Abstract
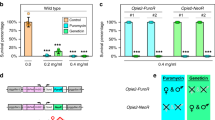
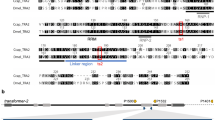
The Sterile Insect Technique is a species-specific and environmentally friendly method of pest control involving mass release of sterilized insects that reduce the wild population through infertile matings1,2,3,4,5. Insects carrying a female-specific autocidal genetic system offer an attractive alternative to conventional sterilization methods6,7 while also eliminating females from the release population7,8,9,10. We exploited sex-specific alternative splicing in insects to engineer female-specific autocidal genetic systems in the Mediterranean fruit fly, Ceratitis capitata. These rely on the insertion of cassette exons from the C. capitata transformer gene into a heterologous tetracycline-repressible transactivator such that the transactivator transcript is disrupted in male splice variants but not in the female-specific one. As the key components of these systems function across a broad phylogenetic range, this strategy addresses the paucity of sex-specific expression systems (e.g., early-acting, female-specific promoters) in insects other than Drosophila melanogaster. The approach may have wide applicability for regulating gene expression in other organisms, particularly for combinatorial control with appropriate promoters.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Keng-Hong, T. (ed.) Area-Wide Control of Fruit Flies and Other Insect Pests (Penerbit Universiti Sains Malaysia, Penang, 2000).
Knipling, E. Possibilities of insect control or eradication through use of sexually sterile males. J. Econ. Entomol. 48, 459–462 (1955).
Koyama, J., Kakinohana, H. & Miyatake, T. Eradication of the melon fly Bactrocera cucurbitae in Japan: importance of behaviour, ecology, genetics and evolution. Annu. Rev. Entomol. 49, 331–349 (2004).
Lindquist, D.A., Abusowa, M. & Hall, M.J. The New World screwworm fly in Libya: a review of its introduction and eradication. Med. Vet. Entomol. 6, 2–8 (1992).
Dyck, V., Hendrichs, J. & Robinson, A. (eds.). Sterile Insect Technique: Principles and Practice in Area-Wide Integrated Pest Management (Springer, Dordrecht, 2005).
Alphey, L. Re-engineering the Sterile Insect Technique. Insect Biochem. Mol. Biol. 32, 1243–1247 (2002).
Thomas, D.D., Donnelly, C.A., Wood, R.J. & Alphey, L.S. Insect population control using a dominant, repressible, lethal genetic system. Science 287, 2474–2476 (2000).
Heinrich, J.C. & Scott, M. A repressible female-specific lethal genetic system for making transgenic insect strains suitable for a sterile-release program. Proc. Natl. Acad. Sci. USA 97, 8229–8232 (2000).
Marec, F. et al. Development of genetic sexing strains in Lepidoptera: from traditional to transgenic approaches. J. Econ. Entomol. 98, 248–259 (2005).
Rendón, P., McInnis, D., Lance, D. & Stewart, J. Medfly (Diptera:Tephritidae) genetic sexing: large-scale field comparison of males-only and bisexual sterile fly releases in Guatemala. J. Econ. Entomol. 97, 1547–1553 (2004).
Franz, G. Genetic sexing strains in mediterranean fruit fly, an example for other species amenable to large-scale rearing for the sterile insect technique. in Sterile Insect Technique. Principles and Practice in Area-Wide Integrated Pest Management (eds. Dyck, V.A., Hendrichs, J. & Robinson, A.S.) 427–451, (Springer, The Netherlands; 2005).
Wimmer, E.A. Eco-friendly insect management. Nat. Biotechnol. 23, 432–433 (2005).
Gould, F. & Schliekelman, P. Population genetics of autocidal control and strain replacement. Annu. Rev. Entomol. 49, 193–217 (2004).
Schliekelman, P. & Gould, F. Pest control by the release of insects carrying a female-killing allele on multiple loci. J. Econ. Entomol. 93, 1566–1579 (2000).
Gossen, M. & Bujard, H. Tight control of gene expression in mammalian cells by tetracycline-responsive promoters. Proc. Natl. Acad. Sci. USA 89, 5547–5551 (1992).
Tortiglione, C. & Bownes, M. Conservation and divergence in the control of yolk protein genes in dipteran insects. Dev. Genes Evol. 207, 264–281 (1997).
Black, D.L. Mechanisms of alternative pre-messenger RNA splicing. Annu. Rev. Biochem. 72, 291–336 (2003).
Park, J.W., Parisky, K., Celotto, A., Reenan, R. & Graveley, B. Identification of alternative splicing regulators by RNA interference in Drosophila. Proc. Natl. Acad. Sci. USA 101, 15974–15979 (2004).
Pane, A., Salvemini, M., Delli Bovi, P., Polito, C. & Saccone, G. The transformer gene in Ceratitis capitata provides a genetic basis for selecting and remembering the sexual fate. Development 129, 3715–3725 (2002).
Saccone, G., Pane, A. & Polito, C. Sex determination in flies, fruitflies and butterflies. Genetica 116, 15–23 (2002).
Gong, P. et al. A dominant lethal genetic system for autocidal control of the Mediterranean fruitfly. Nat. Biotechnol. 23, 453–456 (2005).
Horn, C. & Wimmer, E. A transgene-based, embryo-specific lethality system for insect pest management. Nat. Biotechnol. 21, 64–70 (2003).
Cartegni, L., Chew, S. & Krainer, A. Listening to silence and understanding nonsense: exonic mutations that affect splicing. Nat. Rev. Genet. 3, 285–298 (2002).
Saccone, G. et al. in Area Wide Control of Fruit Flies and Other Insect Species. (ed. Tan, K.) 491–496, (Penerbit Universiti Sains Malaysia, Penang, Malaysia; 2000).
Stoss, O., Stoilov, P., Hartmann, A.M., Nayler, O. & Stamm, S. The in vivo minigene approach to analyze tissue-specific splicing. Brain Res. Brain Res. Protoc. 4, 383–394 (1999).
Stoss, O. et al. The STAR/GSG family protein rSLM-2 regulates the selection of alternative splice sites. J. Biol. Chem. 276, 8665–8673 (2001).
Chiu, Y. & Pin Ouyang, A. Loss of Pnn expression attenuates expression levels of SR family splicing factors and modulates alternative pre-mRNA splicing in vivo. Biochem. Biophys. Res. Commun. 341, 663–671 (2006).
Gaunt, M.W. & Miles, M. An insect molecular clock dates the origin of the insects and accords with palaeontological and biogeographic landmarks. Mol. Biol. Evol. 19, 748–761 (2002).
Pane, A., De Simone, A., Saccone, G. & Polito, C. Evolutionary conservation of Ceratitis capitata transformer gene function. Genetics 171, 615–624 (2005).
Lyman, R.F., Lawrence, F., Nuzhdin, S.V. & Mackay, T.F. Effects of single P-element insertions on bristle number and viability in Drosophila melanogaster. Genetics 143, 277–292 (1996).
Acknowledgements
We thank Pedro Rendón (USDA-APHIS-PPQ-CPHST) for the Toliman wild-type strain, and Trudi Mackay for the SamI236 strains; Sinead O'Connell and Veronica Rodrigues for help with D. melanogaster; Karen Clifton and Lynn Richardson for technical assistance and Sinead O'Connell and Derric Nimmo for critical review of the manuscript. This work was funded in part by the UK Biotechnology and Biological Sciences Research Council.
Author information
Authors and Affiliations
Contributions
G.F. and K.C.C. contributed equally to this work. G.F. and L.J. made the DNA constructs and assisted with the molecular analysis of transgenics. K.C.C. and G.C.C. created the transgenic medfly lines described and conducted most of the phenotypic and molecular analysis. M.J.E. and P.G. performed preliminary experiments in medflies. N.I.M. performed the D. melanogaster experiments. T.H.D. provided advice. L.A. conceived and supervised the project and wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
Those authors affiliated with Oxitec Ltd. are or were employees or collaborative students of this company, which therefore provided salary and other support for the research program. Also, all these employees have or have had shares or share options in Oxitec Ltd. Both Oxitec Ltd. and Oxford University have one or more patents or patent applications related to the subject of this paper.
Supplementary information
Supplementary Fig. 1
Splicing of Cetra intron in Drosophila melanogaster and Ceratitis capitata. (PDF 203 kb)
Supplementary Fig. 2
Sequence of plasmid pLA3077. (PDF 130 kb)
Supplementary Fig. 3
Sequence of plasmid pLA3097. (PDF 132 kb)
Supplementary Table 1
Repressible female-specific lethality in heterozygous transgenic Medfly. (PDF 127 kb)
Supplementary Table 2
Repressible female-specific lethality in homozygous transgenic Medfly. (PDF 106 kb)
Supplementary Table 3
Repressible female-specific lethality in heterozygous transgenic Drosophila. (PDF 120 kb)
Rights and permissions
About this article
Cite this article
Fu, G., Condon, K., Epton, M. et al. Female-specific insect lethality engineered using alternative splicing. Nat Biotechnol 25, 353–357 (2007). https://doi.org/10.1038/nbt1283
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1283
This article is cited by
-
Next-generation genetic sexing strain establishment in the agricultural pest Ceratitis capitata
Scientific Reports (2023)
-
Self-limiting fall armyworm: a new approach in development for sustainable crop protection and resistance management
BMC Biotechnology (2022)
-
A transgenic female killing system for the genetic control of Drosophila suzukii
Scientific Reports (2021)
-
Maintenance management and eradication of established aquatic invaders
Hydrobiologia (2021)
-
Identification and characterization of four Drosophila suzukii cellularization genes and their promoters
BMC Genetics (2020)