercq, W., Van Huffel, S., Vandenabeele, P. & Beyaert, R. Non-specific effects of methyl ketone peptide inhibitors of caspases. FEBS Lett. 442, 117–121 (1999).
Reggiori, F. & Klionsky, D.J. Autophagy in the eukaryotic cell. Eukaryot. Cell 1, 11–21 (2002).
Cataldo, A.M., Hamilton, D.J. & Nixon, R.A. Lysosomal abnormalities in degenerating neurons link neuronal compromise to senile plaque development in Alzheimer disease. Brain Res. 640, 68–80 (1994).
Anglade, P. et al. Apoptosis and autophagy in nigral neurons of patients with Parkinson's disease. Histol. Histopathol. 12, 25–31 (1997).
Liang, X.H. et al. Induction of autophagy and inhibition of tumorigenesis by beclin 1. Nature 402, 672–676 (1999).
Xue, L., Fletcher, G.C. & Tolkovsky, A.M. Autophagy is activated by apoptotic signalling in sympathetic neurons: an alternative mechanism of death execution. Mol. Cell Neurosci 14, 180–198 (1999).
Marino, G. et al. Human autophagins, a family of cysteine proteinases potentially implicated in cell degradation by autophagy. J. Biol. Chem. 278, 3671–3678 (2002).
Deiss, L.P., Galinka, H., Berissi, H., Cohen, O. & Kimchi, A. Catepsin D protease mediates programmed cell death induced by interferon-γ, Fas/APO-1 and TNF-α. EMBO J. 15, 3861–3870 (1996).
Inbal, B., Bialik, S., Sabanay, I., Shani, G. & Kimchi, A. DAP kinase and DRP-1 mediate membrane blebbing and the formation of autophagic vesicles during programmed cell death. J. Cell Biol. 157, 455–468 (2002).
Caro, L.H., Plomp, P.J., Wolvetang, E.J., Kerkhof, C. & Meijer, A.J. 3-Methyladenine, an inhibitor of autophagy, has multiple effects on metabolism. Eur. J. Biochem 175, 325–329 (1988).
Wang, K.K. Calpain and caspase: can you tell the difference? Trends. Neurosci. 23, 20–26 (2000).
Mathiasen, I.S., Hansen, C.M., Foghsgaard, L. & Jäättelä, M. Sensitization to TNF-induced apoptosis by 1,25-dihydroxy vitamin D3 involves up-regulation of the TNF receptor 1 and cathepsin B. Int. J. Cancer 93, 224–231. (2001).
Mathiasen, I.S. et al. Calcium and calpain as key mediators of apoptosis-like death induced by vitamin D compounds in breast cancer cells. J. Biol. Chem. 277, 30738–30745 (2002).
Bao, J.J. et al. Reexpression of the tumor suppressor gene ARHI induces apoptosis in ovarian and breast cancer cells through a caspase-independent calpain-dependent pathway. Cancer Res. 62, 7264–7272 (2002).
Bodmer, J.L., Schneider, P. & Tschopp, J. The molecular architecture of the TNF superfamily. Trends Biochem. Sci. 27, 19–26 (2002).
Locksley, R.M., Killeen, N. & Lenardo, M.J. The TNF and TNF receptor superfamilies: integrating mammalian biology. Cell 104, 487–501 (2001).
Ashkenazi, A. & Dixit, V.M. Death receptors: signaling and modulation. Science 281, 1305–1308 (1998).
Siegel, R.M. et al. Fas preassociation required for apoptosis signaling and dominant inhibition by pathogenic mutations. Science 288, 2354–2357 (2000).
Kischkel, F.C. et al. Cytotoxicity-dependent Apo-1 (Fas/CD95)-associated proteins form a death-inducing signaling complex (DISC) with the receptor. EMBO J. 14, 5579–5588 (1995).
Kischkel, F.C. et al. Death receptor recruitment of endogenous caspase-10 and apoptosis initiation in the absence of caspase-8. J. Biol. Chem. 276, 46639–46646 (2001).
Salvesen, G.S. & Dixit, V.M. Caspase activation: the induced-proximity model. Proc. Natl. Acad. Sci. USA 96, 10964–10967 (1999).
Thome, M. & Tschopp, J. Regulation of lymphocyte proliferation and death by FLIP. Nat. Rev. Immunol. 1, 50–58 (2001).
Kataoka, T. et al. The caspase-8 inhibitor FLIP promotes activation of NF-κB and ERK signaling pathways. Curr. Biol. 10, 640–648 (2000).
Chen, G. & Goeddel, D.V. TNF-R1 signaling: a beautiful pathway. Science 296, 1634–1635 (2002).
Sun, X., Yin, J., Starovasnik, M.A., Fairbrother, W.J. & Dixit, V.M. Identification of a novel homotypic interaction motif required for the phosphorylation of receptor-interacting protein (RIP) by RIP3. J. Biol. Chem. 277, 9505–9511 (2002).
Yeh, W.C. et al. FADD: essential for embryo development and signaling from some, but not all, inducers of apoptosis. Science 279, 1954–1958 (1998).
Zhang, J., Cado, D., Chen, A., Kabra, N.H. & Winoto, A. Fas-mediated apoptosis and activation-induced T-cell proliferation are defective in mice lacking FADD/Mort1. Nature 392, 296–300 (1998).
Varfolomeev, E.E., Boldin, M.P., Goncharov, T.M. & Wallach, D. A potential mechanism of 'cross-talk' between the p55 tumor necrosis factor receptor and Fas/APO1: proteins binding to the death domains of the two receptors also bind to each other. J. Exp. Med. 183, 1271–1275 (1996).
Vercammen, D., Vandenabeele, P., Beyaert, R., Declercq, W. & Fiers, W. Tumour necrosis factor-induced necrosis versus anti-Fas–induced apoptosis in L929 cells. Cytokine 9, 801–808 (1997).
Luschen, S., Ussat, S., Scherer, G., Kabelitz, D. & Adam-Klages, S. Sensitization to death receptor cytotoxicity by inhibition of Fas-associated death domain protein (FADD)/caspase signaling. Requirement of cell cycle progression. J. Biol. Chem. 275, 24670–24678 (2000).
Khwaja, A. & Tatton, L. Resistance to the cytotoxic effects of tumor necrosis factor alpha can be overcome by inhibition of a FADD/caspase-dependent signaling pathway. J. Biol. Chem. 274, 36817–36823 (1999).
Vonarbourg, C. et al. Differential sensitivity of Jurkat and primary T cells to caspase-independent cell death triggered upon Fas stimulation. Eur J. Immunol. 32, 2376–2384 (2002).
Kawahara, A., Ohsawa, Y., Matsumura, H., Uchiyama, Y. & Nagata, S. Caspase-independent cell killing by Fas-associated protein with death domain. J. Cell Biol. 143, 1353–1360 (1998).
Kalai, M., Van Loo, G., Declercq, W. & Vandenabeele, P. Disruption of HSP90 function reverts TNF-induced necrosis to apoptosis. J. Biol. Chem. 278, 5622–5629 (2003).
Kelliher, M.A. et al. The death domain kinase RIP mediates the TNF-induced NF-κB signal. Immunity 8, 297–303 (1998).
Holler, N. et al. Development of improved soluble inhibitors of FasL and CD40L based on oligomerized receptors. J. Immunol. Methods 237, 159–173 (2000).
Goossens, V. et al. Redox regulation of TNF signaling. Biofactors 10, 145–156 (1999).
Kudo, I. & Murakami, M. Phospholipase A2 enzymes. Prostaglandins Other Lipid Mediat. 68–69, 3–58 (2002).
Lin, L.L. et al. cPLA2 is phosphorylated and activated by MAP kinase. Cell 72, 269–278 (1993).
Waterman, W.H., Molski, T.F., Huang, C.K., Adams, J.L. & Sha'afi, R.I. Tumour necrosis factor-α-induced phosphorylation and activation of cytosolic phospholipase A2 are abrogated by an inhibitor of the p38 mitogen-activated protein kinase cascade in human neutrophils. Biochem. J. 319, 17–20 (1996).
Kolesnick, R.N. & Kronke, M. Regulation of ceramide production and apoptosis. Annu. Rev. Physiol. 60, 643–665 (1998).
Kronke, M. & Adam-Klages, S. Role of caspases in TNF-mediated regulation of cPLA2 . FEBS Lett. 531, 18–22 (2002).
Uzzo, R.G. et al. Inhibition of NFκB induces caspase-independent cell death in human T lymphocytes. Biochem. Biophys. Res. Commun. 287, 895–899 (2001).
Hildeman, D.A. et al. Reactive oxygen species regulate activation-induced T cell apoptosis. Immunity 10, 735–744 (1999).
Bidere, N. & Senik, A. Caspase-independent apoptotic pathways in T lymphocytes: a minireview. Apoptosis 6, 371–375 (2001).
Pettersen, R.D., Bernard, G., Olafsen, M.K., Pourtein, M. & Lie, S.O. CD99 signals caspase-independent T cell death. J. Immunol. 166, 4931–4942 (2001).
Doerfler, P., Forbush, K.A. & Perlmutter, R.M. Caspase enzyme activity is not essential for apoptosis during thymocyte development. J. Immunol. 164, 4071–4079 (2000).
Sayers, T.J. et al. T cell lysis of murine renal cancer: multiple signaling pathways for cell death via Fas. J. Leukoc Biol. 68, 81–86 (2000).
O'Connor, L. et al. Bim: a novel member of the Bcl-2 family that promotes apoptosis. EMBO J. 17, 384–395 (1998).
Brunner, T. et al. Cell-autonomous Fas (CD95)/Fas-ligand interaction mediates activation-induced apoptosis in T-cell hybridomas. Nature 373, 441–444 (1995).
Ju, S.T. et al. Fas(CD95)/FasL interactions required for programmed cell death after T-cell activation. Nature 373, 444–448 (1995).
Dhein, J., Walczak, H., Bäumler, C., Debatin, K.-M. & Krammer, P.H. Autocrine T-cell suicide mediated by APO-1/(Fas/CD95). Nature 373, 438–441 (1995).
Zheng, L.X. et al. Induction of apoptosis in mature T cells by tumour necrosis factor. Nature 377, 348–351 (1995).
Scaffidi, C., Kirchhoff, S., Krammer, P.H. & Peter, M.E. Apoptosis signaling in lymphocytes. Curr. Opin. Immunol. 11, 277–285 (1999).
Siegel, R.M., Chan, F.K., Chun, H.J. & Lenardo, M.J. The multifaceted role of Fas signaling in immune cell homeostasis and autoimmunity. Nat. Immunol. 1, 469–474 (2000).
Weih, F., Ryseck, R.P., Chen, L. & Bravo, R. Apoptosis of nur77/N10-transgenic thymocytes involves the Fas/Fas ligand pathway. Proc. Natl. Acad. Sci. USA 93, 5533–5538 (1996).
Mittelstadt, P.R. & Ashwell, J.D. Cyclosporin A-sensitive transcription factor Egr-3 regulates Fas ligand expression. Mol. Cell. Biol. 18, 3744–3751 (1998).
Nagata, S. & Suda, T. Fas and Fas ligand: lpr and gld mutations. Immunol. Today 16, 39–43 (1995).
Refaeli, Y., Van Parijs, L., London, C.A., Tschopp, J. & Abbas, A.K. Biochemical mechanisms of IL-2-regulated Fas-mediated T cell apoptosis. Immunity 8, 615–623 (1998).
Krueger, A., Baumann, S., Krammer, P.H. & Kirchhoff, S. FLICE-inhibitory proteins: regulators of death receptor-mediated apoptosis. Mol. Cell. Biol. 21, 8247–8254 (2001).
Berndt, C., Mopps, B., Angermuller, S., Gierschik, P. & Krammer, P.H. CXCR4 and CD4 mediate a rapid CD95-independent cell death in CD4+ T cells. Proc. Natl. Acad. Sci. USA 95, 12556–12561 (1998).
Davidson, W.F., Haudenschild, C., Kwon, J. & Williams, M.S. T cell receptor ligation triggers novel nonapoptotic cell death pathways that are Fas-independent or Fas-dependent. J. Immunol. 169, 6218–6230 (2002).
Smith, K.G., Strasser, A. & Vaux, D.L. CrmA expression in T lymphocytes of transgenic mice inhibits CD95 (Fas/APO-1)-transduced apoptosis, but does not cause lymphadenopathy or autoimmune disease. EMBO J. 15, 5167–5176 (1996).
Van Parijs, L., Peterson, D.A. & Abbas, A.K. The Fas/Fas ligand pathway and Bcl-2 regulate T cell responses to model self and foreign antigens. Immunity 8, 265–274 (1998).
Strasser, A., Harris, A.W., Huang, D.C.S., Krammer, P.H. & Cory, S. Bcl-2 and Fas/Apo-1 regulate distinct pathways to lymphocyte apoptosis. EMBO J. 14, 6136–6147 (1995).
Scott, D.E., Kisch, W.J. & Steinberg, A.D. Studies of T cell deletion and T cell anergy following in vivo administration of SEB to normal and lupus-prone mice. J. Immunol. 150, 664–672 (1993).
Hildeman, D.A. et al. Activated T cell death in vivo mediated by proapoptotic bcl-2 family member bim. Immunity 16, 759–767 (2002).
Ferraro-Peyret, C., Quemeneur, L., Flacher, M., Revillard, J.P. & Genestier, L. Caspase-independent phosphatidylserine exposure during apoptosis of primary T lymphocytes. J. Immunol. 169, 4805–4810 (2002).
Kennedy, N.J., Kataoka, T., Tschopp, J. & Budd, R.C. Caspase activation is required for T cell proliferation. J. Exp. Med. 190, 1891–1896 (1999).
Alam, A., Cohen, L.Y., Aouad, S. & Sekaly, R.P. Early activation of caspases during T lymphocyte stimulation results in selective substrate cleavage in nonapoptotic cells. J. Exp. Med. 190, 1879–1890 (1999).
Chun, H.J. et al. Pleiotropic defects in lymphocyte activation caused by caspase-8 mutations lead to human immunodeficiency. Nature 419, 395–399 (2002).
Acknowledgements
We thank our colleagues for discussions, M. Leist and H. Everett for help with the text and figures, and the Danish Cancer Society and the Swiss National Science Foundation for financial support.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jäättelä, M., Tschopp, J. Caspase-independent cell death in T lymphocytes. Nat Immunol 4, 416–423 (2003). https://doi.org/10.1038/ni0503-416
Issue Date:
DOI: https://doi.org/10.1038/ni0503-416
This article is cited by
-
Aloe-emodin inhibits African swine fever virus replication by promoting apoptosis via regulating NF-κB signaling pathway
Virology Journal (2023)
-
Clostridioides difficile toxin B alone and with pro-inflammatory cytokines induces apoptosis in enteric glial cells by activating three different signalling pathways mediated by caspases, calpains and cathepsin B
Cellular and Molecular Life Sciences (2022)
-
JNK1 and ERK1/2 modulate lymphocyte homeostasis via BIM and DRP1 upon AICD induction
Cell Death & Differentiation (2020)
-
BIM upregulation and ROS-dependent necroptosis mediate the antitumor effects of the HDACi Givinostat and Sorafenib in Hodgkin lymphoma cell line xenografts
Leukemia (2014)
-
Azacytidine induces necrosis of multiple myeloma cells through oxidative stress
Proteome Science (2013)