Abstract
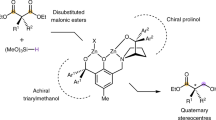
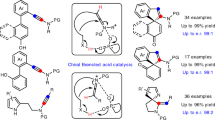
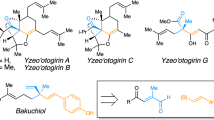
Chiral Brønsted acids (proton donors) have been shown to facilitate a broad range of asymmetric chemical transformations under catalytic conditions without requiring additional toxic or expensive metals1,2,3,4,5,6,7,8. Although the catalysts developed thus far are remarkably effective at activating polarized functional groups, it is not clear whether organic Brønsted acids can be used to catalyse highly enantioselective transformations of unactivated carbon–carbon multiple bonds. This deficiency persists despite the fact that racemic acid-catalysed ‘Markovnikov’ additions to alkenes are well known chemical transformations. Here we show that chiral dithiophosphoric acids can catalyse the intramolecular hydroamination and hydroarylation of dienes and allenes to generate heterocyclic products in exceptional yield and enantiomeric excess. We present a mechanistic hypothesis that involves the addition of the acid catalyst to the diene, followed by nucleophilic displacement of the resulting dithiophosphate intermediate; we also report mass spectroscopic and deuterium labelling studies in support of the proposed mechanism. The catalysts and concepts revealed in this study should prove applicable to other asymmetric functionalizations of unsaturated systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Akiyama, T., Itoh, J., Yokota, K. & Fuchibe, K. Enantioselective Mannich-type reaction catalyzed by a chiral Brønsted acid. Angew. Chem. Int. Edn 43, 1566–1568 (2004)
Uraguchi, D. & Terada, M. Chiral Brønsted acid-catalyzed direct Mannich reactions via electrophilic activation. J. Am. Chem. Soc. 126, 5356–5357 (2004)
Terada, M. Binaphthol-derived phosphoric acid as a versatile catalyst for enantioselective carbon–carbon bond forming reactions. Chem. Commun. 4097–4112 (2008)
Akiyama, T. Stronger Brønsted acids. Chem. Rev. 107, 5744–5758 (2007)
Nakashima, D. & Yamamoto, H. Design of chiral N-triflyl phosphoramide as a strong chiral Brønsted acid and its application to asymmetric Diels−Alder reaction. J. Am. Chem. Soc. 128, 9626–9627 (2006)
Cheon, C. H. & Yamamoto, H. A Brønsted acid catalyst for the enantioselective protonation reaction. J. Am. Chem. Soc. 130, 9246–9247 (2008)
Hatano, M., Maki, T., Moriyama, K., Arinobe, M. & Ishihara, K. Pyridinium 1,1′-binaphthyl-2,2′-disulfonates as highly effective chiral Brønsted acid−base combined salt catalysts for enantioselective Mannich-type reaction. J. Am. Chem. Soc. 130, 16858–16860 (2008)
García-García, P., Lay, F., García-García, P., Rabalakos, C. & List, B. A powerful chiral counteranion motif for asymmetric catalysis. Angew. Chem. Int. Edn 48, 4363–4366 (2009)
Aillaud, I., Collin, J., Hannedouche, J. & Schulz, E. Asymmetric hydroamination of non-activated carbon–carbon multiple bonds. Dalton Trans. 5105–5118 (2007)
Hultzsch, K. C. Transition metal-catalyzed asymmetric hydroamination of alkenes (AHA). Adv. Synth. Catal. 347, 367–391 (2005)
Hultzsch, K. C. Catalytic asymmetric hydroamination of non-activated olefins. Org. Biomol. Chem. 3, 1819–1824 (2005)
Roesky, P. W. & Müller, T. E. Enantioselective catalytic hydroamination of alkenes. Angew. Chem. Int. Edn 42, 2708–2710 (2003)
Schlummer, B. & Hartwig, J. F. Brønsted acid-catalyzed intramolecular hydroamination of protected alkenylamines. Synthesis of pyrrolidines and piperidines. Org. Lett. 4, 1471–1474 (2002)
Rosenfeld, D. C., Shekhar, S., Takemiya, A., Utsunomiya, M. & Hartwig, J. F. Hydroamination and hydroalkoxylation catalyzed by triflic acid. Parallels to reactions initiated with metal triflates. Org. Lett. 8, 4179–4182 (2006)
Li, Z. et al. Brønsted acid catalyzed addition of phenols, carboxylic acids, and tosylamides to simple olefins. Org. Lett. 8, 4175–4178 (2006)
Ackermann, L. & Althammer, A. Phosphoric acid diesters as efficient catalysts for hydroaminations of nonactivated alkenes and an application to asymmetric hydroamination. Synlett 995–998 (2008)
MacMillan, D. W. C. The advent and development of organocatalysis. Nature 455, 304–308 (2008)
Norman, G. R., LeSuer, W. M. & Mastin, T. W. Chemistry of the aliphatic esters of thiophosphoric acids. II. O,O,S-trialkyl thionophosphates by the addition of O,O-dialkyl thiolthionophosphoric acids to olefins. J. Am. Chem. Soc. 74, 161–163 (1952)
Kabachnik, M. I., Mastrukova, T. A., Shipov, A. E. & Melentyeva, T. A. The application of the Hammett equation to the theory of tautomeric equilibrium: thione-thiol equilibrium, acidity, and structure of phosphorous thioacids. Tetrahedron 9, 10–28 (1960)
Mastryukova, T. A., Spivak, L. L., Grigoreva, A. A., Urzhuntseva, E. K. & Kabachnik, M. I. Ionization constants of phosphorus dithio acids in absolute ethanol. J. Gen. Chem. USSR (Engl. Transl.) 41, 1938–1941 (1971)
Leavitt, C. M. et al. Investigations of acidity and nucleophilicity of diphenyldithiophosphinate ligands using theory and gas-phase dissociation reactions. Inorg. Chem. 47, 3056–3064 (2008)
Mueller, W. H. & Oswald, A. A. Organic sulfur compounds. XV. Cationic addition of O,O′-diethylthiophosphoric acid to olefins. J. Org. Chem. 31, 1894–1898 (1966)
Pousse, G. et al. Synthesis of BINOL derived phosphorodithioic acids as new chiral Brønsted acids and an improved synthesis of 3,3′-disubstituted H8-BINOL derivatives. Tetrahedron 65, 10617–10622 (2009)
Cheng, X. C., Wang, Q., Fang, H., Tang, W. & Xu, W. F. Synthesis of new sulfonyl pyrrolidine derivatives as matrix metalloproteinase inhibitors. Bioorg. Med. Chem. 16, 7932–7938 (2008)
Mutel, V. & Wichmann, J. Sulfonyl-pyrrolidine derivatives useful for the treatment of neurological disorders. European patent 1,299. 383 (2005)
Obushak, N. D., Vovk, M. V., Vengrzhanovskii, V. A., Mel'nik, Y. I. & Ganushchak, N. I. Regioselective addition of O,O-diethyl dithiophosphate to 1-aryl-1,3-butadienes. J. Gen. Chem. USSR (Engl. Transl.) 57, 1078–1080 (1987)
Mebah, J. M. N., Mieloszynski, J. L. & Paquer, D. Reactivity of O,O-diethyldithiophosphoric acid toward different ethylenic systems. Phosphorus Sulfur Silicon Relat. Elem. 73, 49–56 (1992)
Houk, K. N., Paddon-Row, M. N. & Rondan, N. G. The origin of syn stereoselectivity in the SN2′ reaction. J. Mol. Struct. 103, 197–208 (1983)
Borrmann, T. & Stohrer, W. What governs the stereochemistry of the SN2′ reaction? Liebigs Ann. 1996, 1593–1597 (1996)
Liu, C. & Widenhoefer, R. A. Gold(I)-catalyzed intramolecular enantioselective hydroarylation of allenes with indoles. Org. Lett. 9, 1935–1938 (2007)
Acknowledgements
We are grateful to D. Benitez and E. Tkatchouk (Goddard Group, California Institute of Technology) for performing DFT calculations, to U. Anderson and T. Iavarone of the QB3/Chemistry Mass Spectrometry Facility, and to A. DiPasquale of the X-ray Crystallography Facility. We acknowledge funding from the Director, Office of Science of the US Department of Energy (contract no. DE-AC02-05CH11231) and from NIHGMS (R01 GM074774). V.R. is grateful for a Natural Sciences and Engineering Research Council of Canada (NSERC) postdoctoral fellowship. G.L.H. is grateful for a Bristol-Myers/Squibb graduate fellowship.
Author information
Authors and Affiliations
Contributions
N.D.S. initiated the hydroamination study. V.R. optimized the catalysts and initiated the hydroarylation study. N.D.S., V.R., G.L.H. and J.W. performed the experiments. N.D.S., G.L.H. and F.D.T. developed the mechanistic concepts. G.L.H. and N.D.S. wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
X-ray crystallographic data have been deposited in the Cambridge Crystallographic Data Centre database (http://www.ccdc.cam.ac.uk/) under code CCDC 800545.
Supplementary information
Supplementary Information
The file contains Supplementary Figures 1-11 with legends, Supplementary Table 1, additional references and Supplementary data (Copies of NMR and HPLC spectra). (PDF 5334 kb)
PowerPoint slides
Rights and permissions
About this article
Cite this article
Shapiro, N., Rauniyar, V., Hamilton, G. et al. Asymmetric additions to dienes catalysed by a dithiophosphoric acid. Nature 470, 245–249 (2011). https://doi.org/10.1038/nature09723
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature09723
This article is cited by
-
Acidic graphene organocatalyst for the superior transformation of wastes into high-added-value chemicals
Nature Communications (2023)
-
Asymmetric dearomatization catalysed by chiral Brønsted acids via activation of ynamides
Nature Chemistry (2021)
-
Magnetic nanoparticles grafted l-carnosine dipeptide: remarkable catalytic activity in water at room temperature
Journal of the Iranian Chemical Society (2017)
-
Asymmetric spiroacetalization catalysed by confined Brønsted acids
Nature (2012)
-
The progression of chiral anions from concepts to applications in asymmetric catalysis
Nature Chemistry (2012)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.