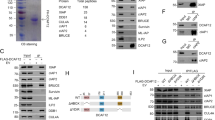
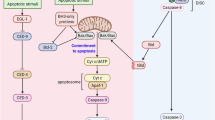
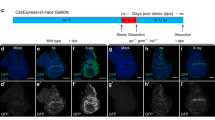
Abstract
Apoptosis is induced by caspases, which are members of the cysteine protease family1. Caspases are synthesized as inactive zymogens and initiator caspases first gain activity by associating with an oligomeric complex of their adaptor proteins, such as the apoptosome2,3. Activated initiator caspases subsequently cleave and activate effector caspases. Although such a proteolytic cascade would predict that a small number of active caspases could irreversibly amplify caspase activity and trigger apoptosis, many cells can maintain moderate levels of caspase activity to perform non-apoptotic roles in cellular differentiation, shape change and migration4. Here we show that the Drosophila melanogaster apoptosome engages in a feedback inhibitory loop, which moderates its activation level in vivo. Specifically, the adaptor protein Apaf-1 lowers the level of its associated initiator caspase Dronc, without triggering apoptosis. Conversely, Dronc lowers Apaf-1 protein levels. This mutual suppression depends on the catalytic site of Dronc and a caspase cleavage site within Apaf-1. Moreover, the Drosophila inhibitor of apoptosis protein 1 (Diap1) is required for this process. We speculate that this feedback inhibition allows cells to regulate the degree of caspase activation for apoptotic and non-apoptotic purposes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Thornberry, N. A. & Lazebnik, Y. Caspases: enemies within. Science 281, 1312–1316 (1998).
Zou, H., Li, Y., Liu, X. & Wang, X. An APAF-1 cytochrome c multimeric complex is a functional apoptosome that activates procaspase-9. J. Biol. Chem. 274, 11549–11556 (1999).
Rodriguez, J. & Lazebnik, Y. Caspase-9 and Apaf-1 form an active holoenzyme. Genes Dev. 13, 3179–3184 (1999).
Kuranaga, E. & Miura, M. Nonapoptotic functions of caspases: caspases as regulatory molecules for immunity and cell-fate determination. Trends Cell Biol. 17, 135–144.
Yu, X., Wang, L., Acehan, D., Wang, X. & Akey, C. W. Three-dimensional structure of a double apoptosome formed by the Drosophila Apaf-1 related killer. J. Mol. Biol. 355, 577–589 (2006).
Rodriguez, A. et al. Dark is a Drosophila homologue of Apaf-1/CED-4 and functions in an evolutionarily conserved death pathway. Nature Cell Biol. 1, 272–279 (1999).
Zhou, L., Song, Z., Tittel, J. & Steller, H. HAC-1, a Drosophila homolog of APAF-1 and CED-4 functions in developmental and radiation-induced apoptosis. Mol. Cell 4, 745–755 (1999).
Kanuka, H. et al. Control of the cell death pathway by Dapaf-1, a Drosophila Apaf-1/CED-4-related caspase activator. Mol. Cell 4, 757–769 (1999).
Srivastava, M. et al. ARK, the Apaf-1 related killer in Drosophila, requires diverse domains for its apoptotic activity. Cell Death Differ. 14, 92–102 (2006).
Chew, S. K. et al. The apical caspase dronc governs programmed and unprogrammed cell death in Drosophila. Dev. Cell 7, 897–907 (2004).
Daish, T. J., Mills, K. & Kumar, S. Drosophila caspase DRONC is required for specific developmental cell death pathways and stress-induced apoptosis. Dev. Cell 7, 909–915 (2004).
Xu, D., Li, Y., Arcaro, M., Lackey, M. & Bergmann, A. The CARD-carrying caspase Dronc is essential for most, but not all, developmental cell death in Drosophila. Development 132, 2125–2134 (2005).
Yan, N. et al. Structure and activation mechanism of the Drosophila initiator caspase Dronc. J. Biol. Chem. 281, 8667–8674 (2006).
Akdemir, F. et al. Autophagy occurs upstream or parallel to the apoptosome during histolytic cell death. Development 113, 1457–1465 (2006).
Clem, R. J., Fechheimer, M. & Miller, K. Prevention of apoptosis by a baculovirus gene during infection of insect cells. Science 254, 1388–1390 (1991).
Meier, P., Silke, J., Leevers, S. J. & Evan, G. I. The Drosophila caspase DRONC is regulated by DIAP1. EMBO J. 19, 598–611 (2000).
Groth, A. C., Fish, M., Nusse, R. & Calos, M. P. Construction of transgenic Drosophila by using the site-specific integrase from phage phiC31. Genetics 166, 1775–1782 (2004).
Bischof, J., Maeda, R. K., Hediger, M., Karch, F. & Basler, K. An optimized transgenesis system for Drosophila using germ-line-specific phiC31 integrases. Proc. Natl Acad. Sci. USA 104, 3312–3317 (2007).
White, K. et al. Genetic control of programmed cell death in Drosophila. Science 264, 677–683 (1994).
Grether, M. E., Abrams, J. M., Agapite, J., White, K. & Steller, H. The head involution defective gene of Drosophila melanogaster functions in programmed cell death. Genes Dev. 9, 1694–1708 (1995).
Chen, P., Nordstrom, W., Gish, B. & Abrams, J. M. grim, a novel cell death gene in Drosophila. Genes Dev. 10, 1773–1782 (1996).
Steller, H. Regulation of apoptosis in Drosophila. Cell Death Differ. 15, 1132–1138 (2008).
Wang, S. L., Hawkins, C. J., Yoo, S. J., Muller, H. A. & Hay, B. A. The Drosophila caspase inhibitor DIAP1 is essential for cell survival and is negatively regulated by HID. Cell 98, 453–63 (1999).
Goyal, L., McCall, K., Agapite, J., Hartwieg, E. & Steller, H. Induction of apoptosis by Drosophila reaper, hid and grim through inhibition of IAP function. EMBO. J. 19, 589–597 (2000).
Lisi, S., Mazzon, I. & White, K. Diverse domains of THREAD/DIAP1 are required to inhibit apoptosis induced by REAPER and HID in Drosophila. Genetics 154, 669–678 (2000).
Wilson, R. et al. The DIAP1 RING finger mediates ubiquitination of Dronc and is indispensable for regulating apoptosis. Nature Cell Biol. 4, 445–450 (2002).
Muro, I., Hay, B. A. & Clem, R. J. The Drosophila DIAP1 protein is required to prevent accumulation of a continuously generated, processed form of the apical caspase DRONC. J. Biol. Chem. 277, 49644–49650 (2002).
Herman-Bachinsky, Y., Ryoo, H. D., Ciechanover, A. & Gonen, H. Regulation of the Drosophila ubiquitin ligase DIAP1 is mediated via several distinct ubiquitin system pathways. Cell Death Differ. 14, 861–871 (2007).
Kornbluth, S. & White, K. Apoptosis in Drosophila: neither fish nor fowl (nor man nor worm). J. Cell Sci. 118, 1779–1787 (2005).
Brand, A. H. & Perrimon, N. Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118, 401–415 (1993).
Golic, K. G. & Lindquist, S. The FLP recombinase of yeast catalyzes site-specific recombination in the Drosophila genome. Cell 59, 499–509 (1989).
Struhl, G. & Basler, K. Organizing activity of wingless protein in Drosophila. Cell 72, 527–540 (1993).
Ryoo, H. D., Gorenc, T. & Steller, H. Apoptotic cells can induce compensatory cell proliferation through the JNK and the Wingless signaling pathways. Dev. Cell 7, 491–501 (2004).
Ryoo, H. D., Domingos, P. M., Kang, M. J. & Steller, H. Unfolded protein response in a Drosophila model for retinal degeneration. EMBO J. 26, 242–252 (2007).
Ryoo, H. D., Bergmann, A., Gonen, H., Ciechanover, A. & Steller, H. Regulation of Drosophila IAP1 degradation and apoptosis by reaper and ubcD1. Nature Cell. Biol. 4, 432–438 (2002).
Acknowledgements
We thank John Abrams, Andreas Bergmann, George Jackson, Pascal Meier for fly stocks; David Sabatini for making available the scanning electron microscope; Milton Adesnik, Ramanuj Dasgupta, Zehra Ordulu, David Sabatini, Greg Suh and Jessica Treisman for advice and comments on the manuscript. P.J.S. is supported by an NIH predoctoral training grant 5T32GM007239-32. H.D.R. is a Special Fellow of the Leukemia-Lymphoma Society, a Basil O'Conner Scholar of the March of Dimes Foundation and an Ellison Medical Foundation New Scholar. This work was supported by a grant to H.D.R. from the National Institutes of Health (1RO1GM079425)
Author information
Authors and Affiliations
Contributions
H.D.R. conceived the project; P.J.S., H.J. and H.D.R. designed the experiments and analysed the data; all authors performed experiments; E.S.R. contributed to the scanning electron microscopy experiments; H.D.R. wrote the paper and all authors read and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 723 kb)
Rights and permissions
About this article
Cite this article
Shapiro, P., Hsu, H., Jung, H. et al. Regulation of the Drosophila apoptosome through feedback inhibition. Nat Cell Biol 10, 1440–1446 (2008). https://doi.org/10.1038/ncb1803
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1803
This article is cited by
-
Wg/Wnt1 and Erasp link ER stress to proapoptotic signaling in an autosomal dominant retinitis pigmentosa model
Experimental & Molecular Medicine (2023)
-
An inhibitor of apoptosis protein (EsIAP1) from Chinese mitten crab Eriocheir sinensis regulates apoptosis through inhibiting the activity of EsCaspase-3/7-1
Scientific Reports (2019)
-
Killers creating new life: caspases drive apoptosis-induced proliferation in tissue repair and disease
Cell Death & Differentiation (2017)
-
More alive than dead: non-apoptotic roles for caspases in neuronal development, plasticity and disease
Cell Death & Differentiation (2017)
-
Drosophila caspase activity is required independently of apoptosis to produce active TNF/Eiger during nociceptive sensitization
Cell Death & Disease (2017)